haloalkanes - Knockhardy
... This form of nucleophilic substitution is known as SN2; it is a bimolecular process. An alternative method involves the initial breaking of the C-X bond to form a carbocation, or carbonium ion, (a unimolecular process - SN1 mechanism), which is then attacked by the nucleophile. SN1 is favoured for t ...
... This form of nucleophilic substitution is known as SN2; it is a bimolecular process. An alternative method involves the initial breaking of the C-X bond to form a carbocation, or carbonium ion, (a unimolecular process - SN1 mechanism), which is then attacked by the nucleophile. SN1 is favoured for t ...
- professional publication
... Reaction. Mechanism and Stereochemistry of S N2 reaction, Mechanism and Stereochemistry of SN1 Reaction. Rearrangement of Carbocation, S N2 versus SN1 Reactions, Reactivity of Alkyl Halides in SN1 and SN2, Factors Affecting SN1 and SN2. ...
... Reaction. Mechanism and Stereochemistry of S N2 reaction, Mechanism and Stereochemistry of SN1 Reaction. Rearrangement of Carbocation, S N2 versus SN1 Reactions, Reactivity of Alkyl Halides in SN1 and SN2, Factors Affecting SN1 and SN2. ...
Senior Science topics Programme
... Teachers may discuss with students how scientists combine basic chemical principles, understanding of the reactivity of different chemical species with experimental observations to deduce the most likely sequence of elementary steps and thus the mechanism of a particular reaction. Knowledge of react ...
... Teachers may discuss with students how scientists combine basic chemical principles, understanding of the reactivity of different chemical species with experimental observations to deduce the most likely sequence of elementary steps and thus the mechanism of a particular reaction. Knowledge of react ...
Elimination Reactions
... Explain how additions of water to an alkene and elimination of an alcohol are opposite mechanisms Describe how to shift equilibrium in favor of elimination or addition Predict the major product according to alkene stability Daily Problems 1. Provide a mechanism for these elimination reactions ...
... Explain how additions of water to an alkene and elimination of an alcohol are opposite mechanisms Describe how to shift equilibrium in favor of elimination or addition Predict the major product according to alkene stability Daily Problems 1. Provide a mechanism for these elimination reactions ...
Workshop 5
... K hydrogenates to form compound M. Give structures for compounds I-M. Explain your reasoning. ...
... K hydrogenates to form compound M. Give structures for compounds I-M. Explain your reasoning. ...
InorgCh14.1
... Ligand (CO) Substitution is important for synthesis of new complexes a) Rate is independent of incoming ligand = D mechanism (for most) Ni(CO)4 Ni(CO)3 18e- to 16e- (slow) Ni(CO)3 + L Ni(CO)3L 16e- to 18e- (fast) b) ...
... Ligand (CO) Substitution is important for synthesis of new complexes a) Rate is independent of incoming ligand = D mechanism (for most) Ni(CO)4 Ni(CO)3 18e- to 16e- (slow) Ni(CO)3 + L Ni(CO)3L 16e- to 18e- (fast) b) ...
SCH4U Organic Chemistry Portfolio Name: This portfolio is due on
... Write a polymerization reaction showing its production from monomers. What type of reaction is this? ...
... Write a polymerization reaction showing its production from monomers. What type of reaction is this? ...
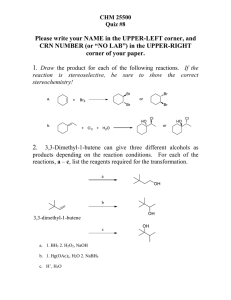
Chemistry 218, Winter 2007 Exam 2 Name: 1.
... 2. In the following reaction, one of the products is formed preferentially over the other one. Circle the product that is more likely to be formed, and explain why using resonance forms. (10 pts) O ...
... 2. In the following reaction, one of the products is formed preferentially over the other one. Circle the product that is more likely to be formed, and explain why using resonance forms. (10 pts) O ...
Types of Reactions in Organic Chemistry Chemistry
... Types of Reactions in Organic Chemistry The mechanism of a reaction is the detailed step-by-step description of how the overall reaction occurs. A substitution reaction is a chemical reaction in which an atom or group of atoms in a molecule is replaced by another atom or group of atoms. A substitut ...
... Types of Reactions in Organic Chemistry The mechanism of a reaction is the detailed step-by-step description of how the overall reaction occurs. A substitution reaction is a chemical reaction in which an atom or group of atoms in a molecule is replaced by another atom or group of atoms. A substitut ...
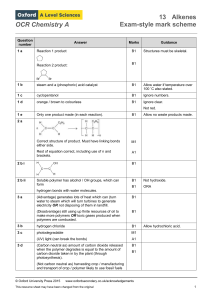
OCR Chemistry A Question number Answer Marks Guidance 1 a
... pair of electrons, so bond breaks when both electrons move to one of the bonded atoms. ...
... pair of electrons, so bond breaks when both electrons move to one of the bonded atoms. ...
Honors Chemistry Organic Chemistry
... 3. Which term doesn’t go with the others? (Circle one) alkyl group substituent division ...
... 3. Which term doesn’t go with the others? (Circle one) alkyl group substituent division ...
Lesson 4 halogenoalkanes
... This means the Rate Determining Step (RDS) must involve a bromoethane molecule and a hydroxide ion ...
... This means the Rate Determining Step (RDS) must involve a bromoethane molecule and a hydroxide ion ...
Chap Thirteen: Alcohols
... Predict the stereochemistry and optical activity of a product from an understanding of its mechanism of formation. Predict the acidity of alcohols relative to one another and relative to other functional groups Recognize structural features of a molecule that are key to its stability and react ...
... Predict the stereochemistry and optical activity of a product from an understanding of its mechanism of formation. Predict the acidity of alcohols relative to one another and relative to other functional groups Recognize structural features of a molecule that are key to its stability and react ...
Outline_CH13_Klein
... Predict the stereochemistry and optical activity of a product from an understanding of its mechanism of formation. Predict the acidity of alcohols relative to one another and relative to other functional groups Recognize structural features of a molecule that are key to its stability and react ...
... Predict the stereochemistry and optical activity of a product from an understanding of its mechanism of formation. Predict the acidity of alcohols relative to one another and relative to other functional groups Recognize structural features of a molecule that are key to its stability and react ...
Chapter 7: Structure and Synthesis of Alkenes
... Ú Procedure: A) assign the substituent a priority for each carbon of the double bond as: #1 and #2. – #1 is the highest priority and is determined by the atomic number (or molecular weight) of the atom directly attached to the C=C carbon B) if a priority cannot be determined using the atom directly ...
... Ú Procedure: A) assign the substituent a priority for each carbon of the double bond as: #1 and #2. – #1 is the highest priority and is determined by the atomic number (or molecular weight) of the atom directly attached to the C=C carbon B) if a priority cannot be determined using the atom directly ...
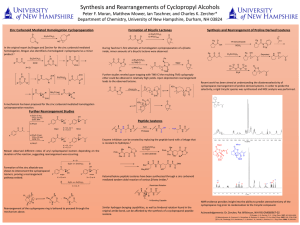
Here is the Original File - University of New Hampshire
... Recent work has been aimed at understanding the diastereoselectivity of cyclopropane rearrangement of proline-derived systems. In order to probe the selectivity, a rigid tricyclic species was synthesized and NOE analysis was performed. ...
... Recent work has been aimed at understanding the diastereoselectivity of cyclopropane rearrangement of proline-derived systems. In order to probe the selectivity, a rigid tricyclic species was synthesized and NOE analysis was performed. ...
Exam 1
... As mentioned in the text, diethyl ether, pentane, and 1-butanol have similar molar masses, but different physical properties. Boiling points are 35oC, 36oC, and 117oC, respectively. Their respective solubilities in water are 7.5g/100mL, insoluble, and 9g/100mL. (i) Draw structures for each of these ...
... As mentioned in the text, diethyl ether, pentane, and 1-butanol have similar molar masses, but different physical properties. Boiling points are 35oC, 36oC, and 117oC, respectively. Their respective solubilities in water are 7.5g/100mL, insoluble, and 9g/100mL. (i) Draw structures for each of these ...
CHEM 201 Name Quiz 10 (Ch 17) ID Q1. Which of the following
... the ester shown below with LiAlH4? O O ...
... the ester shown below with LiAlH4? O O ...
I (21 points) Complete the following reactions by providing starting
... A. (JOC 2008, ASAP, Hartung) The reagent 2-chloro-bis[menth-1-yloxycarbonyl]-1,3,2dioxaphospholane was shown to be useful in determining the enatiomeric purity of chiral alcohols via 31P NMR. The enatiomerically pure phosphorous reagent reacts with chiral alcohols to form diasteromers as shown below ...
... A. (JOC 2008, ASAP, Hartung) The reagent 2-chloro-bis[menth-1-yloxycarbonyl]-1,3,2dioxaphospholane was shown to be useful in determining the enatiomeric purity of chiral alcohols via 31P NMR. The enatiomerically pure phosphorous reagent reacts with chiral alcohols to form diasteromers as shown below ...
Tiffeneau–Demjanov rearrangement

The Tiffeneau–Demjanov rearrangement (TDR) is the chemical reaction of a 1-aminomethyl-cycloalkanol with nitrous acid to form an enlarged cycloketone.The Tiffeneau–Demjanov ring expansion, Tiffeneau–Demjanov rearrangement, or TDR, provides an easy way to increase amino-substituted cycloalkanes and cycloalkanols in size by one carbon. Ring sizes from cyclopropane through cyclooctane are able to undergo Tiffeneau–Demjanov ring expansion with some degree of success. Yields decrease as initial ring size increases, and the ideal use of TDR is for synthesis of five, six, and seven membered rings. A principal synthetic application of Tiffeneau–Demjanov ring expansion is to bicyclic or polycyclic systems. Several reviews on this reaction have been published.