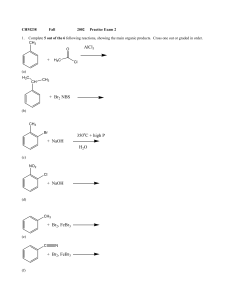
+ NaOH 350 C + high P H2O + H3C AlCl3 + NaOH + Br2, FeBr3
... (b) What is the reactive electrophile in the above reaction? ...
... (b) What is the reactive electrophile in the above reaction? ...
Survey on Conditions Catalysis of Chemical Reactions
... carbon of the substrate. It can also be used to reduce nitro groups and even as a nucleophile to displace halide from an sp3 carbon or open an epoxide. Why does it work? Remember that aluminum is a metal with a low electronegativity; thus, the Al-H bond is strongly polarized with Al positive and H n ...
... carbon of the substrate. It can also be used to reduce nitro groups and even as a nucleophile to displace halide from an sp3 carbon or open an epoxide. Why does it work? Remember that aluminum is a metal with a low electronegativity; thus, the Al-H bond is strongly polarized with Al positive and H n ...
Chem 174-Lecture 15a..
... P. Pauson: Fe(III) salts and cyclopentadiene S. A. Miller: Iron metal and cyclopentadiene at 300 oC ...
... P. Pauson: Fe(III) salts and cyclopentadiene S. A. Miller: Iron metal and cyclopentadiene at 300 oC ...
Nucleophilic Addition: The Grignard reagent
... To the reaction flask that contains the product from day one, add 6.0 mL of 6 M hydrochloric acid dropwise. Use a spatula to break up the solid and cap and shake the vial to help dissolve the solid. Because the left over magnesium will react with the acid to form hydrogen, and the neutralization rea ...
... To the reaction flask that contains the product from day one, add 6.0 mL of 6 M hydrochloric acid dropwise. Use a spatula to break up the solid and cap and shake the vial to help dissolve the solid. Because the left over magnesium will react with the acid to form hydrogen, and the neutralization rea ...
Alcohols - Miller, Jonathan
... (often larger molecular masses) formed when an alcohol reacts with a carboxylic acid in the presence of an acid catalyst (small amount of conc. H2SO4). It is an equilibrium reaction. ...
... (often larger molecular masses) formed when an alcohol reacts with a carboxylic acid in the presence of an acid catalyst (small amount of conc. H2SO4). It is an equilibrium reaction. ...
RULE
... Unexpected outcome - a 1,2-methyl shift (movement of CH3 with electrons) 2-chloro-2,3-dimethylbutane major product from Nu: addition to a 3° carbocation RULE - carbocations rearrangements can occur by CH3:- movement to an adjacent carbon atom (from C1 to C2 - should have been called methide shift, b ...
... Unexpected outcome - a 1,2-methyl shift (movement of CH3 with electrons) 2-chloro-2,3-dimethylbutane major product from Nu: addition to a 3° carbocation RULE - carbocations rearrangements can occur by CH3:- movement to an adjacent carbon atom (from C1 to C2 - should have been called methide shift, b ...
Year 13 Organic Chemistry Test
... Tollen's reagent. What would be seen in a positive test with Tollen's reagent? _____________________________________________________ _____________________________________________________ A second alcohol, 2-methyl propan-2-ol, will not give a positive breathalyzer test. Why not? ____________________ ...
... Tollen's reagent. What would be seen in a positive test with Tollen's reagent? _____________________________________________________ _____________________________________________________ A second alcohol, 2-methyl propan-2-ol, will not give a positive breathalyzer test. Why not? ____________________ ...
Chapter 13 – Alcohols, Phenols, Ethers, and Thioethers
... tend to stick together much more strongly than alkanes and this affects their physical properties. Before going further, please remember that the effects of the –OH group decrease as the alkane chain becomes longer. Thus, any hydrogen bonding effects in methanol are much larger than those in 1-penta ...
... tend to stick together much more strongly than alkanes and this affects their physical properties. Before going further, please remember that the effects of the –OH group decrease as the alkane chain becomes longer. Thus, any hydrogen bonding effects in methanol are much larger than those in 1-penta ...
Recall
... The functionality (OH) has remained at the end of the chain. We could make it even longer by repeating the above sequence. Note attack on less hindered carbon Now a substituted oxirane… ...
... The functionality (OH) has remained at the end of the chain. We could make it even longer by repeating the above sequence. Note attack on less hindered carbon Now a substituted oxirane… ...
Substitution Reactions of Alcohols
... is to convert the alcohol into a sulfonate ester. To make a sulfonate ester, you react an alcohol with an appropriate sulfonyl chloride in the presence of an amine ...
... is to convert the alcohol into a sulfonate ester. To make a sulfonate ester, you react an alcohol with an appropriate sulfonyl chloride in the presence of an amine ...
Converting Alcohols to Alkyl Halides – The Mitsunobu Reaction
... is to convert the alcohol into a sulfonate ester. To make a sulfonate ester, you react an alcohol with an appropriate sulfonyl chloride in the presence of an amine ...
... is to convert the alcohol into a sulfonate ester. To make a sulfonate ester, you react an alcohol with an appropriate sulfonyl chloride in the presence of an amine ...
enzymatic And Limited Industrial Use
... A reaction that can proceed in more than one way to produce different products involving different carbon atoms, where one predominates. It is said to be regioselective. ...
... A reaction that can proceed in more than one way to produce different products involving different carbon atoms, where one predominates. It is said to be regioselective. ...
Topic 16 Test - A
... How many structural isomers, which are aldehydes, have the molecular formula C5H10O? A ...
... How many structural isomers, which are aldehydes, have the molecular formula C5H10O? A ...
T. V. RajanBabu Chemistry, 730 Autumn 1997
... - How to calculate K from Go ( also see later under A values) Conformational analysis of cyclic compounds - cyclohexanes, Chair, boat, twist conformations - proof Substituted cyclohexanes - ‘A’ values of various groups - how to determine ‘A’ values Conformational effects on reactivity - oxidation o ...
... - How to calculate K from Go ( also see later under A values) Conformational analysis of cyclic compounds - cyclohexanes, Chair, boat, twist conformations - proof Substituted cyclohexanes - ‘A’ values of various groups - how to determine ‘A’ values Conformational effects on reactivity - oxidation o ...
730-2005 topics
... - How to calculate K from Go ( also see later under A values) Conformational analysis of cyclic compounds - cyclohexanes, Chair, boat, twist conformations - proof Substituted cyclohexanes - ‘A’ values of various groups - how to determine ‘A’ values Conformational effects on reactivity - oxidation o ...
... - How to calculate K from Go ( also see later under A values) Conformational analysis of cyclic compounds - cyclohexanes, Chair, boat, twist conformations - proof Substituted cyclohexanes - ‘A’ values of various groups - how to determine ‘A’ values Conformational effects on reactivity - oxidation o ...
Principles of Biochemistry 4/e
... different arrangements of their atoms in space • Enantiomers – 2 stereoisomers that are mirror images of one another • Diastereomers – 2 stereoisomers that are not mirror images of one another ...
... different arrangements of their atoms in space • Enantiomers – 2 stereoisomers that are mirror images of one another • Diastereomers – 2 stereoisomers that are not mirror images of one another ...
Lectures 15, 16 and 17
... developed these versatile reagents. • One B—H bond serves as the source of hydride in this reduction. • The (S)-CBS reagent delivers H:- from the front side of the C=O. This generally affords the R alcohol as the major product. • The (R)-CBS reagent delivers H:- from the back side of the C=O. This g ...
... developed these versatile reagents. • One B—H bond serves as the source of hydride in this reduction. • The (S)-CBS reagent delivers H:- from the front side of the C=O. This generally affords the R alcohol as the major product. • The (R)-CBS reagent delivers H:- from the back side of the C=O. This g ...
1. Absorption of what type electromagnetic radiation results in
... proceeds more rapidly than the nitration of benzene and yields predominantly the meta product. proceeds more rapidly than the nitration of benzene and yields predominantly the ortho, para products. proceeds more slowly than the nitration of benzene and yields predominantly the meta product. proceeds ...
... proceeds more rapidly than the nitration of benzene and yields predominantly the meta product. proceeds more rapidly than the nitration of benzene and yields predominantly the ortho, para products. proceeds more slowly than the nitration of benzene and yields predominantly the meta product. proceeds ...
Chapter 14 Selenium reagents
... base, giving alkyl bromides: the overall reaction is ROH RBr with retention of configuration. • Aryl alkyl selenides are preparable either (as above) from electrophilic selenium reagents and carbon ncleophiles or from nucleophilic selenium reagents, e.g. ArSe-Na+, and carbon electrophiles. On oxidat ...
... base, giving alkyl bromides: the overall reaction is ROH RBr with retention of configuration. • Aryl alkyl selenides are preparable either (as above) from electrophilic selenium reagents and carbon ncleophiles or from nucleophilic selenium reagents, e.g. ArSe-Na+, and carbon electrophiles. On oxidat ...
32 GRIGNARD REACTION Alkyl halides can react with magnesium
... Alkyl halides can react with magnesium to give a very versatile synthetic reagent, the Grignard reagent, for which Victor Grignard received the Nobel Prize in 1912. The Grignard reagent, RMgX, is one of the most useful reagents known. Although the carbon magnesium bond is covalent, it is highly pola ...
... Alkyl halides can react with magnesium to give a very versatile synthetic reagent, the Grignard reagent, for which Victor Grignard received the Nobel Prize in 1912. The Grignard reagent, RMgX, is one of the most useful reagents known. Although the carbon magnesium bond is covalent, it is highly pola ...
Chapter 1-
... The reaction is governed by the position of all the equilibria Hydration is favored by addition of a small amount of acid and a large amount of water Dehydration is favored by concentrated acid with very little water present (removal of water produced also helps favor dehydration) ...
... The reaction is governed by the position of all the equilibria Hydration is favored by addition of a small amount of acid and a large amount of water Dehydration is favored by concentrated acid with very little water present (removal of water produced also helps favor dehydration) ...
Chapter 18 - Aldehydes and Ketones
... example illustrating the need to activate the carbonyl by protonation. This reaction is not a very useful reaction in the forward process since the carbonyl hydrate will rearrange to the carbonyl. However, when a carbonyl hydrate is formed during a reaction, the resulting product will be the ketone ...
... example illustrating the need to activate the carbonyl by protonation. This reaction is not a very useful reaction in the forward process since the carbonyl hydrate will rearrange to the carbonyl. However, when a carbonyl hydrate is formed during a reaction, the resulting product will be the ketone ...
Experiment 7 – Dehydration of Methylcyclohexanols
... product. The regiospecificity of the reaction is dependent on Zaitsev’s rule, where the major product tends to be the more substituted alkene. When two different products are possible, these products are constitutional isomers of each other or in this case can be referred to as regioisomers. The typ ...
... product. The regiospecificity of the reaction is dependent on Zaitsev’s rule, where the major product tends to be the more substituted alkene. When two different products are possible, these products are constitutional isomers of each other or in this case can be referred to as regioisomers. The typ ...
Tiffeneau–Demjanov rearrangement

The Tiffeneau–Demjanov rearrangement (TDR) is the chemical reaction of a 1-aminomethyl-cycloalkanol with nitrous acid to form an enlarged cycloketone.The Tiffeneau–Demjanov ring expansion, Tiffeneau–Demjanov rearrangement, or TDR, provides an easy way to increase amino-substituted cycloalkanes and cycloalkanols in size by one carbon. Ring sizes from cyclopropane through cyclooctane are able to undergo Tiffeneau–Demjanov ring expansion with some degree of success. Yields decrease as initial ring size increases, and the ideal use of TDR is for synthesis of five, six, and seven membered rings. A principal synthetic application of Tiffeneau–Demjanov ring expansion is to bicyclic or polycyclic systems. Several reviews on this reaction have been published.























