organic chemistry ii
... exhaustive methylation: strategy to determine ring size glycoside formation ...
... exhaustive methylation: strategy to determine ring size glycoside formation ...
Organic Halides
... Hemiacetals can be converted acetals: Hemiacetal + alcohol +acid(catalyst) acetal + water An aldehyde dissolved in water exists in equilibrium with low concentrations of its hydrate, R-CH(OH)2. Similarly, in excess alcohol, the aldehyde, its hemiacetal, and its acetal all exist in solution. Hemiac ...
... Hemiacetals can be converted acetals: Hemiacetal + alcohol +acid(catalyst) acetal + water An aldehyde dissolved in water exists in equilibrium with low concentrations of its hydrate, R-CH(OH)2. Similarly, in excess alcohol, the aldehyde, its hemiacetal, and its acetal all exist in solution. Hemiac ...
Aromatic Compounds
... Treatment of aryl halides having strongly electronwithdrawing substituents (at the 2- and the 4-position) can also cause substitution reactions But… ...
... Treatment of aryl halides having strongly electronwithdrawing substituents (at the 2- and the 4-position) can also cause substitution reactions But… ...
Organic Chemistry - Centennial College Libraries
... Esters are compounds that contain –COO – between two alkyl groups. They are formed from a carboxylic acid and an alcohol. Amides are compounds that contain the H2NCOO— group. They are formed from ammonia and carboxylic ...
... Esters are compounds that contain –COO – between two alkyl groups. They are formed from a carboxylic acid and an alcohol. Amides are compounds that contain the H2NCOO— group. They are formed from ammonia and carboxylic ...
Chapter 1--Title
... Bonds to sp2-hybridized carbons are shorter, and therefore stronger, than to sp3-hybridized carbons Resonance gives the carbon-halogen bond some double bond character ...
... Bonds to sp2-hybridized carbons are shorter, and therefore stronger, than to sp3-hybridized carbons Resonance gives the carbon-halogen bond some double bond character ...
Rates of Hydrolysis of Some Halogeno-compounds
... hydrogen halide to form an alkene. An alkyne can be formed from the elimination reaction of an alkene. Both elimination and substitution are brought about by basic, electron-rich reagents. Hence there is always competition between the two types of reactions. Halobenzenes are comparatively unreactive ...
... hydrogen halide to form an alkene. An alkyne can be formed from the elimination reaction of an alkene. Both elimination and substitution are brought about by basic, electron-rich reagents. Hence there is always competition between the two types of reactions. Halobenzenes are comparatively unreactive ...
20130409085519
... The aldehyde carbon is always assigned as number one for referencing substituent positions in the name. Therefore, numbering is ...
... The aldehyde carbon is always assigned as number one for referencing substituent positions in the name. Therefore, numbering is ...
Named Reactions Of Haloalkanes and haloarenes
... 4)Swarts reaction– The synthesis of alkyl fluoride is best accomplished by heating n alkyl chloride in the presence of metallic fluoride such as AgF,Hg2F2,CoF2 ...
... 4)Swarts reaction– The synthesis of alkyl fluoride is best accomplished by heating n alkyl chloride in the presence of metallic fluoride such as AgF,Hg2F2,CoF2 ...
Dehydrating Cyclohexanol
... product was 1.4478 at 20oC and the percentage of error was 0.1245%. This meant that although cyclohexanol was still in the products, the percentage was quite small. The reason why the (NH4)2Ce(NO3)6 still turned red was that this test was so sensitive that even small contaminating amount of cyclohex ...
... product was 1.4478 at 20oC and the percentage of error was 0.1245%. This meant that although cyclohexanol was still in the products, the percentage was quite small. The reason why the (NH4)2Ce(NO3)6 still turned red was that this test was so sensitive that even small contaminating amount of cyclohex ...
Chemistry - Tiwariacademy.net
... Both the alkyl halides are primary. However, the substituent −CH 3 is at a greater distance to the carbon atom linked to Br in 1-bromo-3-methylbutane than in 1-bromo2methylbutane. Therefore, the approaching nucleophile is less hindered in case of the former than in case of the latter. Hence, the for ...
... Both the alkyl halides are primary. However, the substituent −CH 3 is at a greater distance to the carbon atom linked to Br in 1-bromo-3-methylbutane than in 1-bromo2methylbutane. Therefore, the approaching nucleophile is less hindered in case of the former than in case of the latter. Hence, the for ...
name Page 1 of 6 Multiple Choice. Choose the best answer for the
... Calculate the degrees of unsaturation for a molecule with the molecular formula C9H10FNO3 (a) (b) (c) (d) (e) ...
... Calculate the degrees of unsaturation for a molecule with the molecular formula C9H10FNO3 (a) (b) (c) (d) (e) ...
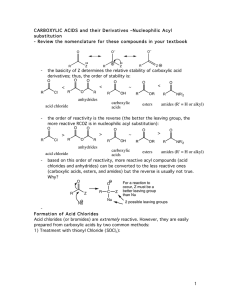
carboxylic acids esters amides (R
... So, remember...anhydrides react just about like acid chlorides. ...
... So, remember...anhydrides react just about like acid chlorides. ...
nucleophilic addition
... Formaldehyde is the most easily oxidized aldehyde. When mixed with another aldehyde that doesn’t have any alphahydrogens and conc. NaOH, all of the formaldehyde is oxidized and all of the other aldehyde is reduced. Crossed Cannizzaro: CH=O ...
... Formaldehyde is the most easily oxidized aldehyde. When mixed with another aldehyde that doesn’t have any alphahydrogens and conc. NaOH, all of the formaldehyde is oxidized and all of the other aldehyde is reduced. Crossed Cannizzaro: CH=O ...
CHEMISTRY 3.5 Paper 1 Describe the structure and reactions of
... acidic conditions, the amino acid forms an ion that will move towards one electrode. In basic conditions, it forms another ion that will move towards the other electrode. Explain how the conditions described above give rise to two ions that will move towards the two different electrodes and state wh ...
... acidic conditions, the amino acid forms an ion that will move towards one electrode. In basic conditions, it forms another ion that will move towards the other electrode. Explain how the conditions described above give rise to two ions that will move towards the two different electrodes and state wh ...
Seminar_1 1. Classification and nomenclature of organic
... Whilst many of the alkanes present in crude oil are aliphatic, having straight–or branched–chain molecules, some of them form rings – they are alicyclic. These compounds are denoted as cycloalkanes. These cycloalkanes have the general formula CnH2n instead of CnH2n+2 for the chain molecules. Cycloal ...
... Whilst many of the alkanes present in crude oil are aliphatic, having straight–or branched–chain molecules, some of them form rings – they are alicyclic. These compounds are denoted as cycloalkanes. These cycloalkanes have the general formula CnH2n instead of CnH2n+2 for the chain molecules. Cycloal ...
7. AS mechanisms
... To understand how the reaction proceeds we must first understand how bonds are broken in organic mechanisms There are two ways to break a covalent bond: ...
... To understand how the reaction proceeds we must first understand how bonds are broken in organic mechanisms There are two ways to break a covalent bond: ...
1 Carbonyl Condensation Reactions (Conjugate Addition) If we look
... Addition of a nucleophile to the double bond to form an enolate, which tautomerizes to the ketone, is called conjugate addition. When carbonyl addition or conjugate addition will occur? In other words, when will a nucleophile add to the carbonyl, or the double bond of a conjugated carbonyl compound? ...
... Addition of a nucleophile to the double bond to form an enolate, which tautomerizes to the ketone, is called conjugate addition. When carbonyl addition or conjugate addition will occur? In other words, when will a nucleophile add to the carbonyl, or the double bond of a conjugated carbonyl compound? ...
Sn1 and Sn2 Chemtivity
... group can undergo two types of reactions. They can undergo substitution reactions, in which the electronegative atom or group is replaced by another atom or group. They can also undergo elimination reactions, in which the electronegative atom or group is eliminated along with a hydrogen from an adja ...
... group can undergo two types of reactions. They can undergo substitution reactions, in which the electronegative atom or group is replaced by another atom or group. They can also undergo elimination reactions, in which the electronegative atom or group is eliminated along with a hydrogen from an adja ...
Organic Chemistry Unit Test! /50
... 1. Thalidomide has caused enormous grief, yet now it is a source of hope. Explain, using key terms and knowledge of organic chemistry, how thalidomide has gone from “zero to hero”. (4 marks) ...
... 1. Thalidomide has caused enormous grief, yet now it is a source of hope. Explain, using key terms and knowledge of organic chemistry, how thalidomide has gone from “zero to hero”. (4 marks) ...
Chapter 17 Allylic and Benzylic Reactivity
... Although the conjugate-base anion of 1,4-pentadiene is doubly allylic and resonance-stabilized, the conjugate-base anion of 1,3-cyclopentadiene is in addition aromatic. (See text p. 726 for a discussion of this case.) Consequently, much less energy is required for the ionization of 1,3-cyclopentadie ...
... Although the conjugate-base anion of 1,4-pentadiene is doubly allylic and resonance-stabilized, the conjugate-base anion of 1,3-cyclopentadiene is in addition aromatic. (See text p. 726 for a discussion of this case.) Consequently, much less energy is required for the ionization of 1,3-cyclopentadie ...
Chemistry 201 C Alkenes
... An alkane -- CnH2n+2 -- is fully saturated with hydrogens. A molecule is unsaturated when it contains fewer hydrogens (and/or halogens and oxygens) than an alkane of corresponding carbon content. The degree of unsaturation or index of hydrogen deficiency equals the number of H2 molecules it would ta ...
... An alkane -- CnH2n+2 -- is fully saturated with hydrogens. A molecule is unsaturated when it contains fewer hydrogens (and/or halogens and oxygens) than an alkane of corresponding carbon content. The degree of unsaturation or index of hydrogen deficiency equals the number of H2 molecules it would ta ...
Tiffeneau–Demjanov rearrangement

The Tiffeneau–Demjanov rearrangement (TDR) is the chemical reaction of a 1-aminomethyl-cycloalkanol with nitrous acid to form an enlarged cycloketone.The Tiffeneau–Demjanov ring expansion, Tiffeneau–Demjanov rearrangement, or TDR, provides an easy way to increase amino-substituted cycloalkanes and cycloalkanols in size by one carbon. Ring sizes from cyclopropane through cyclooctane are able to undergo Tiffeneau–Demjanov ring expansion with some degree of success. Yields decrease as initial ring size increases, and the ideal use of TDR is for synthesis of five, six, and seven membered rings. A principal synthetic application of Tiffeneau–Demjanov ring expansion is to bicyclic or polycyclic systems. Several reviews on this reaction have been published.