File
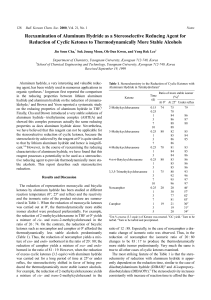
... strong signal at 1691 cm-1. Both signals represent vibration of the same kind of bond. Explain why the absorption in 2-cyclohexenone is at a lower wavenumber, including resonance structures. Both signals represent the IR stretching of the C=O bonds. 2-cyclohexenone has a lower wavenumber absorbance, ...
... strong signal at 1691 cm-1. Both signals represent vibration of the same kind of bond. Explain why the absorption in 2-cyclohexenone is at a lower wavenumber, including resonance structures. Both signals represent the IR stretching of the C=O bonds. 2-cyclohexenone has a lower wavenumber absorbance, ...
CHM 103 Lecture 28 S07
... • all form hydrogen bonds with water. • with 1-5 carbon atoms are soluble in water. ...
... • all form hydrogen bonds with water. • with 1-5 carbon atoms are soluble in water. ...
Carbohydrates: Occurrence, Structures and Chemistry
... Subdivisions are made according to functional groups which may also be present, for example, aminohexoses (C6H13O5N), deoxyhexoses (C6H12O5), and hexuronic acids (C6H10O7). Monosaccharides with fewer (trioses, tetroses) or more carbon atoms (heptoses, octoses, etc.) are rare. A large variety of mono ...
... Subdivisions are made according to functional groups which may also be present, for example, aminohexoses (C6H13O5N), deoxyhexoses (C6H12O5), and hexuronic acids (C6H10O7). Monosaccharides with fewer (trioses, tetroses) or more carbon atoms (heptoses, octoses, etc.) are rare. A large variety of mono ...
carbonyl compound group
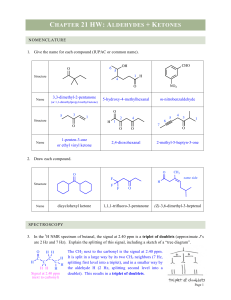
... (vi) Oxime: Oximes are a class of organic compounds having the general formula RR′CNOH, where R is an organic side chain and R′ is either hydrogen or an organic side chain. If R′ is H, then it is known as aldoxime and if R′ is an organic side chain, it is known as ketoxime. ...
... (vi) Oxime: Oximes are a class of organic compounds having the general formula RR′CNOH, where R is an organic side chain and R′ is either hydrogen or an organic side chain. If R′ is H, then it is known as aldoxime and if R′ is an organic side chain, it is known as ketoxime. ...
Chapter 19. Aldehydes and Ketones: Nucleophilic Addition Reactions
... generic name of the beet.] (Chem.) A nitrogenous base, {C5H11NO2}, produced artificially, and also occurring naturally in beet-root molasses and its residues. The listed pronunciation indicates it has the exact same emphasis as “cocaine”. Cocaine \Co"ca*ine\, n. (Chem.) A powerful alkaloid, {C17H2 ...
... generic name of the beet.] (Chem.) A nitrogenous base, {C5H11NO2}, produced artificially, and also occurring naturally in beet-root molasses and its residues. The listed pronunciation indicates it has the exact same emphasis as “cocaine”. Cocaine \Co"ca*ine\, n. (Chem.) A powerful alkaloid, {C17H2 ...
Chapter 19. Aldehydes and Ketones: Nucleophilic Addition Reactions
... generic name of the beet.] (Chem.) A nitrogenous base, {C5H11NO2}, produced artificially, and also occurring naturally in beet-root molasses and its residues. The listed pronunciation indicates it has the exact same emphasis as “cocaine”. Cocaine \Co"ca*ine\, n. (Chem.) A powerful alkaloid, {C17H2 ...
... generic name of the beet.] (Chem.) A nitrogenous base, {C5H11NO2}, produced artificially, and also occurring naturally in beet-root molasses and its residues. The listed pronunciation indicates it has the exact same emphasis as “cocaine”. Cocaine \Co"ca*ine\, n. (Chem.) A powerful alkaloid, {C17H2 ...
AMIDES AND AMINES: ORGANIC NITROGEN COMPOUNDS
... Amides: Unsubstituted amides (except formamide) are solids at room temperature. Many are odorless and colorless. Low molar-mass amides are water soluble. Solubility in water decreases as the molar mass increases. Amides are neutral compounds. The group is capable of hydrogen bonding. Amines: Low mol ...
... Amides: Unsubstituted amides (except formamide) are solids at room temperature. Many are odorless and colorless. Low molar-mass amides are water soluble. Solubility in water decreases as the molar mass increases. Amides are neutral compounds. The group is capable of hydrogen bonding. Amines: Low mol ...
13. Amines. - Mathematic.in
... unshared electron pair of nitrogen atom to be in conjugation with the benzene ring and thus making it less available for protonation. (ii) Methyl amine is a base and dissolves in water to produce hydroxide ions. ...
... unshared electron pair of nitrogen atom to be in conjugation with the benzene ring and thus making it less available for protonation. (ii) Methyl amine is a base and dissolves in water to produce hydroxide ions. ...
Chemistry - Andhra University
... – Baeyer’s strain theory, Sachse and Mohr predictions and Pitzer’s strain theory. Conformational structures of cyclobutane, cyclopentane, cyclohexane. 4. Benzene and its reactivity 7h Concept of resonance, resonance energy. Heat of hydrogenation, heat of combustion of Benezene, mention of C-C bond l ...
... – Baeyer’s strain theory, Sachse and Mohr predictions and Pitzer’s strain theory. Conformational structures of cyclobutane, cyclopentane, cyclohexane. 4. Benzene and its reactivity 7h Concept of resonance, resonance energy. Heat of hydrogenation, heat of combustion of Benezene, mention of C-C bond l ...
Alcohols phenols
... boiling point (b.p.) as compared to compounds of similar molecular weight. In a similar manner, molecules in the lower alcohols associate through H- bonding resulting in higher b.p. than expected. The solubility of lower alcohols in water may also be attributed to the formation of hydrogen bonds wit ...
... boiling point (b.p.) as compared to compounds of similar molecular weight. In a similar manner, molecules in the lower alcohols associate through H- bonding resulting in higher b.p. than expected. The solubility of lower alcohols in water may also be attributed to the formation of hydrogen bonds wit ...
CH3
... Much less POLAR than alcohols Slightly soluble in water More soluble than alkane Lower melting point (MP) and boiling point (BP) than water • Chemically inert • ALL are very flammable ...
... Much less POLAR than alcohols Slightly soluble in water More soluble than alkane Lower melting point (MP) and boiling point (BP) than water • Chemically inert • ALL are very flammable ...
Preparation of d, l-Phenylalanine by Amidocarbonylation of Benzyl
... some way. The effect seems general for amides. It was found that in the presence of a stoichiometric amount of DMF rather than acetamide, aldehydes were indeed formed, both from benzyl halides and alcohols under conditions of the amidocarbonylation reaction.10 The origins of this amide effect are un ...
... some way. The effect seems general for amides. It was found that in the presence of a stoichiometric amount of DMF rather than acetamide, aldehydes were indeed formed, both from benzyl halides and alcohols under conditions of the amidocarbonylation reaction.10 The origins of this amide effect are un ...
Brilliant Preparatory Section, Sitamarhi
... products. The skeletal equation is KMnO4 + HCl → KCl + MnCl2 + H2O + Cl2 If an element is present only one substance in the left hand side of the equation and if the same element is present only one of the substances in the right side, it may be taken up first while balancing the equation. According ...
... products. The skeletal equation is KMnO4 + HCl → KCl + MnCl2 + H2O + Cl2 If an element is present only one substance in the left hand side of the equation and if the same element is present only one of the substances in the right side, it may be taken up first while balancing the equation. According ...
Catalytic Nucleophilic Fluorination of Secondary and Tertiary
... that the copper–N-heterocyclic carbene (NHC) complex [(IPr)CuCl] catalyzed the formation of fluoride 2 a in 42 % yield, with 33 % starting material remaining. Despite this breakthrough, elimination (15 % yield) was an alarming drawback. Control experiments demonstrated that enyne 3 a was not formed ...
... that the copper–N-heterocyclic carbene (NHC) complex [(IPr)CuCl] catalyzed the formation of fluoride 2 a in 42 % yield, with 33 % starting material remaining. Despite this breakthrough, elimination (15 % yield) was an alarming drawback. Control experiments demonstrated that enyne 3 a was not formed ...
Organic Synthesis II
... Mechanisms for many oxidation reactions (even well-known ones) are significantly more complex than drawn throughout this course (and in many cases are not known or understood). Some are based on factual mechanistic data; some should be treated more as a mnemonic than explanation. ...
... Mechanisms for many oxidation reactions (even well-known ones) are significantly more complex than drawn throughout this course (and in many cases are not known or understood). Some are based on factual mechanistic data; some should be treated more as a mnemonic than explanation. ...
Fragmentations Associated with Organic Functional Groups
... It is known that fragmentation of n-alkyl ions occurs by loss of ethene (C2H4), so that the ion of m/z 57 in the mass spectrum of n-octane, above, is probably derived from two pathways, as shown below. ...
... It is known that fragmentation of n-alkyl ions occurs by loss of ethene (C2H4), so that the ion of m/z 57 in the mass spectrum of n-octane, above, is probably derived from two pathways, as shown below. ...
Slide 1
... Note that the nucleophile is NOT alkoxide! Like water, alcohols are not strong acids, so there is no R-O- present. The nucleophile is the whole alcohol molecule., ...
... Note that the nucleophile is NOT alkoxide! Like water, alcohols are not strong acids, so there is no R-O- present. The nucleophile is the whole alcohol molecule., ...
Alcohols, Phenols, and Thiols
... The functional group of alcohols and phenols is the hydroxyl group. In alcohols, this group is connected to an aliphatic carbon, whereas in phenols, it is attached to an aromatic ring. In the IUPAC system of nomenclature, the suffix for alcohols is -ol. Alcohols are classified as primary, secondary, ...
... The functional group of alcohols and phenols is the hydroxyl group. In alcohols, this group is connected to an aliphatic carbon, whereas in phenols, it is attached to an aromatic ring. In the IUPAC system of nomenclature, the suffix for alcohols is -ol. Alcohols are classified as primary, secondary, ...
Unit 5: Oragnic Chemistry Notes (answers)
... any effect on their physical properties. 3. They do give off more heat (more exothermic) when burned (combusted) compare to alkanes. This is because there are more energy stored in the double and triple bonds. When this energy is released during a chemical reaction, more heat is given off. Combustio ...
... any effect on their physical properties. 3. They do give off more heat (more exothermic) when burned (combusted) compare to alkanes. This is because there are more energy stored in the double and triple bonds. When this energy is released during a chemical reaction, more heat is given off. Combustio ...
boehm_rl
... ethyl methylchloroacetate, ethyl phenylchloroacetate, and ethyl diphenylchloroacetate from the corresponding alcohols. Bissinger and Kung (32) studied the effects of varying experimental conditions upon the reactions of propyl alcohols and thionyl chloride . In order to prepare unstable propyl sulfi ...
... ethyl methylchloroacetate, ethyl phenylchloroacetate, and ethyl diphenylchloroacetate from the corresponding alcohols. Bissinger and Kung (32) studied the effects of varying experimental conditions upon the reactions of propyl alcohols and thionyl chloride . In order to prepare unstable propyl sulfi ...
Reexamination of Aluminum Hydride as a Stereoselective Reducing
... the Presence of 2-methylcyclohexanone. The following procedure was used to convert cis-2-methylcyclohexanol to trans-2-methylcyclohexanol. Into a 50 mL flask, dried thoroughly as described above, 5.0 mL of a 2.0 M solution of aluminum hydride in THF (10 mmol) was introduced and the flask was cooled ...
... the Presence of 2-methylcyclohexanone. The following procedure was used to convert cis-2-methylcyclohexanol to trans-2-methylcyclohexanol. Into a 50 mL flask, dried thoroughly as described above, 5.0 mL of a 2.0 M solution of aluminum hydride in THF (10 mmol) was introduced and the flask was cooled ...
Chapter 23 SG5e
... The stereochemistry of the product will be determined by the stereochemistry of the starting material; if the product is racemic, the starting material must have been racemic as well. Problem 23.11 How might you bring about this conversion? OH CH3 NO2 , KOH H2 ,Pt O ethanol CH2 NO2 (Aldol reaction) ...
... The stereochemistry of the product will be determined by the stereochemistry of the starting material; if the product is racemic, the starting material must have been racemic as well. Problem 23.11 How might you bring about this conversion? OH CH3 NO2 , KOH H2 ,Pt O ethanol CH2 NO2 (Aldol reaction) ...
NCERT/CBSE CHEMISTRY CLASS 12 textbook
... C2H5NH2, (C2H5)2NH, (C2H5)3N and NH3 In gas phase there is no hydrogen bonding , therefore stabalisation due to hydrogen bonding is not there. Therfore the only effect to determine the strength is the inductive effect.The +I effect increases with increase in the alkyl group.Therefore the basic stren ...
... C2H5NH2, (C2H5)2NH, (C2H5)3N and NH3 In gas phase there is no hydrogen bonding , therefore stabalisation due to hydrogen bonding is not there. Therfore the only effect to determine the strength is the inductive effect.The +I effect increases with increase in the alkyl group.Therefore the basic stren ...
Lectures 4-6
... Collins Oxidation (CrO3 • 2pyridine) TL 1969, 336 - CrO3 (anhydrous) + pyridine (anhydrous) ...
... Collins Oxidation (CrO3 • 2pyridine) TL 1969, 336 - CrO3 (anhydrous) + pyridine (anhydrous) ...
Haloalkane

The haloalkanes (also known, as halogenoalkanes or alkyl halides) are a group of chemical compounds derived from alkanes containing one or more halogens. They are a subset of the general class of halocarbons, although the distinction is not often made. Haloalkanes are widely used commercially and, consequently, are known under many chemical and commercial names. They are used as flame retardants, fire extinguishants, refrigerants, propellants, solvents, and pharmaceuticals. Subsequent to the widespread use in commerce, many halocarbons have also been shown to be serious pollutants and toxins. For example, the chlorofluorocarbons have been shown to lead to ozone depletion. Methyl bromide is a controversial fumigant. Only haloalkanes which contain chlorine, bromine, and iodine are a threat to the ozone layer, but fluorinated volatile haloalkanes in theory may have activity as greenhouse gases. Methyl iodide, a naturally occurring substance, however, does not have ozone-depleting properties and the United States Environmental Protection Agency has designated the compound a non-ozone layer depleter. For more information, see Halomethane. Haloalkane or alkyl halides are the compounds which have the general formula ″RX″ where R is an alkyl or substituted alkyl group and X is a halogen (F, Cl, Br, I).Haloalkanes have been known for centuries. Chloroethane was produced synthetically in the 15th century. The systematic synthesis of such compounds developed in the 19th century in step with the development of organic chemistry and the understanding of the structure of alkanes. Methods were developed for the selective formation of C-halogen bonds. Especially versatile methods included the addition of halogens to alkenes, hydrohalogenation of alkenes, and the conversion of alcohols to alkyl halides. These methods are so reliable and so easily implemented that haloalkanes became cheaply available for use in industrial chemistry because the halide could be further replaced by other functional groups.While most haloalkanes are human-produced, non-artificial-source haloalkanes do occur on Earth, mostly through enzyme-mediated synthesis by bacteria, fungi, and especially sea macroalgae (seaweeds). More than 1600 halogenated organics have been identified, with bromoalkanes being the most common haloalkanes. Brominated organics in biology range from biologically produced methyl bromide to non-alkane aromatics and unsaturates (indoles, terpenes, acetogenins, and phenols). Halogenated alkanes in land plants are more rare, but do occur, as for example the fluoroacetate produced as a toxin by at least 40 species of known plants. Specific dehalogenase enzymes in bacteria which remove halogens from haloalkanes, are also known.