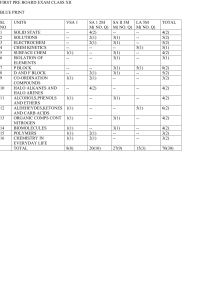
support guide for chemistry 1 south carolina academic standards
... number, a predictable pattern for the addition of electrons exists. This pattern is the basis for the arrangement of elements in the periodic table. The chemical properties of an element are determined by an element’s electron configuration. Elements can react to form chemical compounds/molecules th ...
... number, a predictable pattern for the addition of electrons exists. This pattern is the basis for the arrangement of elements in the periodic table. The chemical properties of an element are determined by an element’s electron configuration. Elements can react to form chemical compounds/molecules th ...
Proton affinities of candidates for positively charged ambient ions in
... Due to limitations in computational resources, we were unable to obtain a result for the PA of pyridine with the W1BD method. In the cases of methylamine, dimethylamine and trimethylamine, we compared all other methods with the W1BD. In the case of pyridine, all results were compared to the PA value ...
... Due to limitations in computational resources, we were unable to obtain a result for the PA of pyridine with the W1BD method. In the cases of methylamine, dimethylamine and trimethylamine, we compared all other methods with the W1BD. In the case of pyridine, all results were compared to the PA value ...
File - cpprashanths Chemistry
... b) Medicines are more effective in colloidal state. c) Alum is added to purify muddy water a) Because the particle size is so small that no scattering of light is possible. 1M b) A colloidal state has a larger surface area. Thus medicines in colloidal state are effectively adsorbed and assimilated a ...
... b) Medicines are more effective in colloidal state. c) Alum is added to purify muddy water a) Because the particle size is so small that no scattering of light is possible. 1M b) A colloidal state has a larger surface area. Thus medicines in colloidal state are effectively adsorbed and assimilated a ...
Electronic structure of molecular van der Waals complexes with
... to describe the tunneling through various flat-lying alkyl derivatives on HOPG under typically used experimental conditions.4–16 Electrons from a metallic STM-tip tunnel through a molecule into the conducting substrate, or vice versa. A schematic energy diagram is displayed in Fig. 1. The workfuncti ...
... to describe the tunneling through various flat-lying alkyl derivatives on HOPG under typically used experimental conditions.4–16 Electrons from a metallic STM-tip tunnel through a molecule into the conducting substrate, or vice versa. A schematic energy diagram is displayed in Fig. 1. The workfuncti ...
c8h18 isomers
... 9) If a substituent of the main chain has sub-branching, name the substituent by applying previous rules and begin numbering at the point of attachment. The complex substituent is set off in parenthesis when writing the complete name of the compound. ...
... 9) If a substituent of the main chain has sub-branching, name the substituent by applying previous rules and begin numbering at the point of attachment. The complex substituent is set off in parenthesis when writing the complete name of the compound. ...
Chapter 21
... 2. overlap of the parallel 2p orbitals forms one torus above the plane of the ring and another below it 3. this orbital represents the lowest-lying pi-bonding molecular orbital ...
... 2. overlap of the parallel 2p orbitals forms one torus above the plane of the ring and another below it 3. this orbital represents the lowest-lying pi-bonding molecular orbital ...
Course No - Chemistry
... (16 Contact Hours) Localised and delocalised chemical bonds: Recapitulation and applications of Inductive and Electromeric effects. Conjugation, Resonance and Hyperconjugation. Dipole-dipole interactions: Hydrogen bond, Van der Waal’s and London forces and their significance with respect to organic ...
... (16 Contact Hours) Localised and delocalised chemical bonds: Recapitulation and applications of Inductive and Electromeric effects. Conjugation, Resonance and Hyperconjugation. Dipole-dipole interactions: Hydrogen bond, Van der Waal’s and London forces and their significance with respect to organic ...
No Slide Title
... Functional Group different functional group different chemical properties different physical properties • Sometimes more than one type of isomerism occurs in the same molecule. • The more carbon atoms there are, the greater the number of possible isomers ...
... Functional Group different functional group different chemical properties different physical properties • Sometimes more than one type of isomerism occurs in the same molecule. • The more carbon atoms there are, the greater the number of possible isomers ...
Isomerism - Knockhardy
... Functional Group different functional group different chemical properties different physical properties • Sometimes more than one type of isomerism occurs in the same molecule. • The more carbon atoms there are, the greater the number of possible isomers ...
... Functional Group different functional group different chemical properties different physical properties • Sometimes more than one type of isomerism occurs in the same molecule. • The more carbon atoms there are, the greater the number of possible isomers ...
The Molecular Structure of Bismuth Oxide by
... strengths, and overall symmetry of metal oxide species. This is not only true for the more common crystalline and solution phases (17), but also for the exotic twodimensional surface phases (18,19). The basic idea behind the Raman analysis is that different molecular structures typically have differ ...
... strengths, and overall symmetry of metal oxide species. This is not only true for the more common crystalline and solution phases (17), but also for the exotic twodimensional surface phases (18,19). The basic idea behind the Raman analysis is that different molecular structures typically have differ ...
Chemistry 3.5 - CashmereChemistry
... morning sickness in early pregnancy in the 1960s, tragically caused the development of serious birth defects (badly deformed limbs, or none at all). In fact, only one of the two optical isomers of thalidomide appear to cause these birth defects, although it could be that once ingested each isomer re ...
... morning sickness in early pregnancy in the 1960s, tragically caused the development of serious birth defects (badly deformed limbs, or none at all). In fact, only one of the two optical isomers of thalidomide appear to cause these birth defects, although it could be that once ingested each isomer re ...
(MgCl2 and CaCl2): Osmotic Pressure Calculations
... charged model mentioned above. An external constraint placed between a Ca2+ ion and oxygen atoms in a theoretical study of Ca2+ binding with Parvalbumin provided evidence of how the charge in the center of the ion by itself is not sufficient to keep the Ca2+ ion stable in its binding site.34 Another t ...
... charged model mentioned above. An external constraint placed between a Ca2+ ion and oxygen atoms in a theoretical study of Ca2+ binding with Parvalbumin provided evidence of how the charge in the center of the ion by itself is not sufficient to keep the Ca2+ ion stable in its binding site.34 Another t ...
CO 2 - TrimbleChemistry
... • A compound is represented by using the symbols for the elements of which it is composed • Subscripts are used to indicate how many atoms of a particular element exist in the compound • If there is only one atom of a particular element, the one is assumed ...
... • A compound is represented by using the symbols for the elements of which it is composed • Subscripts are used to indicate how many atoms of a particular element exist in the compound • If there is only one atom of a particular element, the one is assumed ...
more aromatic chemistry
... electron-withdrawing groups on the ring. Use a very strong base like NaNH2. ...
... electron-withdrawing groups on the ring. Use a very strong base like NaNH2. ...
Jahn−Teller Distortion in the Phosphorescent Excited State of Three
... Luminescent transition metal complexes have been receiving increasing attention due to their role in a variety of optoelectronic applications. For example, molecular light-emitting devices (LEDs) have been increasingly reported1 to exhibit superior efficiencies when phosphorescence is enhanced by th ...
... Luminescent transition metal complexes have been receiving increasing attention due to their role in a variety of optoelectronic applications. For example, molecular light-emitting devices (LEDs) have been increasingly reported1 to exhibit superior efficiencies when phosphorescence is enhanced by th ...
Chapter - Imperial Valley College
... Types of Formula Structural Formula • Structural Formula describe the kinds of elements found in the compound, the numbers of their atoms, order of atom attachment, and the kind of attachment they do not directly describe the 3-dimensional shape, but an experienced chemist can make a good guess a ...
... Types of Formula Structural Formula • Structural Formula describe the kinds of elements found in the compound, the numbers of their atoms, order of atom attachment, and the kind of attachment they do not directly describe the 3-dimensional shape, but an experienced chemist can make a good guess a ...
Chapter 25 Organic and Biological Chemistry
... Reactions of Aromatic Compounds • Unlike in alkenes and alkynes, electrons do not sit between two atoms. • Electrons are delocalized; this stabilizes aromatic compounds. Organic and Biological Chemistry ...
... Reactions of Aromatic Compounds • Unlike in alkenes and alkynes, electrons do not sit between two atoms. • Electrons are delocalized; this stabilizes aromatic compounds. Organic and Biological Chemistry ...
Homoaromaticity

Homoaromaticity in organic chemistry refers to a special case of aromaticity in which conjugation is interrupted by a single sp3 hybridized carbon atom. Although this sp3 center disrupts the continuous overlap of p-orbitals, traditionally thought to be a requirement for aromaticity, considerable thermodynamic stability and many of the spectroscopic, magnetic, and chemical properties associated with aromatic compounds are still observed for such compounds. This formal discontinuity is apparently bridged by p-orbital overlap, maintaining a contiguous cycle of π electrons that is responsible for this preserved chemical stability.The concept of homoaromaticity was pioneered by Saul Winstein in 1959, prompted by his studies of the “tris-homocyclopropenyl” cation. Since the publication of Winstein's paper, much research has been devoted to understanding and classifying these molecules, which represent an additional “class” of aromatic molecules included under the continuously broadening definition of aromaticity. To date, homoaromatic compounds are known to exist as cationic and anionic species, and some studies support the existence of neutral homoaromatic molecules, though these are less common. The 'homotropylium' cation (C8H9+) is perhaps the best studied example of a homoaromatic compound.