II. Nomenclature Rules For Alkenes 1. The parent name will be the
... Many commonly encountered functional groups in organic chemistry are discussed below. An initial “bare bones” list shows the functional groups in their relative priorities from top to bottom (highest priority to lowest priority). A very brief overview of their nomenclature is provided showing the pr ...
... Many commonly encountered functional groups in organic chemistry are discussed below. An initial “bare bones” list shows the functional groups in their relative priorities from top to bottom (highest priority to lowest priority). A very brief overview of their nomenclature is provided showing the pr ...
Organic and Biochemical Molecules
... Geometric isomers of alkenes – If each sp2 C has 2 different groups attached it will have two possible orientations due the fact that pi bonds do not allow for free rotation – If each sp2 C has only one hydrogen you can classify the structure as cis or trans ...
... Geometric isomers of alkenes – If each sp2 C has 2 different groups attached it will have two possible orientations due the fact that pi bonds do not allow for free rotation – If each sp2 C has only one hydrogen you can classify the structure as cis or trans ...
McMurray-Fay Chapter 22 Presentation Slides
... • Write the name as a single word. Use hyphens to separate the different prefixes, and use commas to separate numbers when there are more than one. If two or more different substituent groups are present, list them in alphabetical order. If two or more identical substituent groups are present, use o ...
... • Write the name as a single word. Use hyphens to separate the different prefixes, and use commas to separate numbers when there are more than one. If two or more different substituent groups are present, list them in alphabetical order. If two or more identical substituent groups are present, use o ...
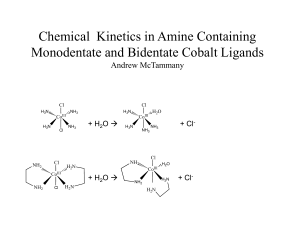
Chemical Kinetics in Monodentate and Bidentate Cobalt Compounds
... repeated. The trans-Co(NH3)4Cl2 should be synthesized using a lower temperature and lower concentration of acid. A mixture of HCl and H2SO4 could be used instead. From there the effect of different ligands can be evaluated. ...
... repeated. The trans-Co(NH3)4Cl2 should be synthesized using a lower temperature and lower concentration of acid. A mixture of HCl and H2SO4 could be used instead. From there the effect of different ligands can be evaluated. ...
Mission Statement
... rearrangement of electrons Understand the Bohr model of the atom Understand how line emission spectra are formed ...
... rearrangement of electrons Understand the Bohr model of the atom Understand how line emission spectra are formed ...
Chapter 7 Carbohydrates - Angelo State University
... • Chiral objects cannot be superimposed on their mirror images — e.g., hands, gloves, and shoes. • Achiral objects can be superimposed on the mirror images — e.g., drinking glasses, spheres, and cubes. • Any carbon atom which is connected to four different groups will be chiral, and will have two no ...
... • Chiral objects cannot be superimposed on their mirror images — e.g., hands, gloves, and shoes. • Achiral objects can be superimposed on the mirror images — e.g., drinking glasses, spheres, and cubes. • Any carbon atom which is connected to four different groups will be chiral, and will have two no ...
RELATIONSHIP BETWEEN Sn1 and E1 REACTIONS
... H Why is the trisubstituted alkene more stable than the other two? The relative stabilities of alkenes is measured by their heat of hydrogenation (much like the relative stabilities of cycloalkanes is measured by their heats of combustion, see ch. 3). The relationship is inverse: the higher the heat ...
... H Why is the trisubstituted alkene more stable than the other two? The relative stabilities of alkenes is measured by their heat of hydrogenation (much like the relative stabilities of cycloalkanes is measured by their heats of combustion, see ch. 3). The relationship is inverse: the higher the heat ...
New trends in the investigations of macrocyclic magnets J. M , A. T
... The growing need for macrocyclic ligands and their complexes has stimulated research efforts for methods of their preparation and for studying their interesting chemical and physical properties. In particular, polynuclear systems are interesting because of their magnetic interaction between metallic ...
... The growing need for macrocyclic ligands and their complexes has stimulated research efforts for methods of their preparation and for studying their interesting chemical and physical properties. In particular, polynuclear systems are interesting because of their magnetic interaction between metallic ...
Tro Ch 3 Lecture PP - Highline Community College
... The empirical formula for the molecular compound oxalic acid is CHO2. This means that there is 1 C atom and 1 H atom for every 2 O atoms in the molecule. The actual molecular formula is C2H2O4. Tro: Chemistry: A Molecular Approach, 2/e ...
... The empirical formula for the molecular compound oxalic acid is CHO2. This means that there is 1 C atom and 1 H atom for every 2 O atoms in the molecule. The actual molecular formula is C2H2O4. Tro: Chemistry: A Molecular Approach, 2/e ...
Incoherent transport through molecules on silicon in the vicinity of a
... technology but also offer a unique platform to study scientific phenomena3,4 with experimental reproducibility. Notably, Si contacts may give rise to novel features such as negative differential resistance,5–7 although the mechanism is not yet established.8 In Si technology, dangling bond 共DB兲 defec ...
... technology but also offer a unique platform to study scientific phenomena3,4 with experimental reproducibility. Notably, Si contacts may give rise to novel features such as negative differential resistance,5–7 although the mechanism is not yet established.8 In Si technology, dangling bond 共DB兲 defec ...
Document
... OH group-containing compounds (8) are by far the most important reactants for isocyanates. They are added under mild conditions, forming carbamic esters (9). Primary alcohols, secondary alcohols, and phenols show decreasing reactivity in that order. ...
... OH group-containing compounds (8) are by far the most important reactants for isocyanates. They are added under mild conditions, forming carbamic esters (9). Primary alcohols, secondary alcohols, and phenols show decreasing reactivity in that order. ...
Matter and Measurement
... carbon chain to indicate the position of the substituent, the C atoms in the longest chain are numbered, starting at the end that will give the lowest number for the position of the first attached group ...
... carbon chain to indicate the position of the substituent, the C atoms in the longest chain are numbered, starting at the end that will give the lowest number for the position of the first attached group ...
Proton Chemical Shift Tensors and Hydrogen Bond Geometry: A 1H
... chemistry, biochemistry, and biology.1-5 Due to its versatility and applicability to molecular systems in all condensed phases, nuclear magnetic resonance (NMR) spectroscopy is commonly used for studying hydrogen-bonding phenomena. The early discovery of the effect of hydrogen bonding on 1H chemical ...
... chemistry, biochemistry, and biology.1-5 Due to its versatility and applicability to molecular systems in all condensed phases, nuclear magnetic resonance (NMR) spectroscopy is commonly used for studying hydrogen-bonding phenomena. The early discovery of the effect of hydrogen bonding on 1H chemical ...
Ca(ii), Cd(ii), Cu(ii) and Pb(ii)
... Electrospray ionization mass spectrometry (ESI-MS)1–4 is currently an effective tool for analysis of a wide variety of noncovalent complexes,5–16 such as those formed in host–guest chemistry. Many studies show that equilibrium distribution of complexes in solution is reflected in the intensities of ...
... Electrospray ionization mass spectrometry (ESI-MS)1–4 is currently an effective tool for analysis of a wide variety of noncovalent complexes,5–16 such as those formed in host–guest chemistry. Many studies show that equilibrium distribution of complexes in solution is reflected in the intensities of ...
enzymatic And Limited Industrial Use
... A stereoselective reaction is one in which a single starting material can yield two or more stereoisomeric products, but one is produced in greater amounts than any other. ...
... A stereoselective reaction is one in which a single starting material can yield two or more stereoisomeric products, but one is produced in greater amounts than any other. ...
CH 2 CH(CH 3 ) - Parkway C-2
... – CH3, -CH2-CH3, -Cl, etc. If there is a tie, start counting from the end that has the most groups on the lowest number ...
... – CH3, -CH2-CH3, -Cl, etc. If there is a tie, start counting from the end that has the most groups on the lowest number ...
Chapter 4
... Not to be confused with isotopes, structural isomers are molecules with the same molecular formula, but there atoms are connected differently (Different connectivity) resulting in different structural formula. Structure determines function and therefore structural isomers function or behave differen ...
... Not to be confused with isotopes, structural isomers are molecules with the same molecular formula, but there atoms are connected differently (Different connectivity) resulting in different structural formula. Structure determines function and therefore structural isomers function or behave differen ...
Chapter 3 An Introduction to Organic Compounds
... In a Newman projection, you are looking down the length of a particular C-C bond. ...
... In a Newman projection, you are looking down the length of a particular C-C bond. ...
Organic Chemistry
... 15.2 Acid Halides (R-C(=O)-X) When Z of R-C(=O)-Z is a halogen atom (X), these compounds are equivalently referred to as acid halides, acyl halides, or alkanoyl halides. Preparation, Reactivity, and Properties (15.2A) The halogen atom of acid halides may be F, Cl, Br, or I, however acid chlorides (X ...
... 15.2 Acid Halides (R-C(=O)-X) When Z of R-C(=O)-Z is a halogen atom (X), these compounds are equivalently referred to as acid halides, acyl halides, or alkanoyl halides. Preparation, Reactivity, and Properties (15.2A) The halogen atom of acid halides may be F, Cl, Br, or I, however acid chlorides (X ...
Solvent effects on nitrogen NMR shieldings in thiazole and
... shielding, a represents the hydrogen bond donor strength of the solvent, p gives its hydrogen bond acceptor strength, n* is its polarity/polarizability and 6 is a correction for polychlorinated solvents (S = 0.5) and aromatic solvents (6 = 1). The corresponding response of the solute nitrogen shield ...
... shielding, a represents the hydrogen bond donor strength of the solvent, p gives its hydrogen bond acceptor strength, n* is its polarity/polarizability and 6 is a correction for polychlorinated solvents (S = 0.5) and aromatic solvents (6 = 1). The corresponding response of the solute nitrogen shield ...
Multinuclear NMR as a tool for studying local order and
... previously reported in the literature. Low temperature deuterium NMR measurements, down to 25K, were carried out to investigate the structural phase transitions of MAPbBr3. Spectral lineshapes allow following the successive phase transitions of MAPbBr3. Finally, quadrupolar NMR ...
... previously reported in the literature. Low temperature deuterium NMR measurements, down to 25K, were carried out to investigate the structural phase transitions of MAPbBr3. Spectral lineshapes allow following the successive phase transitions of MAPbBr3. Finally, quadrupolar NMR ...
6. Low valent of Vanadium catalyst in organic synthesis
... *the coordination of the phosphorus raises the reduction capability and selectivity. *the bulky reductant is liable to approach the bromide from the ...
... *the coordination of the phosphorus raises the reduction capability and selectivity. *the bulky reductant is liable to approach the bromide from the ...
Homoaromaticity

Homoaromaticity in organic chemistry refers to a special case of aromaticity in which conjugation is interrupted by a single sp3 hybridized carbon atom. Although this sp3 center disrupts the continuous overlap of p-orbitals, traditionally thought to be a requirement for aromaticity, considerable thermodynamic stability and many of the spectroscopic, magnetic, and chemical properties associated with aromatic compounds are still observed for such compounds. This formal discontinuity is apparently bridged by p-orbital overlap, maintaining a contiguous cycle of π electrons that is responsible for this preserved chemical stability.The concept of homoaromaticity was pioneered by Saul Winstein in 1959, prompted by his studies of the “tris-homocyclopropenyl” cation. Since the publication of Winstein's paper, much research has been devoted to understanding and classifying these molecules, which represent an additional “class” of aromatic molecules included under the continuously broadening definition of aromaticity. To date, homoaromatic compounds are known to exist as cationic and anionic species, and some studies support the existence of neutral homoaromatic molecules, though these are less common. The 'homotropylium' cation (C8H9+) is perhaps the best studied example of a homoaromatic compound.