Selective Incorporation of Difluoromethylene
... physical, and biological properties. Accordingly, the incorporation of fluorine atom(s) or fluorine-containing functionalities into organic molecules has received considerable attention. For decades, extensive reports and reviews mainly focused on the fluorination and perfluoroalkylation (especially ...
... physical, and biological properties. Accordingly, the incorporation of fluorine atom(s) or fluorine-containing functionalities into organic molecules has received considerable attention. For decades, extensive reports and reviews mainly focused on the fluorination and perfluoroalkylation (especially ...
Solubility
... • We can also determine the concentration of an ion necessary for precipitation to begin. • Assume that precipitation begins when Qsp = Ksp • Example: If a solution contains 0.0020 mol CrO42per liter, what concentration of Ag+ ion must be added as AgNO3 before Ag2CrO4 begins to precipitate. (Neglect ...
... • We can also determine the concentration of an ion necessary for precipitation to begin. • Assume that precipitation begins when Qsp = Ksp • Example: If a solution contains 0.0020 mol CrO42per liter, what concentration of Ag+ ion must be added as AgNO3 before Ag2CrO4 begins to precipitate. (Neglect ...
Name Reactions in Heterocyclic Chemistry-II
... isobutenyl ketone, (in the absence of a hydride donor, this unsaturated ketone replaced the saturated congener, and no isopropyl methyl ketone by rearrangement of the pivaloyl cation could result). An unexpected crystalline material also appeared in small yield when the aqueous solution was saturate ...
... isobutenyl ketone, (in the absence of a hydride donor, this unsaturated ketone replaced the saturated congener, and no isopropyl methyl ketone by rearrangement of the pivaloyl cation could result). An unexpected crystalline material also appeared in small yield when the aqueous solution was saturate ...
Electrophilic Additions: Alkenes Addition of Hydrogen Halides
... Hammond postulate: the transition state will be more similar to the species that it is closer to energetically ...
... Hammond postulate: the transition state will be more similar to the species that it is closer to energetically ...
unit 12 aldehydes, ketones and carboxylic acids
... C=O group. Q.17 Cyclohexanone forms cyanohydrin in good yield but 2,2,6 trimethyl cyclohexanone does not. Why? Ans In 2,2,6 trimethyl cyclohexanone there is stearic hinderance of 3 methyl groups, It does not form cynohydrin in good yield. Q.18 Why is carboxyl group in benzoic acid meta directing? An ...
... C=O group. Q.17 Cyclohexanone forms cyanohydrin in good yield but 2,2,6 trimethyl cyclohexanone does not. Why? Ans In 2,2,6 trimethyl cyclohexanone there is stearic hinderance of 3 methyl groups, It does not form cynohydrin in good yield. Q.18 Why is carboxyl group in benzoic acid meta directing? An ...
organic practice problems
... ____ 60. Which of the following molecules is ethanol? a. C2H6 b. CH3CO2H c. CH3CHO d. CH3CH2OH e. CH3OCH3 ____ 61. Which functional group does not contain an oxygen atom? a. alcohol b. amine c. amide d. ester e. ether ____ 62. The functional group RCO2R' is characteristic of an ________. a. ether b. ...
... ____ 60. Which of the following molecules is ethanol? a. C2H6 b. CH3CO2H c. CH3CHO d. CH3CH2OH e. CH3OCH3 ____ 61. Which functional group does not contain an oxygen atom? a. alcohol b. amine c. amide d. ester e. ether ____ 62. The functional group RCO2R' is characteristic of an ________. a. ether b. ...
Translation of rod-like template sequences into homochiral
... Translation of rod-like template sequences into homochiral assemblies of stacked helical oligomers Quan Gan1†, Xiang Wang1, Brice Kauffmann2, Frédéric Rosu2, Yann Ferrand1 and Ivan Huc1* At the molecular level, translation refers to the production of a new entity according to a template that has a d ...
... Translation of rod-like template sequences into homochiral assemblies of stacked helical oligomers Quan Gan1†, Xiang Wang1, Brice Kauffmann2, Frédéric Rosu2, Yann Ferrand1 and Ivan Huc1* At the molecular level, translation refers to the production of a new entity according to a template that has a d ...
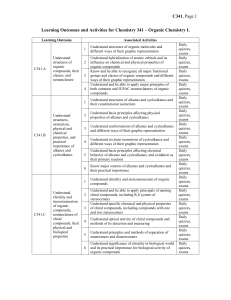
Chem341_outcomes
... Understand structure and nomenclature of alcohols and thiols, including primary, secondary, and tertiary alcohols, diols, and poliols Understand the presence of hydroxyl as a major factor determining specific physical and chemical properties of alcohols, including their high boiling points, their ac ...
... Understand structure and nomenclature of alcohols and thiols, including primary, secondary, and tertiary alcohols, diols, and poliols Understand the presence of hydroxyl as a major factor determining specific physical and chemical properties of alcohols, including their high boiling points, their ac ...
PHOSPHORUS AND SULFUR COSMOCHEMISTRY
... Phosphorus is a key element for life. This work reviews the role of phosphorus in life. Theories on the origin of life are confounded by a lack of reactive phosphorus, and attempts to overcome the dearth of reactive phosphorus must employ unrealistic phosphorus compounds, energetic organic compounds ...
... Phosphorus is a key element for life. This work reviews the role of phosphorus in life. Theories on the origin of life are confounded by a lack of reactive phosphorus, and attempts to overcome the dearth of reactive phosphorus must employ unrealistic phosphorus compounds, energetic organic compounds ...
Lecture 06 Slides
... Step 3: Assign oxidation numbers for the other atoms in the compound in order to make the sum of the oxidation numbers equal to zero ...
... Step 3: Assign oxidation numbers for the other atoms in the compound in order to make the sum of the oxidation numbers equal to zero ...
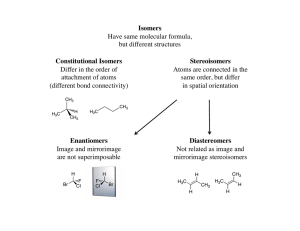
Hydrocarbons Note
... alkanes such as butane (C4H10) and pentane (C5H12). However, it becomes hopeless when larger alkanes are considered. For example there are 5 isomers of hexane (C6H14), 9 isomers of heptane (C7H16) and 75 isomers of decane (C10H22). Another problem arises as far as nomenclature is concerned. Hydrogen ...
... alkanes such as butane (C4H10) and pentane (C5H12). However, it becomes hopeless when larger alkanes are considered. For example there are 5 isomers of hexane (C6H14), 9 isomers of heptane (C7H16) and 75 isomers of decane (C10H22). Another problem arises as far as nomenclature is concerned. Hydrogen ...
Triple resonance for a three-level system of a chiral molecule
... transitions of molecules. We have, however, many examples of C1 chiral molecules in the literatures, for which all a-, b-, and c-types of rotational transitions were observed and reported. Thus the observations on these chiral molecules remained a puzzle for us for a long time. We may wonder how we ...
... transitions of molecules. We have, however, many examples of C1 chiral molecules in the literatures, for which all a-, b-, and c-types of rotational transitions were observed and reported. Thus the observations on these chiral molecules remained a puzzle for us for a long time. We may wonder how we ...
BSA - Sigma
... TMS derivatives and silylating reagents react with and are sensitive to active hydrogen atoms. Do not analyze BSA derivatives on stationary phases with these functional groups (e.g., polyethylene glycol phases). Silicones are the most useful phases for TMS derivatives – they combine inertness and st ...
... TMS derivatives and silylating reagents react with and are sensitive to active hydrogen atoms. Do not analyze BSA derivatives on stationary phases with these functional groups (e.g., polyethylene glycol phases). Silicones are the most useful phases for TMS derivatives – they combine inertness and st ...
Iridium(III) and Rhodium(III) compounds of dipyridyl-N
... have been used as catalysts for a wide range of reactions such as in hydrogenation,6 hydrosilytion,7 amination,8 C-C coupling9,10 and Diels-Alder reactions.11 Furthermore, compounds of this classes exhibited anticancer12 14 and DNA intercalative properties.15,16 Owing to their wide applications, syn ...
... have been used as catalysts for a wide range of reactions such as in hydrogenation,6 hydrosilytion,7 amination,8 C-C coupling9,10 and Diels-Alder reactions.11 Furthermore, compounds of this classes exhibited anticancer12 14 and DNA intercalative properties.15,16 Owing to their wide applications, syn ...
HCN Synthesis from Methane and Ammonia: Mechanisms of Pt
... have already been reported.6,15,22-26 As the C-N bond coupling in reaction 3 constitutes the central part of the theoretical discussion, the key issues are briefly summarized. Further, some details of the consecutive reactions in the Pt+/CH4/NH3 system are included here, which were not fully describ ...
... have already been reported.6,15,22-26 As the C-N bond coupling in reaction 3 constitutes the central part of the theoretical discussion, the key issues are briefly summarized. Further, some details of the consecutive reactions in the Pt+/CH4/NH3 system are included here, which were not fully describ ...
15: Carbonyl Compounds: Esters, Amides, and Related Molecules
... 15.2 Acid Halides (R-C(=O)-X) When Z of R-C(=O)-Z is a halogen atom (X), these compounds are equivalently referred to as acid halides, acyl halides, or alkanoyl halides. Preparation, Reactivity, and Properties (15.2A) The halogen atom of acid halides may be F, Cl, Br, or I, however acid chlorides (X ...
... 15.2 Acid Halides (R-C(=O)-X) When Z of R-C(=O)-Z is a halogen atom (X), these compounds are equivalently referred to as acid halides, acyl halides, or alkanoyl halides. Preparation, Reactivity, and Properties (15.2A) The halogen atom of acid halides may be F, Cl, Br, or I, however acid chlorides (X ...
CHAPTER 11 BONDING AND MOLECULAR STRUCTURE:
... Alkyl group names are used for simple alkyl group substituents. When there are two or more substituents, the positions around the ring are numbered beginning with the substituent first in the alphabet. ...
... Alkyl group names are used for simple alkyl group substituents. When there are two or more substituents, the positions around the ring are numbered beginning with the substituent first in the alphabet. ...
Parent Anions of Iron, Manganese, and Nickel Tetraphenyl
... for nickel than for other transition metals,32 where Hubbard-U approximations are often needed to obtain a qualitatively correct valence, within PBE-GGA, for the Ni ion.33 Nevertheless, this suggests that the relatively large molecular systems discussed here, especially the Ni system, should provide ...
... for nickel than for other transition metals,32 where Hubbard-U approximations are often needed to obtain a qualitatively correct valence, within PBE-GGA, for the Ni ion.33 Nevertheless, this suggests that the relatively large molecular systems discussed here, especially the Ni system, should provide ...
The Effects of Ozone on Compounds in Epicuticular Waxes in Plant
... quercetin, ferulic acid and methyl decanoate were chosen to be representative of compounds found in epicuticular waxes. Each standard was individually exposed to excess amounts of ozone and reacted in a Teflon bottle for a prolonged period prior to analysis. Both the standard and the ozonized produc ...
... quercetin, ferulic acid and methyl decanoate were chosen to be representative of compounds found in epicuticular waxes. Each standard was individually exposed to excess amounts of ozone and reacted in a Teflon bottle for a prolonged period prior to analysis. Both the standard and the ozonized produc ...
CH - UTH e
... Compounds p • Organometallic compound: a compound that contains a carbon-metal carbon metal bond • In this chapter, we focus on organometallic compounds of Mg, Li, and Cu – these classes illustrate the usefulness of organometallics in modern synthetic organic chemistry – they illustrate how the use ...
... Compounds p • Organometallic compound: a compound that contains a carbon-metal carbon metal bond • In this chapter, we focus on organometallic compounds of Mg, Li, and Cu – these classes illustrate the usefulness of organometallics in modern synthetic organic chemistry – they illustrate how the use ...
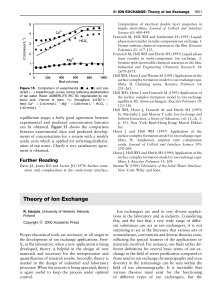
Theory of Ion Exchange
... side of eqn [12] constant), the ratio EA/EB must decrease as CT is decreased when zA'zB. Thus, the relative concentration of ion A must decrease with decreasing CT. This feature, the increased preference of an ion exchanger for the ion having a higher charge with the dilution of the solution, is cal ...
... side of eqn [12] constant), the ratio EA/EB must decrease as CT is decreased when zA'zB. Thus, the relative concentration of ion A must decrease with decreasing CT. This feature, the increased preference of an ion exchanger for the ion having a higher charge with the dilution of the solution, is cal ...
Homoaromaticity

Homoaromaticity in organic chemistry refers to a special case of aromaticity in which conjugation is interrupted by a single sp3 hybridized carbon atom. Although this sp3 center disrupts the continuous overlap of p-orbitals, traditionally thought to be a requirement for aromaticity, considerable thermodynamic stability and many of the spectroscopic, magnetic, and chemical properties associated with aromatic compounds are still observed for such compounds. This formal discontinuity is apparently bridged by p-orbital overlap, maintaining a contiguous cycle of π electrons that is responsible for this preserved chemical stability.The concept of homoaromaticity was pioneered by Saul Winstein in 1959, prompted by his studies of the “tris-homocyclopropenyl” cation. Since the publication of Winstein's paper, much research has been devoted to understanding and classifying these molecules, which represent an additional “class” of aromatic molecules included under the continuously broadening definition of aromaticity. To date, homoaromatic compounds are known to exist as cationic and anionic species, and some studies support the existence of neutral homoaromatic molecules, though these are less common. The 'homotropylium' cation (C8H9+) is perhaps the best studied example of a homoaromatic compound.