Atomic Structure and Periodic Trends
... • Each orbital can hold a maximum of 2 electrons of opposite spin. • Pauli exclusion principle states that no two electrons in an atom can have the same set of 4 quantum numbers ...
... • Each orbital can hold a maximum of 2 electrons of opposite spin. • Pauli exclusion principle states that no two electrons in an atom can have the same set of 4 quantum numbers ...
SOL PS3 Structure of the Atom by GA Tech
... Never express yourself more clearly than you are able to think. Prediction is very difficult, especially about the future. - Niels Bohr ...
... Never express yourself more clearly than you are able to think. Prediction is very difficult, especially about the future. - Niels Bohr ...
eBook AQA GCSE Chemistry Unit C2 Part 1
... Inside bromine Bromine consists of simple molecules. A simple molecule is made up of just a few atoms joined together by strong covalent bonds. The non-metal elements oxygen and hydrogen consist of simple molecules. So do the compounds hydrogen chloride, methane, and ammonia. ...
... Inside bromine Bromine consists of simple molecules. A simple molecule is made up of just a few atoms joined together by strong covalent bonds. The non-metal elements oxygen and hydrogen consist of simple molecules. So do the compounds hydrogen chloride, methane, and ammonia. ...
Vocabulary:
... Bohr’s Atomic Model Planetary System Model – Electrons move around the nucleus of an atom, like the planets around the sun. James Maxwell – Proposed that visible light consists of electromagnetic waves. Maxwell Planck – Suggested that atoms and molecules emit energy in discrete quantities, called qu ...
... Bohr’s Atomic Model Planetary System Model – Electrons move around the nucleus of an atom, like the planets around the sun. James Maxwell – Proposed that visible light consists of electromagnetic waves. Maxwell Planck – Suggested that atoms and molecules emit energy in discrete quantities, called qu ...
CHEMICAL EQUATIONS, SYMBOLS, FORULAS 7
... There are four H atoms on the reactant side (coefficient of 2 x subscript 2) and four H atoms on the product side (coefficient 2 x subscript 2). There are two O atoms on the reactant side (coefficient 2 x (understood) subscript 1) and two O atoms on the product side (understood coefficient 1 x subsc ...
... There are four H atoms on the reactant side (coefficient of 2 x subscript 2) and four H atoms on the product side (coefficient 2 x subscript 2). There are two O atoms on the reactant side (coefficient 2 x (understood) subscript 1) and two O atoms on the product side (understood coefficient 1 x subsc ...
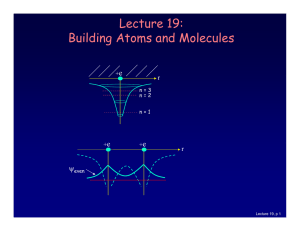
Lecture 19: Building Atoms and Molecules
... • Electrons do not pile up in the lowest energy state. It’s more like filling a bucket with water. • They are distributed among the energy levels according to the Exclusion Principle. • Particles that obey this principle are called “fermions”. Protons and neutrons are also fermions, but photons are ...
... • Electrons do not pile up in the lowest energy state. It’s more like filling a bucket with water. • They are distributed among the energy levels according to the Exclusion Principle. • Particles that obey this principle are called “fermions”. Protons and neutrons are also fermions, but photons are ...
Miss Pang`s 2012 Review
... 18. The "Plum Pudding" model of the atom was first proposed by: A) John Dalton ...
... 18. The "Plum Pudding" model of the atom was first proposed by: A) John Dalton ...
Electron Configurations and Periodicity
... So, going from hydrogen to helium, what’s different? Well, we know that in the case of helium, we have one more electron. If we just show a little cartoon of this here, we have a 2+ nucleus instead of a 1+ nucleus now, and we have two electrons. Again, I draw this little cartoon, but remember, these ...
... So, going from hydrogen to helium, what’s different? Well, we know that in the case of helium, we have one more electron. If we just show a little cartoon of this here, we have a 2+ nucleus instead of a 1+ nucleus now, and we have two electrons. Again, I draw this little cartoon, but remember, these ...
Midterm Review 4
... 53. The ion with a charge of +1 and the same electron configuration as argon is a. potassium b. sodium c. neon d. magnesium 54. The tendency to lose electrons ______________ as we move across a period on the periodic table a. increases b. remains the same c. decreases d. no trend exists 55. Generall ...
... 53. The ion with a charge of +1 and the same electron configuration as argon is a. potassium b. sodium c. neon d. magnesium 54. The tendency to lose electrons ______________ as we move across a period on the periodic table a. increases b. remains the same c. decreases d. no trend exists 55. Generall ...
Light and quantized Energy Section 1
... Einstein’s theory of light’s dual nature accounted for several unexplainable phenomena (photoelectric effect) but not why atomic emission spectra of elements were discontinuous rather continuous. ...
... Einstein’s theory of light’s dual nature accounted for several unexplainable phenomena (photoelectric effect) but not why atomic emission spectra of elements were discontinuous rather continuous. ...
Chemistry SOL Review
... • A 5.0 g object is heated from 25 C to 45 C. If it has a specific heat of 4.5 J/g°C, what is the heat generated by the object? ...
... • A 5.0 g object is heated from 25 C to 45 C. If it has a specific heat of 4.5 J/g°C, what is the heat generated by the object? ...
final study guide answers - Ponce
... d. Scientific theories summarize patterns found in nature. __B_____4. Why are scientific models important? a. They prove scientific theories. b. They help visualize things that are very complex, very large, or very small. c. They make it harder to understand things. d. They never change. ...
... d. Scientific theories summarize patterns found in nature. __B_____4. Why are scientific models important? a. They prove scientific theories. b. They help visualize things that are very complex, very large, or very small. c. They make it harder to understand things. d. They never change. ...
Quantum Mechanics
... The is called the Pauli Exclusion Principle. There are three possibilities for electrons in an orbital. The orbitals have 0,1, or 2 electons contained in them. This is due to the nature of how an atom fills its orbitals which will be discussed later. ...
... The is called the Pauli Exclusion Principle. There are three possibilities for electrons in an orbital. The orbitals have 0,1, or 2 electons contained in them. This is due to the nature of how an atom fills its orbitals which will be discussed later. ...
Unit B review - mvhs
... (A) Be, B, C, N (B) Ne, Ar, Kr, Xe (C) Mg, Ca, Sr, Ba (D) C, P, Se, I (E) Cr, Mn, Fe, Co 22. What is the correct order of decreasing first ionization energies for the elements Be, B, and C? (A) Be>B>C (B) B>Be>C (C) B>C>Be (D) C>Be>B (E) Be>C>B 23. Assume that an element has the following ionization ...
... (A) Be, B, C, N (B) Ne, Ar, Kr, Xe (C) Mg, Ca, Sr, Ba (D) C, P, Se, I (E) Cr, Mn, Fe, Co 22. What is the correct order of decreasing first ionization energies for the elements Be, B, and C? (A) Be>B>C (B) B>Be>C (C) B>C>Be (D) C>Be>B (E) Be>C>B 23. Assume that an element has the following ionization ...
Lab #1 - North Carolina High School Computational Chemistry Server
... known as ”ASA”, otherwise known as ”aspirin”. It is acidic because of the carboxylic acid functional group, consisting of a carbon atom with a double bonded oxygen and an OH ”group” attached. You should immediately identify the carboxylic acid portion of this compound. In this graphic, the carbon at ...
... known as ”ASA”, otherwise known as ”aspirin”. It is acidic because of the carboxylic acid functional group, consisting of a carbon atom with a double bonded oxygen and an OH ”group” attached. You should immediately identify the carboxylic acid portion of this compound. In this graphic, the carbon at ...
ENEE 313, Spr. `09 Midterm I Solutions
... The plane is shown in the figure below, with a top-down (normal-angle) view added on the side √ for convenience. From this, we can see that the nearest neighboring A atoms are a 3/2 apart, as are the nearest neighboring B atoms, while the shortest distance between any atoms are a/2. (c) (7 pts.) Thi ...
... The plane is shown in the figure below, with a top-down (normal-angle) view added on the side √ for convenience. From this, we can see that the nearest neighboring A atoms are a 3/2 apart, as are the nearest neighboring B atoms, while the shortest distance between any atoms are a/2. (c) (7 pts.) Thi ...
Preview Sample 2
... You also notice that the electrons in H2 are evenly distributed among the two atoms. Which two types of bonds are represented in these molecules? A. Covalent bonds in NaCl; ionic bonds in H2. B. Covalent bonds in NaCl; covalent bonds in H2. C. Ionic bonds in NaCl; ionic bonds in H2. D. Ionic bonds i ...
... You also notice that the electrons in H2 are evenly distributed among the two atoms. Which two types of bonds are represented in these molecules? A. Covalent bonds in NaCl; ionic bonds in H2. B. Covalent bonds in NaCl; covalent bonds in H2. C. Ionic bonds in NaCl; ionic bonds in H2. D. Ionic bonds i ...
A Student Introduction to Solar Energy
... Any operation of a semiconductor device depends on the concentration of carriers that transport charge inside the semiconductor and hence cause electrical currents. In order to determine and to understand device operation it is important to know the precise concentration of these charge carriers. In ...
... Any operation of a semiconductor device depends on the concentration of carriers that transport charge inside the semiconductor and hence cause electrical currents. In order to determine and to understand device operation it is important to know the precise concentration of these charge carriers. In ...
A. Atomic and Nuclear Structure
... negative instead of positive. In an uncharged atom, the number of orbital electrons equals the number of protons within the nucleus. When the number of orbital electrons does not equal the number of protons in the nucleus, an overall imbalance of charge exists for the atom. A charged atom is known a ...
... negative instead of positive. In an uncharged atom, the number of orbital electrons equals the number of protons within the nucleus. When the number of orbital electrons does not equal the number of protons in the nucleus, an overall imbalance of charge exists for the atom. A charged atom is known a ...
AP Chemistry
... 8.48 g of a compound containing only C, H and O is completely burned. It gives off 12.42 g of carbon dioxide and 5.08 g of water. What is the mass percentage of each element in this compound? ...
... 8.48 g of a compound containing only C, H and O is completely burned. It gives off 12.42 g of carbon dioxide and 5.08 g of water. What is the mass percentage of each element in this compound? ...
Adv review key
... A) Electrons are transferred between atoms B) Valence electrons- outer shell electrons C) Metals a. Lend valence electrons b. 1 – 4 valence electrons c. Form positive ions ( more protons than electrons) D) Nonmetals a. Borrow valence electrons b. 4 - 8 valence electrons c. Form negative ions (more e ...
... A) Electrons are transferred between atoms B) Valence electrons- outer shell electrons C) Metals a. Lend valence electrons b. 1 – 4 valence electrons c. Form positive ions ( more protons than electrons) D) Nonmetals a. Borrow valence electrons b. 4 - 8 valence electrons c. Form negative ions (more e ...
Chemical bond
A chemical bond is an attraction between atoms that allows the formation of chemical substances that contain two or more atoms. The bond is caused by the electrostatic force of attraction between opposite charges, either between electrons and nuclei, or as the result of a dipole attraction. The strength of chemical bonds varies considerably; there are ""strong bonds"" such as covalent or ionic bonds and ""weak bonds"" such as Dipole-dipole interaction, the London dispersion force and hydrogen bonding.Since opposite charges attract via a simple electromagnetic force, the negatively charged electrons that are orbiting the nucleus and the positively charged protons in the nucleus attract each other. An electron positioned between two nuclei will be attracted to both of them, and the nuclei will be attracted toward electrons in this position. This attraction constitutes the chemical bond. Due to the matter wave nature of electrons and their smaller mass, they must occupy a much larger amount of volume compared with the nuclei, and this volume occupied by the electrons keeps the atomic nuclei relatively far apart, as compared with the size of the nuclei themselves. This phenomenon limits the distance between nuclei and atoms in a bond.In general, strong chemical bonding is associated with the sharing or transfer of electrons between the participating atoms. The atoms in molecules, crystals, metals and diatomic gases—indeed most of the physical environment around us—are held together by chemical bonds, which dictate the structure and the bulk properties of matter.All bonds can be explained by quantum theory, but, in practice, simplification rules allow chemists to predict the strength, directionality, and polarity of bonds. The octet rule and VSEPR theory are two examples. More sophisticated theories are valence bond theory which includes orbital hybridization and resonance, and the linear combination of atomic orbitals molecular orbital method which includes ligand field theory. Electrostatics are used to describe bond polarities and the effects they have on chemical substances.