FREE Sample Here
... A. Odd number of neutrons and odd number of protons B. Even number of neutrons and odd number of protons C. Odd number of neutrons and even number of protons D. Even number of neutrons and even number of protons E. None of the options above results in significantly more stable nuclei. ...
... A. Odd number of neutrons and odd number of protons B. Even number of neutrons and odd number of protons C. Odd number of neutrons and even number of protons D. Even number of neutrons and even number of protons E. None of the options above results in significantly more stable nuclei. ...
Introduction to chemistry Multiple Choice 1. Which SI prefix means
... 63. When expressed in proper scientific notation the number 4289 is A. 4.289 X 10-4 B. 4.289 X 10-3 C. 4.289 X 103 D. 4.289 X104 Answer: C; Difficulty: easy; Reference: Section 2.1 64. When expressed in proper scientific notation the number 286 is A. 2.86 X 101 B. 2.86 X 102 C. 2.86 X 10-2 D. 28.6 X ...
... 63. When expressed in proper scientific notation the number 4289 is A. 4.289 X 10-4 B. 4.289 X 10-3 C. 4.289 X 103 D. 4.289 X104 Answer: C; Difficulty: easy; Reference: Section 2.1 64. When expressed in proper scientific notation the number 286 is A. 2.86 X 101 B. 2.86 X 102 C. 2.86 X 10-2 D. 28.6 X ...
Difficulty: how to deal accurately with both the core and
... Numerical atomic orbitals Numerical solution of the Kohn-Sham Hamiltonian for the isolated pseudoatom with the same approximations (xc,pseudos) as for the condensed system ...
... Numerical atomic orbitals Numerical solution of the Kohn-Sham Hamiltonian for the isolated pseudoatom with the same approximations (xc,pseudos) as for the condensed system ...
Chapter 3 - Cloudfront.net
... there is a strong attraction between them. • A similar attraction exists when neutrons are very close to each other or when protons and neutrons are very close together. • The short-range proton-neutron, proton-proton, and neutron-neutron forces that hold the nuclear particles together are referred ...
... there is a strong attraction between them. • A similar attraction exists when neutrons are very close to each other or when protons and neutrons are very close together. • The short-range proton-neutron, proton-proton, and neutron-neutron forces that hold the nuclear particles together are referred ...
The Coordination Chemistry of Solvated Metal Ions in DMPU
... rearrangements of atoms. Around this conglomerate of ideas he and a few others started building the foundation of the chemistry we know today.[6] The main blemish in Dalton’s atomic theory was a presupposed fact that water consists of one hydrogen atom and one oxygen atom, whereas the true ratio is ...
... rearrangements of atoms. Around this conglomerate of ideas he and a few others started building the foundation of the chemistry we know today.[6] The main blemish in Dalton’s atomic theory was a presupposed fact that water consists of one hydrogen atom and one oxygen atom, whereas the true ratio is ...
Atomic orbitals of finite range as basis sets
... The same Hilbert space can be expanded if we use the difference, with the advantage that now the second-ζ vanishes at rm (more efficient) ...
... The same Hilbert space can be expanded if we use the difference, with the advantage that now the second-ζ vanishes at rm (more efficient) ...
chapter 3 - Denton ISD
... Charge and Mass of the Electron Cathode rays have identical properties regardless of the element used to produce them. Therefore, it was concluded that electrons are present in atoms of all elements. Thus, cathode-ray experiments provided evidence that atoms are divisible and that one of the atom’s ...
... Charge and Mass of the Electron Cathode rays have identical properties regardless of the element used to produce them. Therefore, it was concluded that electrons are present in atoms of all elements. Thus, cathode-ray experiments provided evidence that atoms are divisible and that one of the atom’s ...
Chapter 2 - Atoms and the Periodic Table (test bank)
... In the early 1900s, Ernest Rutherford performed an experiment with thin foils of gold and alpha particles to probe the structure of the atoms. He observed that most of these alpha particles penetrated the foil and were not deflected. Realizing that atoms are electrically neutral (that is, they have ...
... In the early 1900s, Ernest Rutherford performed an experiment with thin foils of gold and alpha particles to probe the structure of the atoms. He observed that most of these alpha particles penetrated the foil and were not deflected. Realizing that atoms are electrically neutral (that is, they have ...
Alchemy Invesigation III
... Purpose: The various physical and chemical properties of the elements can be traced to the electrons. By studying electrons further we may be able to unlock the key to creating substances similar to gold. This lesson will reveal the arrangement of electrons within atoms. ...
... Purpose: The various physical and chemical properties of the elements can be traced to the electrons. By studying electrons further we may be able to unlock the key to creating substances similar to gold. This lesson will reveal the arrangement of electrons within atoms. ...
CHE 110 Dr. Nicholas Bizier Office DS 337b email
... The number of protons and neutrons is equal to the atomic mass. ...
... The number of protons and neutrons is equal to the atomic mass. ...
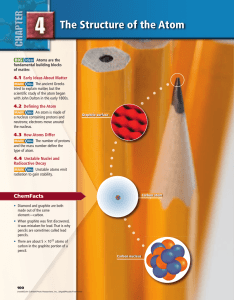
Chapter 4: The Structure of the Atom
... MAIN Idea An atom is made of a nucleus containing protons and neutrons; electrons move around the nucleus. Real-World Reading Link If you have ever accidentally bitten into a peach pit, you know that your teeth pass easily through the fruit, but cannot dent the hard pit. Similarly, many particles th ...
... MAIN Idea An atom is made of a nucleus containing protons and neutrons; electrons move around the nucleus. Real-World Reading Link If you have ever accidentally bitten into a peach pit, you know that your teeth pass easily through the fruit, but cannot dent the hard pit. Similarly, many particles th ...
Document
... – Students need to be protons, neutrons, and electrons in the correct orbitals. • Boys neutrons, girls protons in nucleus? ...
... – Students need to be protons, neutrons, and electrons in the correct orbitals. • Boys neutrons, girls protons in nucleus? ...
Chapter 23 Metals and Metallurgy
... • Transition metals often have more than one common oxidation state. Most have +2 state due to loss of s electrons. Oxidation numbers greater than 2 are due to loss of d electrons as well as s. Metals and Metallurgy ...
... • Transition metals often have more than one common oxidation state. Most have +2 state due to loss of s electrons. Oxidation numbers greater than 2 are due to loss of d electrons as well as s. Metals and Metallurgy ...
2(g)
... 2. Write information…mols….masse under the chemical/element that it relates to. 3. ***Convert information given in question into mols.*** 4. Find the chemical species the question wants to know about and put a N for (need to find) above it. 5. Put a G for (got information about this) over the specie ...
... 2. Write information…mols….masse under the chemical/element that it relates to. 3. ***Convert information given in question into mols.*** 4. Find the chemical species the question wants to know about and put a N for (need to find) above it. 5. Put a G for (got information about this) over the specie ...
C 4 The Atomic Theory
... to produce a new substance, the reactants (hydrogen and oxygen) always reacted in the same proportions by mass. In other words, if 1 gram of hydrogen reacted with 8 grams of oxygen, then 2 grams of hydrogen would react with 16 grams of oxygen, and 3 grams of hydrogen would react with 24 grams of oxy ...
... to produce a new substance, the reactants (hydrogen and oxygen) always reacted in the same proportions by mass. In other words, if 1 gram of hydrogen reacted with 8 grams of oxygen, then 2 grams of hydrogen would react with 16 grams of oxygen, and 3 grams of hydrogen would react with 24 grams of oxy ...