Unit 1 Section 4 - Atomic Structure PPT
... pieces - protons, neutrons and electrons • The nucleus contains protons and neutrons • The nucleus is only about 10-13 cm in diameter • The electrons move outside the nucleus with an average distance of about 10-8 cm ...
... pieces - protons, neutrons and electrons • The nucleus contains protons and neutrons • The nucleus is only about 10-13 cm in diameter • The electrons move outside the nucleus with an average distance of about 10-8 cm ...
1 Unit 2: Atomic Theory Unit Notes Name: Period: ______
... Objective: To define the parts of an atom Please watch the following video: Bill Nye Atoms Each subatomic particle has very unique physical properties that will in turn; affect the chemical properties of the atoms they make up. The following reading passage will help you to better understand the str ...
... Objective: To define the parts of an atom Please watch the following video: Bill Nye Atoms Each subatomic particle has very unique physical properties that will in turn; affect the chemical properties of the atoms they make up. The following reading passage will help you to better understand the str ...
Section 2.7 An Introduction to the Periodic Table
... the overall size of the atom. Extremely dense; accounts for almost all of the atom’s mass. ...
... the overall size of the atom. Extremely dense; accounts for almost all of the atom’s mass. ...
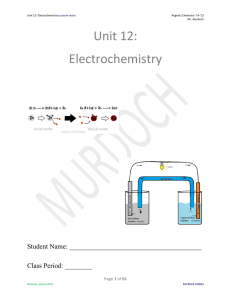
Unit 12: Electrochemistry
... ionization energy; they (and what it means for the can easily lose electrons element) - Table S when energy is added ...
... ionization energy; they (and what it means for the can easily lose electrons element) - Table S when energy is added ...
redox reaction - Seattle Central College
... Most ionic compounds dissolve in water to produce solutions that conduct electricity. The degree to which a solution will conduct electricity is used to determine if a solute is a strong or weak electrolyte. Solutions that do not conduct electricity are called nonelectrolytes. Molecular compounds th ...
... Most ionic compounds dissolve in water to produce solutions that conduct electricity. The degree to which a solution will conduct electricity is used to determine if a solute is a strong or weak electrolyte. Solutions that do not conduct electricity are called nonelectrolytes. Molecular compounds th ...
Symbol Protons Neutons Electrons Name
... their position in the Periodic Table. Cations have the same name as the atoms from which they are derived. • B-group (transition) metals can form two or more different ions. To name them, a Roman Numeral following the symbol is used to indicate the charge on the ion. ...
... their position in the Periodic Table. Cations have the same name as the atoms from which they are derived. • B-group (transition) metals can form two or more different ions. To name them, a Roman Numeral following the symbol is used to indicate the charge on the ion. ...
101
... To assign oxidation numbers to the atoms in a water molecule, you can consider all the bonding electrons to be “owned” by the more electronegative oxygen atom, as shown in Figure 10.4B. Thus, each hydrogen atom in a water molecule is considered to have no electrons, as hydrogen would in a hydrogen ...
... To assign oxidation numbers to the atoms in a water molecule, you can consider all the bonding electrons to be “owned” by the more electronegative oxygen atom, as shown in Figure 10.4B. Thus, each hydrogen atom in a water molecule is considered to have no electrons, as hydrogen would in a hydrogen ...
Atomic Theory of Matter
... Rules for predicting charges on monatomic ions Most of the main group metals form cations with the charge equal to their group number. The charge on a monatomic anion for a nonmetal equals the group number minus 8. Most transition elements form more than one ion, each with a different charge. Chapte ...
... Rules for predicting charges on monatomic ions Most of the main group metals form cations with the charge equal to their group number. The charge on a monatomic anion for a nonmetal equals the group number minus 8. Most transition elements form more than one ion, each with a different charge. Chapte ...
honors chemistry harvard-westlake second semester final exam
... 17. Consider the reaction below: 1.7 kJ + NaCl(s) + H2O(ℓ) Na+(aq) + Cl-(aq) Which of the following combinations for H and S is correct? a. +,- b. -,+ c. -,- d. +,+ 18. How many grams of sodium hydroxide pellets are required to prepare 50 mL (to the nearest 1 mL) of a 0.15 M solution? a. 0.30 b ...
... 17. Consider the reaction below: 1.7 kJ + NaCl(s) + H2O(ℓ) Na+(aq) + Cl-(aq) Which of the following combinations for H and S is correct? a. +,- b. -,+ c. -,- d. +,+ 18. How many grams of sodium hydroxide pellets are required to prepare 50 mL (to the nearest 1 mL) of a 0.15 M solution? a. 0.30 b ...
Review Study Guide for the Final
... What is it called when you have more electrons than protons? ...
... What is it called when you have more electrons than protons? ...
Lecture 1 - Алтайский государственный технический
... The diameters of atomic nuclei are about 10-4A. Thus, the nuclei are about 0.01% the diameter of the atom as a whole. If the nucleus had a diameter equal to that of a pinhead, then the atom itself would have a diameter of some 10 meters (about 39 and a half feet). The nucleus of an atom is therefor ...
... The diameters of atomic nuclei are about 10-4A. Thus, the nuclei are about 0.01% the diameter of the atom as a whole. If the nucleus had a diameter equal to that of a pinhead, then the atom itself would have a diameter of some 10 meters (about 39 and a half feet). The nucleus of an atom is therefor ...
atoms - WordPress.com
... All the positive charge, and almost all the mass is concentrated in a small area in the center. He called this a “nucleus” The nucleus is composed of protons and neutrons (they make the nucleus!) The electrons distributed around the nucleus, and occupy most of the volume His model was called ...
... All the positive charge, and almost all the mass is concentrated in a small area in the center. He called this a “nucleus” The nucleus is composed of protons and neutrons (they make the nucleus!) The electrons distributed around the nucleus, and occupy most of the volume His model was called ...
FREE Sample Here
... Skill: Knowledge/Comprehension 2) Trace elements are those required by an organism in only minute quantities. Which of the following is a trace element that is required by humans and other vertebrates? A) nitrogen B) calcium C) iodine D) sodium E) phosphorus Answer: C Topic: Concept 2.1 Skill: Knowl ...
... Skill: Knowledge/Comprehension 2) Trace elements are those required by an organism in only minute quantities. Which of the following is a trace element that is required by humans and other vertebrates? A) nitrogen B) calcium C) iodine D) sodium E) phosphorus Answer: C Topic: Concept 2.1 Skill: Knowl ...
13.0 Redox Reactions PowerPoint
... Notice that both of these half-reactions are balanced by mass (same number of atoms/ions of each element on both sides) and by charge (same total charge on both sides) ...
... Notice that both of these half-reactions are balanced by mass (same number of atoms/ions of each element on both sides) and by charge (same total charge on both sides) ...
Biology, 8e (Campbell) Chapter 2 The Chemical Context of Life
... Topic: Concept 2.1 Skill: Knowledge/Comprehension 2) Trace elements are those required by an organism in only minute quantities. Which of the following is a trace element that is required by humans and other vertebrates? A) nitrogen B) calcium C) iodine D) sodium E) phosphorus Answer: C Topic: Conce ...
... Topic: Concept 2.1 Skill: Knowledge/Comprehension 2) Trace elements are those required by an organism in only minute quantities. Which of the following is a trace element that is required by humans and other vertebrates? A) nitrogen B) calcium C) iodine D) sodium E) phosphorus Answer: C Topic: Conce ...
FREE Sample Here
... Topic: Concept 2.1 Skill: Knowledge/Comprehension 2) Trace elements are those required by an organism in only minute quantities. Which of the following is a trace element that is required by humans and other vertebrates? A) nitrogen B) calcium C) iodine D) sodium E) phosphorus Answer: C Topic: Conce ...
... Topic: Concept 2.1 Skill: Knowledge/Comprehension 2) Trace elements are those required by an organism in only minute quantities. Which of the following is a trace element that is required by humans and other vertebrates? A) nitrogen B) calcium C) iodine D) sodium E) phosphorus Answer: C Topic: Conce ...
FREE Sample Here
... Skill: Knowledge/Comprehension 2) Trace elements are those required by an organism in only minute quantities. Which of the following is a trace element that is required by humans and other vertebrates? A) nitrogen B) calcium C) iodine D) sodium E) phosphorus Answer: C Topic: Concept 2.1 Skill: Knowl ...
... Skill: Knowledge/Comprehension 2) Trace elements are those required by an organism in only minute quantities. Which of the following is a trace element that is required by humans and other vertebrates? A) nitrogen B) calcium C) iodine D) sodium E) phosphorus Answer: C Topic: Concept 2.1 Skill: Knowl ...
FREE Sample Here
... Topic: Concept 2.1 Skill: Knowledge/Comprehension 2) Trace elements are those required by an organism in only minute quantities. Which of the following is a trace element that is required by humans and other vertebrates? A) nitrogen B) calcium C) iodine D) sodium E) phosphorus Answer: C Topic: Conce ...
... Topic: Concept 2.1 Skill: Knowledge/Comprehension 2) Trace elements are those required by an organism in only minute quantities. Which of the following is a trace element that is required by humans and other vertebrates? A) nitrogen B) calcium C) iodine D) sodium E) phosphorus Answer: C Topic: Conce ...