Atoms, Molecules, and Ions
... and environmental exposure, with the outcome difficult to predict. Early detection of biomarkers, substances that indicate an organism’s disease or physiological state, could allow diagnosis and treatment before a condition becomes serious or irreversible. Recent studies have shown that your exhaled ...
... and environmental exposure, with the outcome difficult to predict. Early detection of biomarkers, substances that indicate an organism’s disease or physiological state, could allow diagnosis and treatment before a condition becomes serious or irreversible. Recent studies have shown that your exhaled ...
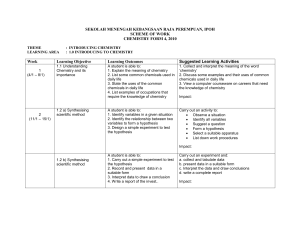
SEKOLAH MENENGAH KEBANGSAAN RAJA PEREMPUAN, IPOH
... Suggested Learning Activities Discuss and explain the particulate nature of matter. Use models or view computer simulations to discuss 1. the kinetic theory of matter 2 .the meaning of atoms, molecules and ions 3. conduct an activity to investigate diffusion of particles in solid, liquid and gas. Im ...
... Suggested Learning Activities Discuss and explain the particulate nature of matter. Use models or view computer simulations to discuss 1. the kinetic theory of matter 2 .the meaning of atoms, molecules and ions 3. conduct an activity to investigate diffusion of particles in solid, liquid and gas. Im ...
C5H12 + 8 O2 → 5 CO2 + 6 H2O
... e.g.: O2, halogens, H2O2, HNO3, Cr2O7–, MnO4– • Reducing agents: Elements or compounds that reduce the other reactant. e.g.: H2, C, metals ...
... e.g.: O2, halogens, H2O2, HNO3, Cr2O7–, MnO4– • Reducing agents: Elements or compounds that reduce the other reactant. e.g.: H2, C, metals ...
Inside the atom - Oxford University Press
... In the early 19th century, over a thousand years later, English chemist John Dalton built on Democritus’s idea of indivisible particles. He also suggested that different substances were made up of different particles that had specific masses and properties – elements. In other words, the particles t ...
... In the early 19th century, over a thousand years later, English chemist John Dalton built on Democritus’s idea of indivisible particles. He also suggested that different substances were made up of different particles that had specific masses and properties – elements. In other words, the particles t ...
Power Point over chemistry
... retaining heat is important to our climate. It means that our climate stays much more stable than it would if there were less water on Earth. TAKS Need to Know ...
... retaining heat is important to our climate. It means that our climate stays much more stable than it would if there were less water on Earth. TAKS Need to Know ...
chapter 1 - Louisiana Tech University
... Which of the following statements concerning the chemical symbols for the elements is correct? a) All chemical symbols start with the first letter of the element’s English name. b) Most elements have two-letter chemical symbols. c) Synthetic elements have one-letter symbols and naturally-occurring e ...
... Which of the following statements concerning the chemical symbols for the elements is correct? a) All chemical symbols start with the first letter of the element’s English name. b) Most elements have two-letter chemical symbols. c) Synthetic elements have one-letter symbols and naturally-occurring e ...
atom - Physicsland
... • to date, 115 are known – 90 occur in nature – others produced in laboratory are unstable Words atom and element can be used interchangeably Copyright © 2008 Pearson Education, Inc., publishing as Pearson Addison-Wesley ...
... • to date, 115 are known – 90 occur in nature – others produced in laboratory are unstable Words atom and element can be used interchangeably Copyright © 2008 Pearson Education, Inc., publishing as Pearson Addison-Wesley ...
Chapter 1: Matter and Measurement
... radioactive isotopes of elements of low atomic number. Its percent natural abundance among K isotopes is 0.012%. How many 40K atoms do you ingest by drinking one cup of whole milk containing 371 mg of K? Want atoms of 40K, need atoms of K, Want atoms of K, need moles of K, Want moles of K, need mass ...
... radioactive isotopes of elements of low atomic number. Its percent natural abundance among K isotopes is 0.012%. How many 40K atoms do you ingest by drinking one cup of whole milk containing 371 mg of K? Want atoms of 40K, need atoms of K, Want atoms of K, need moles of K, Want moles of K, need mass ...
Chapter 2 Atoms, Molecules, and Ions
... • If the anion is an element, change its ending to -ide; if the anion is a polyatomic ion, simply write the name of the polyatomic ion. • If the cation can have more than one possible charge, write the charge as a Roman numeral in parentheses. Atoms, Molecules, and Ions ...
... • If the anion is an element, change its ending to -ide; if the anion is a polyatomic ion, simply write the name of the polyatomic ion. • If the cation can have more than one possible charge, write the charge as a Roman numeral in parentheses. Atoms, Molecules, and Ions ...
Chapter 2 – Atoms, Ions, and the Periodic Table
... The atomic number of an atom is equal to the number of protons. If you know the name of the element, you can find the atomic number by finding the element on the periodic table. For example, for iron (Fe), you can find the atomic number, 26, listed with the element symbol in the fourth period of the ...
... The atomic number of an atom is equal to the number of protons. If you know the name of the element, you can find the atomic number by finding the element on the periodic table. For example, for iron (Fe), you can find the atomic number, 26, listed with the element symbol in the fourth period of the ...
CHEMISTRY SOL REVIEW MATERIAL Name SCIENTIFIC
... He tried to explain the bright-line spectrum of hydrogen with a model of the atom in which electrons occupy fixed energy levels and circle the nucleus in orbits, like planets around the sun. MENDELEEV He came up with the first periodic table and predicted the properties of a few elements that had no ...
... He tried to explain the bright-line spectrum of hydrogen with a model of the atom in which electrons occupy fixed energy levels and circle the nucleus in orbits, like planets around the sun. MENDELEEV He came up with the first periodic table and predicted the properties of a few elements that had no ...
The Atom - cloudfront.net
... Tlte nucleus is a very small region located at the center of the atom. The nucleus includes at least one positively charged particle called a proton and usually one or more neutral particles called neutrons. Surrounding the nucleus is a much larger region that contains negatively charged particles c ...
... Tlte nucleus is a very small region located at the center of the atom. The nucleus includes at least one positively charged particle called a proton and usually one or more neutral particles called neutrons. Surrounding the nucleus is a much larger region that contains negatively charged particles c ...
atoms
... • Because in the real world we use large amounts of atoms and molecules, we use average masses in calculations. • Average mass is calculated from the isotopes of an element weighted by their relative abundances. ...
... • Because in the real world we use large amounts of atoms and molecules, we use average masses in calculations. • Average mass is calculated from the isotopes of an element weighted by their relative abundances. ...
Section 2.9 Molar Mass: Counting Atoms by Weighing Them
... Each element is made up of tiny indestructible particles called atoms. All atoms of a given element have the same mass and other properties that distinguish them from atoms of other elements Atoms combine in simple whole number ratios to form compounds. Atoms of one element cannot change into atoms ...
... Each element is made up of tiny indestructible particles called atoms. All atoms of a given element have the same mass and other properties that distinguish them from atoms of other elements Atoms combine in simple whole number ratios to form compounds. Atoms of one element cannot change into atoms ...
Atoms
... • Each electron shell can hold a specific maximum number of electrons. • The valence electrons are in the outermost electron shell of an atom. Electrons that are not valence electrons are called core electrons. (cont.) © 2004 Key Curriculum Press. ...
... • Each electron shell can hold a specific maximum number of electrons. • The valence electrons are in the outermost electron shell of an atom. Electrons that are not valence electrons are called core electrons. (cont.) © 2004 Key Curriculum Press. ...
Chapter 2: Atoms, Ions, and the Periodic Table
... C) Protons and neutrons Ans: C 18. The number of ________ determines the identity of an element. A) electrons D) neutrons plus protons B) protons E) protons plus electrons C) neutrons Ans: B 19. The atomic number of an element represents A) the number of electrons its atom can gain. B) the number of ...
... C) Protons and neutrons Ans: C 18. The number of ________ determines the identity of an element. A) electrons D) neutrons plus protons B) protons E) protons plus electrons C) neutrons Ans: B 19. The atomic number of an element represents A) the number of electrons its atom can gain. B) the number of ...