Electron spin and the periodic table
... We can check this by putting the particle through an inhomogeneous field, because Fz = μz dB/dz. This was the idea behind the Stern-Gerlach experiment, in which the electron passes through a field and is deflected by it. ...
... We can check this by putting the particle through an inhomogeneous field, because Fz = μz dB/dz. This was the idea behind the Stern-Gerlach experiment, in which the electron passes through a field and is deflected by it. ...
Unit 2: Biochem Notes
... - A solution with a pH __________ 7, has more OH- ions than H+ ions, and is basic. - A solution with a pH _________ 7, has more H+ ions than OH- ions, and is acidic. b. buffer – Weak acids or bases that can react with strong acids or bases to prevent sharp, sudden changes in pH. Buffers make acidic ...
... - A solution with a pH __________ 7, has more OH- ions than H+ ions, and is basic. - A solution with a pH _________ 7, has more H+ ions than OH- ions, and is acidic. b. buffer – Weak acids or bases that can react with strong acids or bases to prevent sharp, sudden changes in pH. Buffers make acidic ...
Exit Slip: Atomic Structure and Nuclear Chemistry-1
... C. splitting of a uranium nucleus into barium and krypton nuclei D. return of ground state of electrons that have been excited to a higher energy level 3. Nuclei with too many or too few neutrons are (11.a) A. never found. C. unstable. B. unnatural. D. stable. 4. Arrange the forms of radiation from ...
... C. splitting of a uranium nucleus into barium and krypton nuclei D. return of ground state of electrons that have been excited to a higher energy level 3. Nuclei with too many or too few neutrons are (11.a) A. never found. C. unstable. B. unnatural. D. stable. 4. Arrange the forms of radiation from ...
January 1999
... the rod is always pointed in the radial direction. Calculate the tension at the center of the rod. ...
... the rod is always pointed in the radial direction. Calculate the tension at the center of the rod. ...
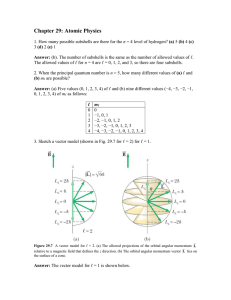
AP Semester I Review: Free Response Questions
... that represents the different particles present in the solution. Base the particles in your drawing on the particles shown in the representations above. Include only one formula unit of LiCl and no more than ten molecules of water. ...
... that represents the different particles present in the solution. Base the particles in your drawing on the particles shown in the representations above. Include only one formula unit of LiCl and no more than ten molecules of water. ...
The Periodic Table OL Page 1 of 2 G. Galvin Name: Periodic Table
... Defn: The mass number of an element is the sum of the number of protons and neutrons in the nucleus of an atom of that element. No. of neutrons in an atom = Mass Number (A) – Atomic Number (Z) Defn: Isotopes are atoms of the same element (i.e. they have the same atomic number) which have different m ...
... Defn: The mass number of an element is the sum of the number of protons and neutrons in the nucleus of an atom of that element. No. of neutrons in an atom = Mass Number (A) – Atomic Number (Z) Defn: Isotopes are atoms of the same element (i.e. they have the same atomic number) which have different m ...
Van der Waals Forces Between Atoms
... The perfect gas equation of state PV = NkT is manifestly incapable of describing actual gases at low temperatures, since they undergo a discontinuous change of volume and become liquids. In the 1870’s, the Dutch physicist Van der Waals came up with an improvement: a gas law that recognized the m ...
... The perfect gas equation of state PV = NkT is manifestly incapable of describing actual gases at low temperatures, since they undergo a discontinuous change of volume and become liquids. In the 1870’s, the Dutch physicist Van der Waals came up with an improvement: a gas law that recognized the m ...
4 colour slides per page
... • We have modified the Bohr model to account for waveparticle duality and the uncertainty principle. • An additional quantum number, ms, is required to explain magnetic field effects. • Schrödinger’s equation can be written for any electron system, but cannot be solved exactly for more than one elec ...
... • We have modified the Bohr model to account for waveparticle duality and the uncertainty principle. • An additional quantum number, ms, is required to explain magnetic field effects. • Schrödinger’s equation can be written for any electron system, but cannot be solved exactly for more than one elec ...
Summary
... With the realization of coherent, laser-like atoms in the form of Bose-Einstein condensates it has become possible to explore matter-wave amplification, a process in which the number of atoms in a quantum state is amplified due to bosonic stimulation. In previous amplifiers based on superradiant Ray ...
... With the realization of coherent, laser-like atoms in the form of Bose-Einstein condensates it has become possible to explore matter-wave amplification, a process in which the number of atoms in a quantum state is amplified due to bosonic stimulation. In previous amplifiers based on superradiant Ray ...
NASC 1110
... Electrons may have any energy within bands, but not within band gaps. Electrons are distributed from the lowest energy levels up in agreement with the exclusion principle. In metals, the highest energy band is partially full. In insulators, it is completely full. ...
... Electrons may have any energy within bands, but not within band gaps. Electrons are distributed from the lowest energy levels up in agreement with the exclusion principle. In metals, the highest energy band is partially full. In insulators, it is completely full. ...
Atomic Theory electron charge: -1.6 X 10-19C
... nucleus contains the positively charged protons (and also neutrons in most atoms) with electrons orbitting about it. Compared to the total volume of the atom, which is determined by the orbital space of the electrons, the volume of the nucleus is minute. For the most part, matter is filled with empt ...
... nucleus contains the positively charged protons (and also neutrons in most atoms) with electrons orbitting about it. Compared to the total volume of the atom, which is determined by the orbital space of the electrons, the volume of the nucleus is minute. For the most part, matter is filled with empt ...
Midterm Review Packet - Mrs. McKenzie`s Chemistry and ICP Classes
... formed during ___________________________ ______________________________. 6. Atoms with filled outermost energy levels tend _____________ to participate in chemical reactions. 7. A pure substance that cannot be broken down is called an ____________________. 8. The simplest part of an element is an _ ...
... formed during ___________________________ ______________________________. 6. Atoms with filled outermost energy levels tend _____________ to participate in chemical reactions. 7. A pure substance that cannot be broken down is called an ____________________. 8. The simplest part of an element is an _ ...
IntroRedoxDCIAns
... b. Identify two characteristics common to these equations. The first three reactions show an element, in this case oxygen, converted to the combined form of oxygen in a compound. An element was converted to a compound in the reactions. In the fourth reaction, a compound decomposed into its elements. ...
... b. Identify two characteristics common to these equations. The first three reactions show an element, in this case oxygen, converted to the combined form of oxygen in a compound. An element was converted to a compound in the reactions. In the fourth reaction, a compound decomposed into its elements. ...
Introduction to Oxidation Reduction
... b. Identify two characteristics common to these equations. The first three reactions show an element, in this case oxygen, converted to the combined form of oxygen in a compound. An element was converted to a compound in the reactions. In the fourth reaction, a compound decomposed into its elements. ...
... b. Identify two characteristics common to these equations. The first three reactions show an element, in this case oxygen, converted to the combined form of oxygen in a compound. An element was converted to a compound in the reactions. In the fourth reaction, a compound decomposed into its elements. ...
Answers to questions on test #2
... Without Slater’s rules: e− #1 spends half its time closer to the nucleus than e− #2. This suggests that e− #1 fells Z = 2 half the time, Z = 1 half the time, and Zef f = 1.5 on average: Zef f ≈ 1.5. (note: this would be true only if, at all times, one e− is much closer to the nucleus than the other. ...
... Without Slater’s rules: e− #1 spends half its time closer to the nucleus than e− #2. This suggests that e− #1 fells Z = 2 half the time, Z = 1 half the time, and Zef f = 1.5 on average: Zef f ≈ 1.5. (note: this would be true only if, at all times, one e− is much closer to the nucleus than the other. ...
Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.The word atom comes from the Ancient Greek adjective atomos, meaning ""uncuttable"". 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. While seemingly apropos, around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called ""uncuttable atom"" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all. Since atoms were found to be divisible, physicists later invented the term ""elementary particles"" to describe the ""uncuttable"", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.