AP Chemistry 2013 Semester 1 Final Exam Review Problems
... Chapter 2: Atoms, Molecules and Ions Topics: protons, electrons, and neutrons: development of atomic structure; atomic number and atomic mass; isotopes; atomic mass; periodic table basics; molecules, compounds and formulas; Ionic Compounds: formulas, names and properties; Molecular Compounds: formul ...
... Chapter 2: Atoms, Molecules and Ions Topics: protons, electrons, and neutrons: development of atomic structure; atomic number and atomic mass; isotopes; atomic mass; periodic table basics; molecules, compounds and formulas; Ionic Compounds: formulas, names and properties; Molecular Compounds: formul ...
The Bohr Atom
... in the case of the hydrogen atom, as the electron loses energy, it moves into an orbit of smaller radius, loses energy more rapidly and spirals into the nucleus within about 10−10 seconds. This cannot be correct since atoms certainly exist. This problem was solved by Bohr in an remarkable leap of th ...
... in the case of the hydrogen atom, as the electron loses energy, it moves into an orbit of smaller radius, loses energy more rapidly and spirals into the nucleus within about 10−10 seconds. This cannot be correct since atoms certainly exist. This problem was solved by Bohr in an remarkable leap of th ...
Teacher quality grant - Gulf Coast State College
... An atom that loses one or more electrons becomes positively charged, while an atom that acquires electrons becomes negatively charged. This transfer of electrons is driven by the fact that atoms with full outer electron shells are more stable. Donated electron ...
... An atom that loses one or more electrons becomes positively charged, while an atom that acquires electrons becomes negatively charged. This transfer of electrons is driven by the fact that atoms with full outer electron shells are more stable. Donated electron ...
Teacher quality grant
... An atom that loses one or more electrons becomes positively charged, while an atom that acquires electrons becomes negatively charged. This transfer of electrons is driven by the fact that atoms with full outer electron shells are more stable. Donated electron ...
... An atom that loses one or more electrons becomes positively charged, while an atom that acquires electrons becomes negatively charged. This transfer of electrons is driven by the fact that atoms with full outer electron shells are more stable. Donated electron ...
Science Olympiad
... ______ 7. In moving from left to right across a period in the periodic table of the elements (A) ionization energy decreases due to increases shielding effect. (B) atomic radius decreases due to an increase in effective nuclear charge. (C) electronegativity decreases due to an increase in atomic rad ...
... ______ 7. In moving from left to right across a period in the periodic table of the elements (A) ionization energy decreases due to increases shielding effect. (B) atomic radius decreases due to an increase in effective nuclear charge. (C) electronegativity decreases due to an increase in atomic rad ...
1 The Photoelectric Effect 2 Line Spectra and Energy Levels
... scintillation: momentary flash caused by a particle being absorbed and re-emitted by certain materials. Geiger counter : a device commonly used to detect radiation. nucleus: positively charged, dense core of the atom consisting of neutrons (neutral) and protons (positive). Bohr model : a mechanical ...
... scintillation: momentary flash caused by a particle being absorbed and re-emitted by certain materials. Geiger counter : a device commonly used to detect radiation. nucleus: positively charged, dense core of the atom consisting of neutrons (neutral) and protons (positive). Bohr model : a mechanical ...
Chapter 28 notes
... scintillation: momentary flash caused by a particle being absorbed and re-emitted by certain materials. Geiger counter : a device commonly used to detect radiation. nucleus: positively charged, dense core of the atom consisting of neutrons (neutral) and protons (positive). Bohr model : a mechanical ...
... scintillation: momentary flash caused by a particle being absorbed and re-emitted by certain materials. Geiger counter : a device commonly used to detect radiation. nucleus: positively charged, dense core of the atom consisting of neutrons (neutral) and protons (positive). Bohr model : a mechanical ...
- BUGS McGill
... the energy, degeneracy and the quantum numbers of the state(s) that derive from the original 27h2/8ma2 energy level. ...
... the energy, degeneracy and the quantum numbers of the state(s) that derive from the original 27h2/8ma2 energy level. ...
CHM 111: General Physical Chemistry 3 Units
... Historical development of the atom: definition of atoms, Daltons atomic theory, relative atomic masses. Fundamental particles of the atom and atomic structure. Modern electronic theory of atoms; electronic configuration of the elements. Periodicity of the elements. Radioactivity: Stoichiometry: mole ...
... Historical development of the atom: definition of atoms, Daltons atomic theory, relative atomic masses. Fundamental particles of the atom and atomic structure. Modern electronic theory of atoms; electronic configuration of the elements. Periodicity of the elements. Radioactivity: Stoichiometry: mole ...
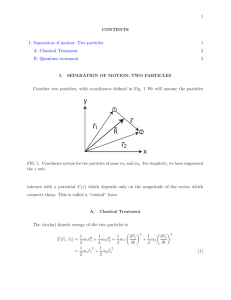
Separation of internal and center-of
... Now, let us define two new coordinates: the position of the center-of-mass ~ = m1 ~r1 + m2 ~r2 R M M ~ is the mass-weighted average of the positions of the two where M = m1 + m2 (note that R particles) and the separation between the two particles ~r = ~r2 − ~r1 ~ and ~r, the positions of the two par ...
... Now, let us define two new coordinates: the position of the center-of-mass ~ = m1 ~r1 + m2 ~r2 R M M ~ is the mass-weighted average of the positions of the two where M = m1 + m2 (note that R particles) and the separation between the two particles ~r = ~r2 − ~r1 ~ and ~r, the positions of the two par ...
energy levels
... energy levels in an atom – Tells you how far away the electron is from the nucleus – There are 1-7 energy levels, correlates with period numbers – Each level has same n number of sublevels – Maximum number of 2n2 electrons per level ...
... energy levels in an atom – Tells you how far away the electron is from the nucleus – There are 1-7 energy levels, correlates with period numbers – Each level has same n number of sublevels – Maximum number of 2n2 electrons per level ...
Chapter 2 - profpaz.com
... The law of definite proportions states that all samples of a given compound, regardless of their source or preparation, have the same proportions of their constituent elements. ...
... The law of definite proportions states that all samples of a given compound, regardless of their source or preparation, have the same proportions of their constituent elements. ...
BS5-Ch 2.
... the different masses of one element that combine with the same mass of the other element are in the ratio of small whole numbers. • cements the idea that atoms react as complete (whole) particles. • chemical formulas indicate whole numbers of atoms- not fractions ...
... the different masses of one element that combine with the same mass of the other element are in the ratio of small whole numbers. • cements the idea that atoms react as complete (whole) particles. • chemical formulas indicate whole numbers of atoms- not fractions ...
Grade 11 Chemistry E.. - hrsbstaff.ednet.ns.ca
... g. Na2SO4(aq) + BaCl2(aq) → BaSO4(s) + NaCl(aq) h. CH3OH(l) + O2(g) → CO2(g) + H2O(g) 25. Classify each of the above according to the 5 types of reactions (composition, decomposition, single replacement, double replacement and combustion). 26. Write the formula for each material correctly and then b ...
... g. Na2SO4(aq) + BaCl2(aq) → BaSO4(s) + NaCl(aq) h. CH3OH(l) + O2(g) → CO2(g) + H2O(g) 25. Classify each of the above according to the 5 types of reactions (composition, decomposition, single replacement, double replacement and combustion). 26. Write the formula for each material correctly and then b ...
Midterm Review 2017
... electrons. An ion of the same element has a total of 10 electrons. Which statement describes the charge and radius of the ion? 1) The ion is positively charged and its radius is smaller than the radius of the atom. 2) The ion is positively charged and its radius is larger than the radius of the atom ...
... electrons. An ion of the same element has a total of 10 electrons. Which statement describes the charge and radius of the ion? 1) The ion is positively charged and its radius is smaller than the radius of the atom. 2) The ion is positively charged and its radius is larger than the radius of the atom ...
03 Atoms – Nuclides
... a positively charged alpha particle (α), which is the same as a helium nuclei consisting of two neutrons and two protons a negatively charged beta minus particle (β-), which is the same as an electron a positively charged beta plus particle (β+), which is the same as a positron, a particle of equal ...
... a positively charged alpha particle (α), which is the same as a helium nuclei consisting of two neutrons and two protons a negatively charged beta minus particle (β-), which is the same as an electron a positively charged beta plus particle (β+), which is the same as a positron, a particle of equal ...
Kvantfysik Lecture Notes No. 4x
... We can naively understand why we cannot have Bohr’s ` = 0 state. This would correspond to an electron at rest sitting on top of the proton. If this were the case, then we would know both the position and momentum of the electron exactly, violating Heisenberg uncertainty. It turns out that the ` in ( ...
... We can naively understand why we cannot have Bohr’s ` = 0 state. This would correspond to an electron at rest sitting on top of the proton. If this were the case, then we would know both the position and momentum of the electron exactly, violating Heisenberg uncertainty. It turns out that the ` in ( ...
Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.The word atom comes from the Ancient Greek adjective atomos, meaning ""uncuttable"". 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. While seemingly apropos, around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called ""uncuttable atom"" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all. Since atoms were found to be divisible, physicists later invented the term ""elementary particles"" to describe the ""uncuttable"", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.