ChemicalBondingPowerpoint
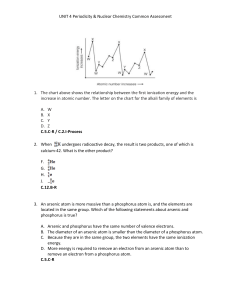
... Each element has a characteristic number of protons. The number of neutrons can vary; forms of an element with different numbers of neutrons are called isotopes. ...
... Each element has a characteristic number of protons. The number of neutrons can vary; forms of an element with different numbers of neutrons are called isotopes. ...
Laboratory Exercise: The Electronic Structure of the Hydrogen Atom
... In this laboratory exercise, we will probe the behavior of electrons within atoms using Emission Spectroscopy. In particular, we will focus on the behavior of the electron in the simplest atom, Hydrogen, and this atom's emission spectrum. For comparison, we will look at the emission spectrum of the ...
... In this laboratory exercise, we will probe the behavior of electrons within atoms using Emission Spectroscopy. In particular, we will focus on the behavior of the electron in the simplest atom, Hydrogen, and this atom's emission spectrum. For comparison, we will look at the emission spectrum of the ...
Bio_130_files/Chemistry Review
... – Elements are the simplest form of matter with unique chemical properties. They are charted on the periodic table based on some of their chemical characteristics. • There are 24 major elements that have various roles in the body. – These include structural, enzymatic, and homeostatic balance. • Com ...
... – Elements are the simplest form of matter with unique chemical properties. They are charted on the periodic table based on some of their chemical characteristics. • There are 24 major elements that have various roles in the body. – These include structural, enzymatic, and homeostatic balance. • Com ...
what is energy?
... SPACE AS A PHOTON (PACKET OF ENERGY). • IT IS PART OF THE ELECTROMAGNETIC SPECTRUM. • LIGHT CAN BE REFLECTED, REFRACTED AND DIFFRACTED ...
... SPACE AS A PHOTON (PACKET OF ENERGY). • IT IS PART OF THE ELECTROMAGNETIC SPECTRUM. • LIGHT CAN BE REFLECTED, REFRACTED AND DIFFRACTED ...
atomic structure + mass spectrometer
... i) 2.5 moles of Ammonia molecules, NH3 ii) 0.3 moles of Rubidium atoms, Rb iii) 1.51 moles of Bromine molecules, Br2 iv) 4.275 moles of hydrated sodium carbonate, Na2CO3.10H2O 3. How many moles are there in? i) 140g of Calcium atoms, Ca ii) 8.34g of Silicon dioxide molecules, SiO2 iii) 0.522g of Cop ...
... i) 2.5 moles of Ammonia molecules, NH3 ii) 0.3 moles of Rubidium atoms, Rb iii) 1.51 moles of Bromine molecules, Br2 iv) 4.275 moles of hydrated sodium carbonate, Na2CO3.10H2O 3. How many moles are there in? i) 140g of Calcium atoms, Ca ii) 8.34g of Silicon dioxide molecules, SiO2 iii) 0.522g of Cop ...
NYS Regents Chemistry June 21, 2002
... 1: II. PERIODIC TABLE\1. Properties of Elements\A. Metals\1. Metals - (32) 2: II. PERIODIC TABLE\2. Valence Electrons\A. Electron / Ionic Configuration\2. Ionic Configuration - (10, 30) 2: II. PERIODIC TABLE\4. Properties of Periods\C. Electronegativity\1. Electronegativity - (11, 13) 1: II. PERIODI ...
... 1: II. PERIODIC TABLE\1. Properties of Elements\A. Metals\1. Metals - (32) 2: II. PERIODIC TABLE\2. Valence Electrons\A. Electron / Ionic Configuration\2. Ionic Configuration - (10, 30) 2: II. PERIODIC TABLE\4. Properties of Periods\C. Electronegativity\1. Electronegativity - (11, 13) 1: II. PERIODI ...
Glossary Chapter 4
... electromagnetic radiation a form of energy that exhibits wavelike behavior as it travels through space (91) electromagnetic spectrum all the forms of electromagnetic radiation (91) electron configuration the arrangement of electrons in an atom (105) excited state a state in which an atom has a highe ...
... electromagnetic radiation a form of energy that exhibits wavelike behavior as it travels through space (91) electromagnetic spectrum all the forms of electromagnetic radiation (91) electron configuration the arrangement of electrons in an atom (105) excited state a state in which an atom has a highe ...
first chapter - damtp - University of Cambridge
... some of the energy, W , is absorbed by the other particles in the metal.) However, a free particle can scatter the photon, so changing its energy and therefore its frequency; at the same time the particle recoils. The kinematics of the process can be worked out using (1.2), (1.4) and the relativisti ...
... some of the energy, W , is absorbed by the other particles in the metal.) However, a free particle can scatter the photon, so changing its energy and therefore its frequency; at the same time the particle recoils. The kinematics of the process can be worked out using (1.2), (1.4) and the relativisti ...
Spectra and atomic structure
... by considering gases. Looking at light emitted by a source through a prism or a diffraction grating can tell us a great deal, not only what the substance is but also much about its atomic structure! Emission spectra You should have a look at various spectra (some of which are shown in the photograph ...
... by considering gases. Looking at light emitted by a source through a prism or a diffraction grating can tell us a great deal, not only what the substance is but also much about its atomic structure! Emission spectra You should have a look at various spectra (some of which are shown in the photograph ...
All you need to know about Additional Science
... If we have a solution containing 100 g of sodium hydroxide, how much chlorine gas should we pass through the solution to make bleach? Too much, and some chlorine will be wasted, too little and not all of the sodium hydroxide will react. ...
... If we have a solution containing 100 g of sodium hydroxide, how much chlorine gas should we pass through the solution to make bleach? Too much, and some chlorine will be wasted, too little and not all of the sodium hydroxide will react. ...
Lesson 1 - Working With Chemicals
... o A nucleus – a central region that is positively charged and contains most of the mass - protons are heavy positive particles within the nucleus o Electrons – particles with a negative charge and are very light (compared to protons). - Electrons circle around the nucleus o Empty space surrounding t ...
... o A nucleus – a central region that is positively charged and contains most of the mass - protons are heavy positive particles within the nucleus o Electrons – particles with a negative charge and are very light (compared to protons). - Electrons circle around the nucleus o Empty space surrounding t ...
4_POSER_FAEN
... observed to cause electrons to be ejected from a metal's surface. The classical explanation was that the metal's electrons would oscillate with the light and eventually break a way from the surface with a kinetic energy that would depend on the intensity of the incident radiation. However, the kinet ...
... observed to cause electrons to be ejected from a metal's surface. The classical explanation was that the metal's electrons would oscillate with the light and eventually break a way from the surface with a kinetic energy that would depend on the intensity of the incident radiation. However, the kinet ...
Chemical equations must be balanced.
... This equation is not balanced. There is one C on each side of the equation, so C is balanced. However, on the left side, H has a subscript of 4, which means there are four hydrogen atoms. On the right side, H has a subscript of 2, which means there are two hydrogen atoms. Also, there are two oxygen ...
... This equation is not balanced. There is one C on each side of the equation, so C is balanced. However, on the left side, H has a subscript of 4, which means there are four hydrogen atoms. On the right side, H has a subscript of 2, which means there are two hydrogen atoms. Also, there are two oxygen ...
Chapter 5/6 Notes
... BIG PROBLEM: **Rutherford’s atomic model could not explain the chemical properties of elements.** ...
... BIG PROBLEM: **Rutherford’s atomic model could not explain the chemical properties of elements.** ...
Atomic number - River Dell Regional School District
... D. Joseph Proust (1779) Develops Law of Definite Composition- all samples of a specific substance contain the same mass ratio of the same elements a. ex: all samples of CO2 contains 27.3% carbon and 72.7% oxygen b. therefore ‘elements’ are combining in a whole number ratio ...
... D. Joseph Proust (1779) Develops Law of Definite Composition- all samples of a specific substance contain the same mass ratio of the same elements a. ex: all samples of CO2 contains 27.3% carbon and 72.7% oxygen b. therefore ‘elements’ are combining in a whole number ratio ...
The Trouble with Gravity Summary/Review
... – Direct measurements of the amount of dark and luminous matter imply one last constraint. Together, all three of these constraints imply a small, positive cosmological constant. – We don’t have a complete understanding of all the sources of the vacuum energy density, but if we look at the quantum ...
... – Direct measurements of the amount of dark and luminous matter imply one last constraint. Together, all three of these constraints imply a small, positive cosmological constant. – We don’t have a complete understanding of all the sources of the vacuum energy density, but if we look at the quantum ...
r - Purdue Physics
... Planck’s relation and its interpretation by Einstein suggests that the discrete spectral lines of H 2 gas ( See Fig. 18.17 on page 399) with fixed can be interpreted as an electron transition from one stable orbit with energy level E1 ( r1 ) with radius r1 to another stable orbit with energy leve ...
... Planck’s relation and its interpretation by Einstein suggests that the discrete spectral lines of H 2 gas ( See Fig. 18.17 on page 399) with fixed can be interpreted as an electron transition from one stable orbit with energy level E1 ( r1 ) with radius r1 to another stable orbit with energy leve ...
NOTES Atomic Structure Number Mass.docx
... are using it. If the basis for sorting is carefully chosen, classification may also reveal relationships that would go unnoticed. The best basis for sorting data depends on how they will be used and who will be using them. Initially, scientists tried classifying elements on the basis of their proper ...
... are using it. If the basis for sorting is carefully chosen, classification may also reveal relationships that would go unnoticed. The best basis for sorting data depends on how they will be used and who will be using them. Initially, scientists tried classifying elements on the basis of their proper ...
Early Modern Physics
... • Late 19th Century: try to derive Wien and StefanBoltzman Laws and shape of observed light spectra • used Statistical Mechanics (we’ll do later in 461) to determine relative probability for any wavelength l • need::number of states (“nodes”) for any l - energy of any state probability versus energy ...
... • Late 19th Century: try to derive Wien and StefanBoltzman Laws and shape of observed light spectra • used Statistical Mechanics (we’ll do later in 461) to determine relative probability for any wavelength l • need::number of states (“nodes”) for any l - energy of any state probability versus energy ...
Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.The word atom comes from the Ancient Greek adjective atomos, meaning ""uncuttable"". 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. While seemingly apropos, around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called ""uncuttable atom"" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all. Since atoms were found to be divisible, physicists later invented the term ""elementary particles"" to describe the ""uncuttable"", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.