Intro to Chapter 5 Development of the Periodic Table
... What properties of atoms is responsible for the periodic variations? To understand how, it s necessary to look first at the nature of visible line and other forms of radiant energy. Historically, studies of the interaction of radiant energy with matter provided immense insight into the atomic struct ...
... What properties of atoms is responsible for the periodic variations? To understand how, it s necessary to look first at the nature of visible line and other forms of radiant energy. Historically, studies of the interaction of radiant energy with matter provided immense insight into the atomic struct ...
Проф - Atomic physics department
... symmetry breaking. Higgs boson. Neutrinos -masses and oscillations. History of the early Universe. Seminars/Practical exercises 1 Units in the atomic and nuclear physics. Lorenz transformations. Relativistic kinematics. 2 Wave and corpuscular properties of the light. 3 Atomic spectra. Rydberg consta ...
... symmetry breaking. Higgs boson. Neutrinos -masses and oscillations. History of the early Universe. Seminars/Practical exercises 1 Units in the atomic and nuclear physics. Lorenz transformations. Relativistic kinematics. 2 Wave and corpuscular properties of the light. 3 Atomic spectra. Rydberg consta ...
qp2
... would be where it could form a closed circular standing wave, where the ends meet up as if in a perfect circle. In such a case, the circumference would have to be an integer multiple of the electrons wavelength i.e. 2πr=nλ, where n is any integer (see figure). This explains why electrons could only ...
... would be where it could form a closed circular standing wave, where the ends meet up as if in a perfect circle. In such a case, the circumference would have to be an integer multiple of the electrons wavelength i.e. 2πr=nλ, where n is any integer (see figure). This explains why electrons could only ...
The Pauli-Exclusion Principle Indistinguishability
... anti-symmetric combination of hydrogen orbitals. Thus, we can obtain an approximate solution and learn a lot about atomic structure if we just neglect electron repulsion compared to the nuclear attraction. The ground state solution is approximated by filling hydrogen orbitals with electrons starting ...
... anti-symmetric combination of hydrogen orbitals. Thus, we can obtain an approximate solution and learn a lot about atomic structure if we just neglect electron repulsion compared to the nuclear attraction. The ground state solution is approximated by filling hydrogen orbitals with electrons starting ...
South Pasadena • AP Chemistry
... 12. “Wafting” is the proper technique for a) neutralizing a spilled acid. b) putting out burning clothing. c) washing chemicals from the eye. d) smelling a chemical substance. e) observing the color of a chemical. 13. You measure the density of a slab of lead as 11.10 g/mL. The accepted value is 11. ...
... 12. “Wafting” is the proper technique for a) neutralizing a spilled acid. b) putting out burning clothing. c) washing chemicals from the eye. d) smelling a chemical substance. e) observing the color of a chemical. 13. You measure the density of a slab of lead as 11.10 g/mL. The accepted value is 11. ...
Lecture XIII_XIV
... hits it. Therefore it reflects no radiation and appears perfectly black. It is also a perfect emitter of radiation. At a particular temperature the black body would emit the maximum amount of energy possible for that temperature. ...
... hits it. Therefore it reflects no radiation and appears perfectly black. It is also a perfect emitter of radiation. At a particular temperature the black body would emit the maximum amount of energy possible for that temperature. ...
Chapter 2
... • _________ -a substance that cannot be broken down to other substances by chemical reactions. • -92 naturally-occurring elements. • Each element has a unique symbol, usually from the first one or two letters of the name, often from Latin or German. ...
... • _________ -a substance that cannot be broken down to other substances by chemical reactions. • -92 naturally-occurring elements. • Each element has a unique symbol, usually from the first one or two letters of the name, often from Latin or German. ...
Honors Chemistry Ms. K Pages 66
... for this relative scale has been chosen as the carbon-12 isotope. Carbon-12 has been arbitrarily assigned a mass of exactly 12 atomic mass units or amu. atomic mass unit (amu) ? 1/12 the mass of a carbon-12 atom The atomic mass of any atom is determined by comparing the mass of that atom to the mass ...
... for this relative scale has been chosen as the carbon-12 isotope. Carbon-12 has been arbitrarily assigned a mass of exactly 12 atomic mass units or amu. atomic mass unit (amu) ? 1/12 the mass of a carbon-12 atom The atomic mass of any atom is determined by comparing the mass of that atom to the mass ...
PHYS 481/681 Quantum Mechanics Stephen Lepp August 29, 2016
... Introduction to Quantum Mechanics nd the interpretation of its solutions, the uncertainty principles, one-dimensional problems, harmonic oscillator, angular momentum, the hydrogen atom. 3 credits. • Class MW 11:30-12:45 BPB 249. • Office Hours TTh 12:45-1:30 or by arrangement. • Textbook “Quantum Me ...
... Introduction to Quantum Mechanics nd the interpretation of its solutions, the uncertainty principles, one-dimensional problems, harmonic oscillator, angular momentum, the hydrogen atom. 3 credits. • Class MW 11:30-12:45 BPB 249. • Office Hours TTh 12:45-1:30 or by arrangement. • Textbook “Quantum Me ...
Document
... Quantum Hall edge states with a Bose gas Ultracold atomic systems can be used to model condensed-matter physics, providing precise control of system variables often not achievable in real materials. This involves inducing charge-neutral particles to behave as if they were charged particles in a magn ...
... Quantum Hall edge states with a Bose gas Ultracold atomic systems can be used to model condensed-matter physics, providing precise control of system variables often not achievable in real materials. This involves inducing charge-neutral particles to behave as if they were charged particles in a magn ...
chem final review
... 28) Why did J. J. Thomson reason that electrons must be a part of the atoms of all elements? A) Cathode rays were always accompanied by anode rays. B) Cathode rays can be deflected by magnets. C) Cathode rays are always made of electrons, regardless of the gas used. D) Cathode rays are negatively-ch ...
... 28) Why did J. J. Thomson reason that electrons must be a part of the atoms of all elements? A) Cathode rays were always accompanied by anode rays. B) Cathode rays can be deflected by magnets. C) Cathode rays are always made of electrons, regardless of the gas used. D) Cathode rays are negatively-ch ...
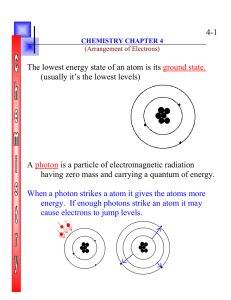
4-1 The lowest energy state of an atom is its ground state. (usually
... (usually it’s the lowest levels) ...
... (usually it’s the lowest levels) ...
Physics 272: Electricity and Magnetism
... are positive or negative ions, or electrons • Amount of energy required to remove bare nuclei or protons from inside the surface atoms is enormous ...
... are positive or negative ions, or electrons • Amount of energy required to remove bare nuclei or protons from inside the surface atoms is enormous ...
Metals
... •Ordered elements by atomic mass •Repeating pattern of properties •Elements with similar properties in the same column Periodic Law – when the elements are arranged in order of increasing atomic mass, certain sets of ...
... •Ordered elements by atomic mass •Repeating pattern of properties •Elements with similar properties in the same column Periodic Law – when the elements are arranged in order of increasing atomic mass, certain sets of ...
Chemical Principles – by Steven Zumdahl (5 ) Chapter 1
... States of Matter: We classify matter into several states because it helps us classify information in a systematic way. Solid State: Solids are rigid. They have definite shapes. The dimensions of solids change slightly with temperature and pressure. Liquid State: Liquids flow and assume the shape of ...
... States of Matter: We classify matter into several states because it helps us classify information in a systematic way. Solid State: Solids are rigid. They have definite shapes. The dimensions of solids change slightly with temperature and pressure. Liquid State: Liquids flow and assume the shape of ...
Waves - Valdosta State University
... • Energy states correspond to orbits • Orbits are numbered: principal quantum number (n) • n = 1, is closest to nucleus (lowest energy) • Electrons can only move between orbits by absorbing and emitting energy in quanta (h). • Ground State –electron is in its lowest energy orbit. • Excited State – ...
... • Energy states correspond to orbits • Orbits are numbered: principal quantum number (n) • n = 1, is closest to nucleus (lowest energy) • Electrons can only move between orbits by absorbing and emitting energy in quanta (h). • Ground State –electron is in its lowest energy orbit. • Excited State – ...
- Aboriginal Access to Engineering
... Chemistry is one of the subjects which engineers study in a pure, theoretical form and then apply to solve problems in the real world. For instance, did you know that the making of steel is chemistry? Steel is a very strong and versatile metal. It is used in hundreds of ways, to reinforce bridges an ...
... Chemistry is one of the subjects which engineers study in a pure, theoretical form and then apply to solve problems in the real world. For instance, did you know that the making of steel is chemistry? Steel is a very strong and versatile metal. It is used in hundreds of ways, to reinforce bridges an ...
Atomic theory
In chemistry and physics, atomic theory is a scientific theory of the nature of matter, which states that matter is composed of discrete units called atoms. It began as a philosophical concept in ancient Greece and entered the scientific mainstream in the early 19th century when discoveries in the field of chemistry showed that matter did indeed behave as if it were made up of atoms.The word atom comes from the Ancient Greek adjective atomos, meaning ""uncuttable"". 19th century chemists began using the term in connection with the growing number of irreducible chemical elements. While seemingly apropos, around the turn of the 20th century, through various experiments with electromagnetism and radioactivity, physicists discovered that the so-called ""uncuttable atom"" was actually a conglomerate of various subatomic particles (chiefly, electrons, protons and neutrons) which can exist separately from each other. In fact, in certain extreme environments, such as neutron stars, extreme temperature and pressure prevents atoms from existing at all. Since atoms were found to be divisible, physicists later invented the term ""elementary particles"" to describe the ""uncuttable"", though not indestructible, parts of an atom. The field of science which studies subatomic particles is particle physics, and it is in this field that physicists hope to discover the true fundamental nature of matter.