Packet #7- Chemical Reactions
... the total mass of products after the reaction is the same as the total mass of the reactants at the start. This fact allows you to work out the mass of one substance in a reaction if the masses of the other substances are known. For example: Carbon reacts with oxygen to form carbon dioxide: C + O2 → ...
... the total mass of products after the reaction is the same as the total mass of the reactants at the start. This fact allows you to work out the mass of one substance in a reaction if the masses of the other substances are known. For example: Carbon reacts with oxygen to form carbon dioxide: C + O2 → ...
Regents Exam In Chemistry Review Homework #1
... ______________________________________________________________________________________________ ...
... ______________________________________________________________________________________________ ...
mechanisms - Manasquan Public Schools
... • But this gives us no info on HOW FAST reaction goes from reactants to products. ...
... • But this gives us no info on HOW FAST reaction goes from reactants to products. ...
FINAL EXAM Spring 2012
... 10) Determine the pH of the following solution. Initial concentrations are given. [HF] = 1.296 M, [HCl] = 1.045 M, Ka for HF is 6.6 × 10-4 A) -0.019 B) 0.60 C) 1.3 D) 3.1 E) 3.2 11) When 0.10 mol of solid silver nitrate is added to 1.0 L of a clear, saturated solution of Ag2CrO4 (Ksp = 2.4 x 10-12), ...
... 10) Determine the pH of the following solution. Initial concentrations are given. [HF] = 1.296 M, [HCl] = 1.045 M, Ka for HF is 6.6 × 10-4 A) -0.019 B) 0.60 C) 1.3 D) 3.1 E) 3.2 11) When 0.10 mol of solid silver nitrate is added to 1.0 L of a clear, saturated solution of Ag2CrO4 (Ksp = 2.4 x 10-12), ...
Class Notes
... only those particles that are directly involved in the chemical change. Net ionic equations differentiate between ions that react to form a precipitate, a gas, or water, and ions that simply remain in aqueous solution. In the example above, the NO3-1(aq) and the Na+1(aq) are both spectator ions. The ...
... only those particles that are directly involved in the chemical change. Net ionic equations differentiate between ions that react to form a precipitate, a gas, or water, and ions that simply remain in aqueous solution. In the example above, the NO3-1(aq) and the Na+1(aq) are both spectator ions. The ...
AP Review Chp 1 and Chp 2 Wed 10/9/2013 1. Near room
... I) Magnesium displaces copper from a dilute solution of copper ( II) sulfate; the pure copper will settle out of the solution. Mg(s) + CuSO4(aq) MgSO4(aq) + Cu(s) A copper(II) sulfate solution is mixed by dissolving 25.000 g of copper(II) sulfate, and then it is treated with an excess of magnesium ...
... I) Magnesium displaces copper from a dilute solution of copper ( II) sulfate; the pure copper will settle out of the solution. Mg(s) + CuSO4(aq) MgSO4(aq) + Cu(s) A copper(II) sulfate solution is mixed by dissolving 25.000 g of copper(II) sulfate, and then it is treated with an excess of magnesium ...
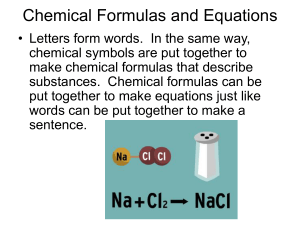
Chemical Formulas and Equations
... Chemical Formulas and Equations • Letters form words. In the same way, chemical symbols are put together to make chemical formulas that describe substances. Chemical formulas can be put together to make equations just like words can be put together to make a ...
... Chemical Formulas and Equations • Letters form words. In the same way, chemical symbols are put together to make chemical formulas that describe substances. Chemical formulas can be put together to make equations just like words can be put together to make a ...
Final Exam Review- no solutions
... sodium hydroxide in 25.00 mL of NaOH solution. What is the molarity of the NaOH solution? ...
... sodium hydroxide in 25.00 mL of NaOH solution. What is the molarity of the NaOH solution? ...
EOC Review - Dorman Freshman Campus
... created nor destroyed •The amount of mass you started with before the reaction is the same after the reaction. •Ex: burning a log ...
... created nor destroyed •The amount of mass you started with before the reaction is the same after the reaction. •Ex: burning a log ...
Chapter 3 Powerpoint
... 1023), but 1 mole sample of different substances will have different masses. • The mass of a single atom of an element (in amu) is numerically equal to the mass (in grams) of 1 mole of that element. • The mass in grams of 1 mole of a substance is called the molar mass of the substance. ...
... 1023), but 1 mole sample of different substances will have different masses. • The mass of a single atom of an element (in amu) is numerically equal to the mass (in grams) of 1 mole of that element. • The mass in grams of 1 mole of a substance is called the molar mass of the substance. ...
Name: Northwest Vista College Chem 1311
... 44. (6 pts) How many liters of chlorine gas at 25°C and 0.950 atm can be produced by the reaction of 12.0 g of MnO2 with excess HCl(aq) according to the following chemical equation? MnO2(s) + 4HCl(aq) MnCl2(aq) + 2H2O(l) + Cl2(g) Answer: First find the number of moles Cl2 formed from the reaction ...
... 44. (6 pts) How many liters of chlorine gas at 25°C and 0.950 atm can be produced by the reaction of 12.0 g of MnO2 with excess HCl(aq) according to the following chemical equation? MnO2(s) + 4HCl(aq) MnCl2(aq) + 2H2O(l) + Cl2(g) Answer: First find the number of moles Cl2 formed from the reaction ...
Name - Deans Community High School
... b) Is the forward reaction is exothermic or endothermic. ............................................ 1 c) Gold and platinum both catalyse the reaction. For the forward reaction EA using gold is 30 kJ, while EA using platinum is 40 kJ. i) using different dotted lines add this information to the grap ...
... b) Is the forward reaction is exothermic or endothermic. ............................................ 1 c) Gold and platinum both catalyse the reaction. For the forward reaction EA using gold is 30 kJ, while EA using platinum is 40 kJ. i) using different dotted lines add this information to the grap ...
CHEM 1405 Practice Exam 3 (2015)
... 6) How many moles of chlorine gas react with 1 mol of hydrogen gas according to the balanced chemical equation? H2(g) + Cl2(g) → 2 HCl(g) A) 1 mol ...
... 6) How many moles of chlorine gas react with 1 mol of hydrogen gas according to the balanced chemical equation? H2(g) + Cl2(g) → 2 HCl(g) A) 1 mol ...
Chapter 6 Chemical Reactions: An Introduction
... 2 x [2 NH3(g) + 2.5 O2(g) → 2 NO(g) + 3 H2O(g)] 4 NH3(g) + 5 O2(g) → 4 NO(g) + 6 H2O(g) ...
... 2 x [2 NH3(g) + 2.5 O2(g) → 2 NO(g) + 3 H2O(g)] 4 NH3(g) + 5 O2(g) → 4 NO(g) + 6 H2O(g) ...
Ch 19 test_take-home
... 9) In the Haber process, ammonia is synthesized from nitrogen and hydrogen: N2 (g) + 3H2 (g) → 2NH3 (g) △G° at 298 oK for this reaction is -33.3 kJ/mol. What is the value of △G at 298 K for a reaction mixture that consists of 1.9 atm N2, 1.6 atm H2, and 0.65 atm NH3? ...
... 9) In the Haber process, ammonia is synthesized from nitrogen and hydrogen: N2 (g) + 3H2 (g) → 2NH3 (g) △G° at 298 oK for this reaction is -33.3 kJ/mol. What is the value of △G at 298 K for a reaction mixture that consists of 1.9 atm N2, 1.6 atm H2, and 0.65 atm NH3? ...
Questions 1-2
... HOBr is a weaker acid than HBrO3. Account for this fact in terms of molecular structure. ...
... HOBr is a weaker acid than HBrO3. Account for this fact in terms of molecular structure. ...
Study Guide – Unit Test (9-27-13)
... There will be other examples included on the test. (Look at notes/old quizzes and worksheets) ...
... There will be other examples included on the test. (Look at notes/old quizzes and worksheets) ...
Chemical reactions alter arrangements of atoms.
... reaction. In the burning of natural gas, for example, methane (CH4) and oxygen (O2) are the reactants in the chemical reaction. Products are the substances formed by a chemical reaction. In the burning of natural gas, carbon dioxide (CO2) and water (H2O) are the products formed by the reaction. Reac ...
... reaction. In the burning of natural gas, for example, methane (CH4) and oxygen (O2) are the reactants in the chemical reaction. Products are the substances formed by a chemical reaction. In the burning of natural gas, carbon dioxide (CO2) and water (H2O) are the products formed by the reaction. Reac ...
Stoichiometry notes 1
... Can be used as a conversion factor between moles and Number of representative particles. Reminder: The representative particles for 1. an element is an atom 2. for a molecular compound is a molecule 3. for an ionic compound is a formula unit ...
... Can be used as a conversion factor between moles and Number of representative particles. Reminder: The representative particles for 1. an element is an atom 2. for a molecular compound is a molecule 3. for an ionic compound is a formula unit ...
Lecture 8
... A 30.5-g sample of acrylic acid, used in the manufacture of acrylic plastics, is found to contain 15.25 g C, 1.71 g H, and 13.54 g O. In a separate mass spectrometer experiment, the acrylic acid is found to have a molar mass of approximately 72 g/mol. What are the empirical and molecular formulas of ...
... A 30.5-g sample of acrylic acid, used in the manufacture of acrylic plastics, is found to contain 15.25 g C, 1.71 g H, and 13.54 g O. In a separate mass spectrometer experiment, the acrylic acid is found to have a molar mass of approximately 72 g/mol. What are the empirical and molecular formulas of ...
Introduction to Stoichiometry
... What is Stoichiometry? The proportional relationship between two or more substances during a chemical reaction. In other words, using dimensional analysis to convert one substance to another There are many different types, but they are all similar. So, let’s start small. How small? ...
... What is Stoichiometry? The proportional relationship between two or more substances during a chemical reaction. In other words, using dimensional analysis to convert one substance to another There are many different types, but they are all similar. So, let’s start small. How small? ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.