PPT - Unit 5
... -(C2H2(g) + 5/2O2(g) → 2CO2(g) + H2O(l) ΔH = -1300. kJ) 2( C(s) + O2(g) → CO2(g) ) 2(ΔH = -394 kJ) H2(g) + 1/2O2(g) → H2O(l) ΔH = -286 kJ Calculate ΔH for the following reaction: 2C(s) + H2(g) → C2H2(g) 2C(s) + 2O2(g) → 2CO2(g) ΔH = -788 kJ 2CO2(g) + H2O(l) → C2H2(g) + 5/2O2(g) ΔH = +1300 kJ H2(g) + ...
... -(C2H2(g) + 5/2O2(g) → 2CO2(g) + H2O(l) ΔH = -1300. kJ) 2( C(s) + O2(g) → CO2(g) ) 2(ΔH = -394 kJ) H2(g) + 1/2O2(g) → H2O(l) ΔH = -286 kJ Calculate ΔH for the following reaction: 2C(s) + H2(g) → C2H2(g) 2C(s) + 2O2(g) → 2CO2(g) ΔH = -788 kJ 2CO2(g) + H2O(l) → C2H2(g) + 5/2O2(g) ΔH = +1300 kJ H2(g) + ...
Chapter 11 Chemical Reactions
... Nitric acid dissolved in water reacts with solid sodium carbonate to form liquid water and carbon dioxide gas and sodium nitrate dissolved in water. ...
... Nitric acid dissolved in water reacts with solid sodium carbonate to form liquid water and carbon dioxide gas and sodium nitrate dissolved in water. ...
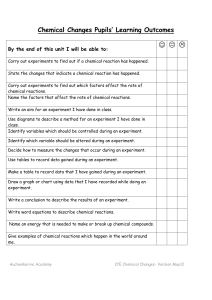
Chemical Reactions Unit Pupils` Learning Outcomes
... Identify variables which should be controlled during an experiment. Identify which variable should be altered during an experiment. Decide how to measure the changes that occur during an experiment. Use tables to record data gained during an experiment. Make a table to record data that I have gained ...
... Identify variables which should be controlled during an experiment. Identify which variable should be altered during an experiment. Decide how to measure the changes that occur during an experiment. Use tables to record data gained during an experiment. Make a table to record data that I have gained ...
Atomic Theory Practice Test
... a. the atoms share electrons evenly c. the atoms share electrons, but not evenly b. the atoms take or give electrons d. the atoms give or take protons ____ 16. Given the compound CO2, what type of bond is it? Electronegativities are C=2.5 O=3.5 ...
... a. the atoms share electrons evenly c. the atoms share electrons, but not evenly b. the atoms take or give electrons d. the atoms give or take protons ____ 16. Given the compound CO2, what type of bond is it? Electronegativities are C=2.5 O=3.5 ...
Chapter 1
... new white substance B and a gas C. The gas has exactly the same properties as the product obtained when carbon is burned in an excess of oxygen. Based on these observations, can we determine whether solids A and B and the gas C are elements or compounds? .A – compound B – cannot be determined, C - c ...
... new white substance B and a gas C. The gas has exactly the same properties as the product obtained when carbon is burned in an excess of oxygen. Based on these observations, can we determine whether solids A and B and the gas C are elements or compounds? .A – compound B – cannot be determined, C - c ...
Chemistry
... terms of moles and concentrations in terms of mol dm . Redox titrations are limited to manganate(VII) – iron(II) and iodine - thiosulfate. Back titration involving addition of known excess of acid or alkali to a reactant solution and determining the excess by titration. Gravimetric methods of analys ...
... terms of moles and concentrations in terms of mol dm . Redox titrations are limited to manganate(VII) – iron(II) and iodine - thiosulfate. Back titration involving addition of known excess of acid or alkali to a reactant solution and determining the excess by titration. Gravimetric methods of analys ...
Chapter 7
... • A chemical reaction is a chemical change resulting from a collision of atoms or molecules. • The original substances are reactants • The substances produced by the reaction are called products for example: carbon can collide with oxygen and make carbon dioxide Chemical Equation: ...
... • A chemical reaction is a chemical change resulting from a collision of atoms or molecules. • The original substances are reactants • The substances produced by the reaction are called products for example: carbon can collide with oxygen and make carbon dioxide Chemical Equation: ...
1. some basic concepts of chemistry
... Mole is the unit of amount of substance. It is defined as the amount of substance that contains as many particles as there are atoms in exactly 12 g C12 isotope. 1 mole of any substance contains 6.022 x 1023 atoms. This number is known as Avogadro number or Avogadro constant (NA or N0). 1 mol of hyd ...
... Mole is the unit of amount of substance. It is defined as the amount of substance that contains as many particles as there are atoms in exactly 12 g C12 isotope. 1 mole of any substance contains 6.022 x 1023 atoms. This number is known as Avogadro number or Avogadro constant (NA or N0). 1 mol of hyd ...
semester two review sheet
... arranged in the order that we learned the concepts. If there are problems you do not understand, or need additional practice on, please revisit that unit’s review sheet from my website. In addition to a periodic table, scientific calculator and ion chart, you may also use this HANDWRITTEN review she ...
... arranged in the order that we learned the concepts. If there are problems you do not understand, or need additional practice on, please revisit that unit’s review sheet from my website. In addition to a periodic table, scientific calculator and ion chart, you may also use this HANDWRITTEN review she ...
CHEMISTRY IM 06 SYLLABUS 1
... terms of moles and concentrations in terms of mol dm . Redox titrations are limited to manganate(VII) – iron(II) and iodine - thiosulfate. Back titration involving addition of known excess of acid or alkali to a reactant solution and determining the excess by titration. Gravimetric methods of analys ...
... terms of moles and concentrations in terms of mol dm . Redox titrations are limited to manganate(VII) – iron(II) and iodine - thiosulfate. Back titration involving addition of known excess of acid or alkali to a reactant solution and determining the excess by titration. Gravimetric methods of analys ...
exo and endo experiments
... The Law of Conservation of Mass The Law of Conservation of Mass was officially established in the year 1789 by the French Chemist, Antoine Lavoisier. The Law of Conservation of Mass states that mass is neither lost nor gained in chemical reactions, it states that it simply changes form. For that rea ...
... The Law of Conservation of Mass The Law of Conservation of Mass was officially established in the year 1789 by the French Chemist, Antoine Lavoisier. The Law of Conservation of Mass states that mass is neither lost nor gained in chemical reactions, it states that it simply changes form. For that rea ...
Exam Review - hrsbstaff.ednet.ns.ca
... The reaction of solutions of ammonium phosphate and barium nitrate gives a precipitate of barium phosphate. The equation that best represents this statement is a) 2(NH4)3PO4(s) + 3Ba(NO3)2(aq) → Ba3(PO4)2(aq) + 6NH4NO3(s). b) 2(NH4)3PO4(aq) + 3Ba(NO3)2(aq) → Ba3(PO4)2(s) + 6NH4NO3(aq). c) 2(NH4)3PO4 ...
... The reaction of solutions of ammonium phosphate and barium nitrate gives a precipitate of barium phosphate. The equation that best represents this statement is a) 2(NH4)3PO4(s) + 3Ba(NO3)2(aq) → Ba3(PO4)2(aq) + 6NH4NO3(s). b) 2(NH4)3PO4(aq) + 3Ba(NO3)2(aq) → Ba3(PO4)2(s) + 6NH4NO3(aq). c) 2(NH4)3PO4 ...
Chemistry I – Fall 2004
... 15. A pure substance melts at 113 °C and does not conduct electricity in either the solid or liquid state. What conclusions can be drawn concerning the bonding in this substance? ...
... 15. A pure substance melts at 113 °C and does not conduct electricity in either the solid or liquid state. What conclusions can be drawn concerning the bonding in this substance? ...
Practice Exam #2
... is __________, and therefore heat is __________ by the reaction. A) exothermic, released B) exothermic, absorbed C) endothermic, absorbed D) endothermic, released E) thermoneutral, neither released nor absorbed 17) The value of ΔE for a system that performs 213 kJ of work on its surroundings and los ...
... is __________, and therefore heat is __________ by the reaction. A) exothermic, released B) exothermic, absorbed C) endothermic, absorbed D) endothermic, released E) thermoneutral, neither released nor absorbed 17) The value of ΔE for a system that performs 213 kJ of work on its surroundings and los ...
CHEM 20 FINAL EXAM: STUDY HEADINGS Jan 2012
... the following equation: NH4NO3(s) → N2O(g) + 2 H2O(g) When 1.000 g of ammonium nitrate is decomposed in this way, 0.550 g of dinitrogen oxide is produced. What mass of water would also be produced in this reaction? a) 1.10 g b) 0.450 g c) 0.900 g d) 0.550 g ...
... the following equation: NH4NO3(s) → N2O(g) + 2 H2O(g) When 1.000 g of ammonium nitrate is decomposed in this way, 0.550 g of dinitrogen oxide is produced. What mass of water would also be produced in this reaction? a) 1.10 g b) 0.450 g c) 0.900 g d) 0.550 g ...
Chemistry Test Review - Greenslime Home Page
... used to describe it. b. Physical changes occur when you alter the shape or size of an object, but it is still made of the same “stuff” and with the same properties c. Chemical changes occur when materials react chemically with each other to form a new material with different properties than the orig ...
... used to describe it. b. Physical changes occur when you alter the shape or size of an object, but it is still made of the same “stuff” and with the same properties c. Chemical changes occur when materials react chemically with each other to form a new material with different properties than the orig ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.