Learning Activities
... 15. Know that science plays a role in many different kinds of careers and activities (e.g., public service, volunteers, public office holders, researchers, teachers, doctors, nurses, technicians, farmers, ranchers). ...
... 15. Know that science plays a role in many different kinds of careers and activities (e.g., public service, volunteers, public office holders, researchers, teachers, doctors, nurses, technicians, farmers, ranchers). ...
practice test2
... A) a reactant in a chemical reaction B) a product in a chemical reaction C) a substance that speeds up a reaction without being consumed in the reaction D) a substance that increases the energy of the products ...
... A) a reactant in a chemical reaction B) a product in a chemical reaction C) a substance that speeds up a reaction without being consumed in the reaction D) a substance that increases the energy of the products ...
Semester II Exam Review Questions
... 10. Aqueous sodium phosphate and liquid water are formed when aqueous sodium hydroxide reacts with phosphoric acid. ...
... 10. Aqueous sodium phosphate and liquid water are formed when aqueous sodium hydroxide reacts with phosphoric acid. ...
Welcome to AP Chemistry! I am glad that you decided to take on the
... Princeton Review’s “Cracking the AP Chemistry Exam.” You can also check my webpage under Pre-AP Skills for notes that will help you. If you have difficulty, you are more than welcome to call or email me at any point during the summer at the telephone number or email address below. If I am not at hom ...
... Princeton Review’s “Cracking the AP Chemistry Exam.” You can also check my webpage under Pre-AP Skills for notes that will help you. If you have difficulty, you are more than welcome to call or email me at any point during the summer at the telephone number or email address below. If I am not at hom ...
formula`s - Montville.net
... 17.2 Avogadro’s Number The Avogadro number was named in honor of Amedeo Avogadro who discovered that a mole of any gas under the same conditions has the same number of molecules. Johann Josef Loschmidt, a German physicist, named and discovered the Avogadro number. Loschmidt realized that a mo ...
... 17.2 Avogadro’s Number The Avogadro number was named in honor of Amedeo Avogadro who discovered that a mole of any gas under the same conditions has the same number of molecules. Johann Josef Loschmidt, a German physicist, named and discovered the Avogadro number. Loschmidt realized that a mo ...
Atom (A) or Ion (I)
... 66. A sample of sulfur has a mass of 223 g. How many moles of sulfur are in the sample? 67. Calculate the mass of 3.89 moles of ethanol (C 2H5OH). 68. Determine the mass of 0.187 mol of tin (II) sulfate (SnSO 4) 69. Calculate the number of atoms in 27.2 g of plutonium. 70. If a student has 9.22 x 10 ...
... 66. A sample of sulfur has a mass of 223 g. How many moles of sulfur are in the sample? 67. Calculate the mass of 3.89 moles of ethanol (C 2H5OH). 68. Determine the mass of 0.187 mol of tin (II) sulfate (SnSO 4) 69. Calculate the number of atoms in 27.2 g of plutonium. 70. If a student has 9.22 x 10 ...
Chem 171 Review - Exam 1
... early experiments and scientists that established our modern understanding of the atom and its structure protons, electrons, neutrons - know the characteristics of these particles including relative masses and electrical charge atomic structure - where do the various subatomic particles reside? rela ...
... early experiments and scientists that established our modern understanding of the atom and its structure protons, electrons, neutrons - know the characteristics of these particles including relative masses and electrical charge atomic structure - where do the various subatomic particles reside? rela ...
chemistry 110 final exam
... FORM A -------------------------------------------------------------------------------------1. The highly exothermic thermite reaction, in which aluminum reduces iron (III) oxide to elemental iron, has been used by railroad repair crews to weld rails together. 2Al(s) + Fe2O3(s) ! 2Fe(s) + Al2O3(s) ...
... FORM A -------------------------------------------------------------------------------------1. The highly exothermic thermite reaction, in which aluminum reduces iron (III) oxide to elemental iron, has been used by railroad repair crews to weld rails together. 2Al(s) + Fe2O3(s) ! 2Fe(s) + Al2O3(s) ...
Slide 1 - Herricks
... 4. Balance the elements one at a time by using coefficients. When no coefficient is written, it is assumed to be 1. Begin by balancing elements that appear only once on each side of the equation. Never balance an equation by changing the subscripts in a chemical formula. Each substance has only one ...
... 4. Balance the elements one at a time by using coefficients. When no coefficient is written, it is assumed to be 1. Begin by balancing elements that appear only once on each side of the equation. Never balance an equation by changing the subscripts in a chemical formula. Each substance has only one ...
File
... A) Atoms are neutral because they contain the same number of protons and electrons B) All atoms of a given element must contain the same number protons, electrons, and neutrons. C) Most of the volume of an atom contains only electrons D) The nucleus is positively charged E) Almost all of the mass of ...
... A) Atoms are neutral because they contain the same number of protons and electrons B) All atoms of a given element must contain the same number protons, electrons, and neutrons. C) Most of the volume of an atom contains only electrons D) The nucleus is positively charged E) Almost all of the mass of ...
Chemistry Name: LeChâtlier`s Principle Date: Chemical Equilibrium
... Total pressure is increased (or volume decreased),toward side with less moles of gas Total pressure is decreased (or volume increased),toward side with more moles of gas Increase the temperature, shift away from heat Decrease the temperature, shift toward heat Endothermic = heat is a reactant (+∆H) ...
... Total pressure is increased (or volume decreased),toward side with less moles of gas Total pressure is decreased (or volume increased),toward side with more moles of gas Increase the temperature, shift away from heat Decrease the temperature, shift toward heat Endothermic = heat is a reactant (+∆H) ...
South Pasadena • AP Chemistry
... 11. When H2SO4 and Ba(OH)2 are reacted in a double replacement reaction, one of the products of the reaction is… a) H2 d) BaH2 b) H2O e) SO2 c) BaS 12. In the double replacement reaction between the weak acid, HC2H3O2 and strong base, NaOH, which ion(s) are spectator ions? a) Na+, C2H3O2– d) H+, C2 ...
... 11. When H2SO4 and Ba(OH)2 are reacted in a double replacement reaction, one of the products of the reaction is… a) H2 d) BaH2 b) H2O e) SO2 c) BaS 12. In the double replacement reaction between the weak acid, HC2H3O2 and strong base, NaOH, which ion(s) are spectator ions? a) Na+, C2H3O2– d) H+, C2 ...
Unit 8 Packet
... 4. When sodium hydroxide reacts with sulfuric acid (H2SO4), water and sodium sulfate are the products. Calculate the mass of sodium sulfate produced when 15.5 g of sodium hydroxide are reacted with 46.7 g of sulfuric acid. [Hint: which unit is used in all stoichiometry reasoning?] ...
... 4. When sodium hydroxide reacts with sulfuric acid (H2SO4), water and sodium sulfate are the products. Calculate the mass of sodium sulfate produced when 15.5 g of sodium hydroxide are reacted with 46.7 g of sulfuric acid. [Hint: which unit is used in all stoichiometry reasoning?] ...
2A Final Exam Review Worksheet
... 11. 100.0 mL of 2.00 M sulfuric acid neutralizes with what mass of potassium hydroxide? 12. A. What are the products to the following reaction that occurs in solution? (NH4)2S + Al(C2H3O2)3 → ? B. Which will ppt? Sr(NO3)2, BaSO4, BaS, AgBr, AuBr 13. What are all the strong acids and strong bases fro ...
... 11. 100.0 mL of 2.00 M sulfuric acid neutralizes with what mass of potassium hydroxide? 12. A. What are the products to the following reaction that occurs in solution? (NH4)2S + Al(C2H3O2)3 → ? B. Which will ppt? Sr(NO3)2, BaSO4, BaS, AgBr, AuBr 13. What are all the strong acids and strong bases fro ...
atom a very small particle that makes up most kinds of matters and
... smells, color, etc. a substance that has the same chemical composition through and cannot be separated into its parts by physical means ...
... smells, color, etc. a substance that has the same chemical composition through and cannot be separated into its parts by physical means ...
Thermodynamics Test Study Guide—AP _____ 1. The entropy
... 80.0oC. The final temperature of the mixture is 75.3oC. Assuming that the specific heat of water is 1.00 cal/g-oC and that no heat is lost to or gained from the surroundings, what is the specific heat of copper, in cal/g-oC? 11. The combustion of 0.100 gram of ethane causes a temperature rise of 2.0 ...
... 80.0oC. The final temperature of the mixture is 75.3oC. Assuming that the specific heat of water is 1.00 cal/g-oC and that no heat is lost to or gained from the surroundings, what is the specific heat of copper, in cal/g-oC? 11. The combustion of 0.100 gram of ethane causes a temperature rise of 2.0 ...
enthalpy worksheet
... If a chemical reaction occurs at constant pressure, as all of our chemical reactions do we can consider a property called enthalpy. Enthalpy (H) is the energy (heat) content of a system at constant pressure. You cannot measure the actual energy or enthalpy of a substance, but you can measure the cha ...
... If a chemical reaction occurs at constant pressure, as all of our chemical reactions do we can consider a property called enthalpy. Enthalpy (H) is the energy (heat) content of a system at constant pressure. You cannot measure the actual energy or enthalpy of a substance, but you can measure the cha ...
Chapter 3 Atomic Mass
... How many moles of sulfuric acid would be needed to produce 4.80 moles of molecular iodine (I2) according to the following balanced equation. 10HI + 2KMnO4 + 3H2SO4 5I2 + 2MnSO4 + K2SO4 + 8H2O ...
... How many moles of sulfuric acid would be needed to produce 4.80 moles of molecular iodine (I2) according to the following balanced equation. 10HI + 2KMnO4 + 3H2SO4 5I2 + 2MnSO4 + K2SO4 + 8H2O ...
Chapter 5 CHEM 121
... BALANCED CHEMICAL EQUATIONS • A balanced chemical equation is one in which the number of atoms of each element in the reactants is equal to the number of atoms of that same element in the products. • A reaction can be balanced by applying the law of conservation of matter. • Coefficients (in red be ...
... BALANCED CHEMICAL EQUATIONS • A balanced chemical equation is one in which the number of atoms of each element in the reactants is equal to the number of atoms of that same element in the products. • A reaction can be balanced by applying the law of conservation of matter. • Coefficients (in red be ...
Review - gbschemphys
... produced if a given amount of moles of reactant was reacted. Which quantities would be essential in order to solve such a problem? Bubble in all that apply - but only those that are essential to this calculation. a. The molar mass of the reactant b. The molar mass of the product c. The coefficients ...
... produced if a given amount of moles of reactant was reacted. Which quantities would be essential in order to solve such a problem? Bubble in all that apply - but only those that are essential to this calculation. a. The molar mass of the reactant b. The molar mass of the product c. The coefficients ...
Fall Exam 4
... The rate of evaporation begins high and decreases; the rate of condensation begins low and increases until it is the same as the rate of evaporation. The rate of condensation starts at zero and increases until it becomes constant and matches the constant rate of evaporation. ...
... The rate of evaporation begins high and decreases; the rate of condensation begins low and increases until it is the same as the rate of evaporation. The rate of condensation starts at zero and increases until it becomes constant and matches the constant rate of evaporation. ...
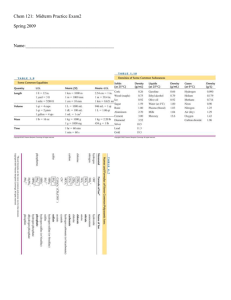
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.