South Pasadena • AP Chemistry
... 11. When H2SO4 and Ba(OH)2 are reacted in a double replacement reaction, one of the products of the reaction is… a) H2 d) BaH2 b) H2O e) SO2 c) BaS 12. In the double replacement reaction between the weak acid, HC2H3O2 and strong base, NaOH, which ion(s) are spectator ions? a) Na+, C2H3O2– d) H+, C2 ...
... 11. When H2SO4 and Ba(OH)2 are reacted in a double replacement reaction, one of the products of the reaction is… a) H2 d) BaH2 b) H2O e) SO2 c) BaS 12. In the double replacement reaction between the weak acid, HC2H3O2 and strong base, NaOH, which ion(s) are spectator ions? a) Na+, C2H3O2– d) H+, C2 ...
Regents Chemistry Review Questions
... Write the chemical reaction that occurs when solutions of sodium chloride and silver nitrate are mixed together. Does a precipitate form? If so, name the precipitate. Write the chemical reaction that occurs when solutions of ammonium sulfide and sodium hydrogen carbonate are mixed together. Does a p ...
... Write the chemical reaction that occurs when solutions of sodium chloride and silver nitrate are mixed together. Does a precipitate form? If so, name the precipitate. Write the chemical reaction that occurs when solutions of ammonium sulfide and sodium hydrogen carbonate are mixed together. Does a p ...
THE MOLE (pp. 159
... 3. But one _______________ read this reaction in terms of mass amounts. 4. If we convert from moles to mass, we can then obtain information regarding the masses of the reactants and products involved: ...
... 3. But one _______________ read this reaction in terms of mass amounts. 4. If we convert from moles to mass, we can then obtain information regarding the masses of the reactants and products involved: ...
Atoms and Molecules
... In the human body, the toxic compound hydrogen cyanide is neutralized by the acid, H 2S2O3, according to the reaction: HCN + H2S2O3 HCNS + H2SO3. If 1.000 mg of H2S2O3, is available in the body, will this be enough to neutralize 2.000 mg of HCN swallowed by a person? [hint – focus on the mole rati ...
... In the human body, the toxic compound hydrogen cyanide is neutralized by the acid, H 2S2O3, according to the reaction: HCN + H2S2O3 HCNS + H2SO3. If 1.000 mg of H2S2O3, is available in the body, will this be enough to neutralize 2.000 mg of HCN swallowed by a person? [hint – focus on the mole rati ...
Example - Schoolwires.net
... Example : How many milliliters of liquid water can be produced by the combustion of 775 mL of octane with oxygen? Assume that the volumes of the octane and the water are measured at 20oC where the densities are 0.7025 g/mL for octane and 0.9982 g/mL for water. C8H18(l) + O2(g) CO2(g) + H2O(l) ...
... Example : How many milliliters of liquid water can be produced by the combustion of 775 mL of octane with oxygen? Assume that the volumes of the octane and the water are measured at 20oC where the densities are 0.7025 g/mL for octane and 0.9982 g/mL for water. C8H18(l) + O2(g) CO2(g) + H2O(l) ...
Energy and Matter
... cooking food, burning wood, decomposing. Chemical reactions are processes where one substance is changed into a new one. A reactant is what goes into a reaction and the product is what comes out. If the product is solid, it is called a precipitate. ...
... cooking food, burning wood, decomposing. Chemical reactions are processes where one substance is changed into a new one. A reactant is what goes into a reaction and the product is what comes out. If the product is solid, it is called a precipitate. ...
Final Exam Practice Problems Set 2
... 2) All atoms of a given element are identical; the atoms of different elements are different and have different properties. 3) Atoms of an element are not changed into different types of atoms by chemical reactions; atoms are neither created nor destroyed in chemical reactions. 4) Compounds are form ...
... 2) All atoms of a given element are identical; the atoms of different elements are different and have different properties. 3) Atoms of an element are not changed into different types of atoms by chemical reactions; atoms are neither created nor destroyed in chemical reactions. 4) Compounds are form ...
Thermal Energy from Chemical Reactions
... Measuring Heat Released During a Reaction (Enthalpy Change) • Measured by a Calorimeter • When a reaction takes place, the heat change causes a rise or fall in temperature in the contents of the calorimeter. • Before use calorimeter must be calibrated • Find out how much energy is needed to change ...
... Measuring Heat Released During a Reaction (Enthalpy Change) • Measured by a Calorimeter • When a reaction takes place, the heat change causes a rise or fall in temperature in the contents of the calorimeter. • Before use calorimeter must be calibrated • Find out how much energy is needed to change ...
Chapter 1 - TamAPChemistryHart
... The Modern View of Atomic Structure and Atomic Weights: Exercises: pp. 71,72: # 17, 20, 23, 25, 29, 31 17. The radius of an atom of krypton (Kr) is about 1.9 Å (10-10 m). a) Express this distance in nanometers (nm) and in picometers (pm). b) How many krypton atoms would have to be lined up to span 1 ...
... The Modern View of Atomic Structure and Atomic Weights: Exercises: pp. 71,72: # 17, 20, 23, 25, 29, 31 17. The radius of an atom of krypton (Kr) is about 1.9 Å (10-10 m). a) Express this distance in nanometers (nm) and in picometers (pm). b) How many krypton atoms would have to be lined up to span 1 ...
Final Exam Chemistry B2A Mr. Kimball`s Class 2003
... 60. When this equation is balanced, what are the coefficients? ___ Mg(ClO3)2 ------> ___ MgCl2 + ___ O2 a) 1,1,1 b) 1,1,3 c) 2,2,3 d) 1,1,6 e) 3,2,2 61.When this equation is balanced, what are the coefficients? ___ P4 + ___ O2 ------> ___ P2O5 a) 1,1,1 b) 5,1,2 c) 1,3,1 d) 2,1,3 e) 1,5,2 62. When th ...
... 60. When this equation is balanced, what are the coefficients? ___ Mg(ClO3)2 ------> ___ MgCl2 + ___ O2 a) 1,1,1 b) 1,1,3 c) 2,2,3 d) 1,1,6 e) 3,2,2 61.When this equation is balanced, what are the coefficients? ___ P4 + ___ O2 ------> ___ P2O5 a) 1,1,1 b) 5,1,2 c) 1,3,1 d) 2,1,3 e) 1,5,2 62. When th ...
Name:…………………………………… Index Number
... Determine the value of a and b in the equation. (2mks) (a) Sulphur atomic number 16 and oxygen atomic number 8 are elements in group 6 of the periodic table. Sulphur is a solid at room temperature while oxygen is a gas at room temperature. Explain why hydrogen sulphide with molecular mass 34 is a ga ...
... Determine the value of a and b in the equation. (2mks) (a) Sulphur atomic number 16 and oxygen atomic number 8 are elements in group 6 of the periodic table. Sulphur is a solid at room temperature while oxygen is a gas at room temperature. Explain why hydrogen sulphide with molecular mass 34 is a ga ...
Stoichiometry …like a beautiful sunset on a serene lake – NOT!
... • CH4 + O2 → CO2 + H2O • In a chemical reaction, atoms have been rearranged but have NOT been created or destroyed. This is why we have to balance every chemical reaction/equation. Java-Balancing! • Balance CH4 + O2 → CO2 + H2O • Balance HCl + NaHCO3 → CO2 + H2O + NaCl • When balancing equations, t ...
... • CH4 + O2 → CO2 + H2O • In a chemical reaction, atoms have been rearranged but have NOT been created or destroyed. This is why we have to balance every chemical reaction/equation. Java-Balancing! • Balance CH4 + O2 → CO2 + H2O • Balance HCl + NaHCO3 → CO2 + H2O + NaCl • When balancing equations, t ...
Chapter 4 (additional powerpoint)
... CHEMICAL EQUATIONS • Chemical equations are a convenient way to represent chemical reactions. Chemical equations are written in terms of reactants and products. • A symbol is written in parentheses to the right of each reactant and product to indicate the state or form in which the substance exists ...
... CHEMICAL EQUATIONS • Chemical equations are a convenient way to represent chemical reactions. Chemical equations are written in terms of reactants and products. • A symbol is written in parentheses to the right of each reactant and product to indicate the state or form in which the substance exists ...
Example - Request a Spot account
... 1. Catalysts are substances that speed up chemical reactions a. Allow reactions to occur that might not otherwise take place (due to low temperature for example) b. Lower activation energy for a chemical reaction 2. Participation of catalysts in a chemical reaction a. They may undergo a chemical cha ...
... 1. Catalysts are substances that speed up chemical reactions a. Allow reactions to occur that might not otherwise take place (due to low temperature for example) b. Lower activation energy for a chemical reaction 2. Participation of catalysts in a chemical reaction a. They may undergo a chemical cha ...
A.P. Chemistry
... Calculations using Titration Data: M1V1 = M2V2 M = mol/L Volume in liters (be sure to convert mL L) (p. 145-147) Problem: What volume of 16 M sulfuric acid must be used to prepare 1.5 L of a 0.10 M H2SO4 solution? ...
... Calculations using Titration Data: M1V1 = M2V2 M = mol/L Volume in liters (be sure to convert mL L) (p. 145-147) Problem: What volume of 16 M sulfuric acid must be used to prepare 1.5 L of a 0.10 M H2SO4 solution? ...
1 Mole
... e.x. sulfate appears on both sides of the reaction so SO4 can be treated like one atom: Mg(s) + CuSO4(aq) MgSO4(aq) + Cu(s) e.x. carbonate breaks apart so atoms must be balanced individually: CaCO3(aq) + HCl(aq) CaCl2(aq) + H2O(l) + CO2(g) ...
... e.x. sulfate appears on both sides of the reaction so SO4 can be treated like one atom: Mg(s) + CuSO4(aq) MgSO4(aq) + Cu(s) e.x. carbonate breaks apart so atoms must be balanced individually: CaCO3(aq) + HCl(aq) CaCl2(aq) + H2O(l) + CO2(g) ...
PDF(343KB)
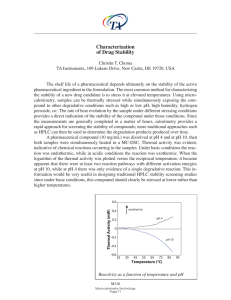
... rapid approach for screening the stability of compounds; more traditional approaches such as HPLC can then be used to determine the degradation products produced over time. A pharmaceutical compound (10 mg/mL) was dissolved at pH 4 and at pH 10, then both samples were simultaneously heated in a MC-D ...
... rapid approach for screening the stability of compounds; more traditional approaches such as HPLC can then be used to determine the degradation products produced over time. A pharmaceutical compound (10 mg/mL) was dissolved at pH 4 and at pH 10, then both samples were simultaneously heated in a MC-D ...
2. NH3 - Huffman Chemistry Website!
... a. What does the number 235 tell you about uranium? _______________________________ b. Write the symbol for this atom using subscripts to show the mass number and atomic number. ...
... a. What does the number 235 tell you about uranium? _______________________________ b. Write the symbol for this atom using subscripts to show the mass number and atomic number. ...
L1 – CHEMISTRY FINAL REVIEW
... capillary action; high specific heat (it takes a lot of energy to raise the temperature of water); only substance in which the solid state is less dense than the liquid state; unusually high BP when compared to other molecular compounds with similar molecular weight. 34. How is a saturation point an ...
... capillary action; high specific heat (it takes a lot of energy to raise the temperature of water); only substance in which the solid state is less dense than the liquid state; unusually high BP when compared to other molecular compounds with similar molecular weight. 34. How is a saturation point an ...
Ch. 9 Review
... 2 multiple choice: coefficients & mole ratio 8 problems moles to moles (2) moles to grams grams to moles grams to grams (2) percent yield with grams to grams percent yield, with grams to grams, find actual yield given percent yield ...
... 2 multiple choice: coefficients & mole ratio 8 problems moles to moles (2) moles to grams grams to moles grams to grams (2) percent yield with grams to grams percent yield, with grams to grams, find actual yield given percent yield ...
Final Review
... c. 22.4 liters d. It depends on the gas since all gases have different densities 5. Which of the following is NOT true at STP conditions? a. Temperature is at 0°C and pressure is at 1 atm b. Temperature is at 273 K and pressure is at 760 mmHg c. The volume of 1 mol of He gas is 22.4 L d. The volume ...
... c. 22.4 liters d. It depends on the gas since all gases have different densities 5. Which of the following is NOT true at STP conditions? a. Temperature is at 0°C and pressure is at 1 atm b. Temperature is at 273 K and pressure is at 760 mmHg c. The volume of 1 mol of He gas is 22.4 L d. The volume ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.