Final Review
... c. 22.4 liters d. It depends on the gas since all gases have different densities 5. Which of the following is NOT true at STP conditions? a. Temperature is at 0°C and pressure is at 1 atm b. Temperature is at 273 K and pressure is at 760 mmHg c. The volume of 1 mol of He gas is 22.4 L d. The volume ...
... c. 22.4 liters d. It depends on the gas since all gases have different densities 5. Which of the following is NOT true at STP conditions? a. Temperature is at 0°C and pressure is at 1 atm b. Temperature is at 273 K and pressure is at 760 mmHg c. The volume of 1 mol of He gas is 22.4 L d. The volume ...
8.5DF: Chemical Formulas and Equations
... reacting with water to produce carbon dioxide gas. This gas produces the “holes” in the cake that give the cake its light, fluffy texture. A similar type of reaction occurs when baking soda is mixed with vinegar. Work with your child to investigate, either online or via textbook, the chemical formul ...
... reacting with water to produce carbon dioxide gas. This gas produces the “holes” in the cake that give the cake its light, fluffy texture. A similar type of reaction occurs when baking soda is mixed with vinegar. Work with your child to investigate, either online or via textbook, the chemical formul ...
Grade 11 Chemistry E.. - hrsbstaff.ednet.ns.ca
... g. Na2SO4(aq) + BaCl2(aq) → BaSO4(s) + NaCl(aq) h. CH3OH(l) + O2(g) → CO2(g) + H2O(g) 25. Classify each of the above according to the 5 types of reactions (composition, decomposition, single replacement, double replacement and combustion). 26. Write the formula for each material correctly and then b ...
... g. Na2SO4(aq) + BaCl2(aq) → BaSO4(s) + NaCl(aq) h. CH3OH(l) + O2(g) → CO2(g) + H2O(g) 25. Classify each of the above according to the 5 types of reactions (composition, decomposition, single replacement, double replacement and combustion). 26. Write the formula for each material correctly and then b ...
Free response review
... 1. A 50.0 gram sample of solid ammonium carbonate is placed in a closed evacuated 3.00L flask and heated to 400° C. It decomposed to produce ammonia, water, and carbon dioxide. The equilibrium constant, Kp, for the reaction is 0.295 at 400° C. a. Write the balanced equation. b. Write the Kp equilibr ...
... 1. A 50.0 gram sample of solid ammonium carbonate is placed in a closed evacuated 3.00L flask and heated to 400° C. It decomposed to produce ammonia, water, and carbon dioxide. The equilibrium constant, Kp, for the reaction is 0.295 at 400° C. a. Write the balanced equation. b. Write the Kp equilibr ...
Molecular and Empirical Formulas
... Atomic weight of an element is the average of the masses of it’s isotopes. – For hydrogen, this value is 1.00794 atomic mass units, for chlorine its 35.453 amu – But since a mole of amu’s is a gram, we can use these exact same numbers for atomic weights in “grams per mole.” Hydrogen weighs 1.00794 g ...
... Atomic weight of an element is the average of the masses of it’s isotopes. – For hydrogen, this value is 1.00794 atomic mass units, for chlorine its 35.453 amu – But since a mole of amu’s is a gram, we can use these exact same numbers for atomic weights in “grams per mole.” Hydrogen weighs 1.00794 g ...
The Advanced Placement Examination in Chemistry Part I – Multiple
... (C) 34 g (D) 85 g (E) 140 g 32. Which of the following oxides is a gas at 25°C and 1 atm ? (A) Rb2O (B) N2O (C) Na2O2 (D) SiO2 (E) La2O3 52. Propane gas, C3H8, burns in excess oxygen gas. When the equation for the reaction is correctly balanced and all coefficients are reduced to their lowest whole- ...
... (C) 34 g (D) 85 g (E) 140 g 32. Which of the following oxides is a gas at 25°C and 1 atm ? (A) Rb2O (B) N2O (C) Na2O2 (D) SiO2 (E) La2O3 52. Propane gas, C3H8, burns in excess oxygen gas. When the equation for the reaction is correctly balanced and all coefficients are reduced to their lowest whole- ...
Regents Review Packet B2 Answer Key
... Three elements, represented by D, E, and Q, are located in Period 3. Some properties of these elements are listed in the table below. A student's experimental result indicates that the density of element Q is , at room temperature and standard pressure. ...
... Three elements, represented by D, E, and Q, are located in Period 3. Some properties of these elements are listed in the table below. A student's experimental result indicates that the density of element Q is , at room temperature and standard pressure. ...
Exam practice answers 5
... white solid and MgBr2 would give a cream solid. If dilute ammonia, NH3, is added the white precipitate dissolves. The cream precipitate will only dissolve in concentrated NH3. There are other ways of doing this such as: Both solutions would conduct electricity. If electricity is passed through t ...
... white solid and MgBr2 would give a cream solid. If dilute ammonia, NH3, is added the white precipitate dissolves. The cream precipitate will only dissolve in concentrated NH3. There are other ways of doing this such as: Both solutions would conduct electricity. If electricity is passed through t ...
Chemistry 102B What`s in an atom? Before “Chemistry” Other Early
... Developed the “Law of conservation of Mass”. • Joseph Proust (early 1800s) – discovered that a given compound always contained the same proportions of certain elements by mass. “Law of Definite Proportions” • John Dalton (early 1800s) – noted that elements that combined to form more than one kind of ...
... Developed the “Law of conservation of Mass”. • Joseph Proust (early 1800s) – discovered that a given compound always contained the same proportions of certain elements by mass. “Law of Definite Proportions” • John Dalton (early 1800s) – noted that elements that combined to form more than one kind of ...
C4C5C6
... • Step 2: Calculate the moles for the part you have the information for. (moles of aluminium = 135/ 27 = 5) • Step 3: Look at the ratio to give you the moles for the part that you want. (4 moles of Al react to form 2 molesAl2O3 so 5 moles would give 2.5 moles of aluminium oxide) • Step 4: Use the eq ...
... • Step 2: Calculate the moles for the part you have the information for. (moles of aluminium = 135/ 27 = 5) • Step 3: Look at the ratio to give you the moles for the part that you want. (4 moles of Al react to form 2 molesAl2O3 so 5 moles would give 2.5 moles of aluminium oxide) • Step 4: Use the eq ...
Stoichiometry: Calculations with Chemical Formulas and Equations
... Avogadro’s Number • In a lab, we cannot work with individual molecules. They are too small. • 6.02 × 1023 atoms or molecules is an amount that brings us to lab size. It is ONE MOLE. • One mole of 12C has a mass of 12.000 g. © 2015 Pearson Education, Inc. ...
... Avogadro’s Number • In a lab, we cannot work with individual molecules. They are too small. • 6.02 × 1023 atoms or molecules is an amount that brings us to lab size. It is ONE MOLE. • One mole of 12C has a mass of 12.000 g. © 2015 Pearson Education, Inc. ...
Form A 1 Chem 130 Name______________________________
... If we look at the reaction, there are three moles of gas on the reactant side and only two moles of gas on the product side, therefore, we expect the S for this process to be negative. So, we will have a situation where we have a negative H and a negative S. Since G = H - TS, the spontaneity o ...
... If we look at the reaction, there are three moles of gas on the reactant side and only two moles of gas on the product side, therefore, we expect the S for this process to be negative. So, we will have a situation where we have a negative H and a negative S. Since G = H - TS, the spontaneity o ...
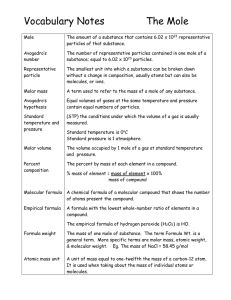
Vocabulary Notes
... without a change in composition, usually atoms but can also be molecules, or ions. ...
... without a change in composition, usually atoms but can also be molecules, or ions. ...
This `practice exam`
... 17. An organic solvent has the empirical formula CH. If the molar mass of this solvent is 78.11 g/mol, what is the molecular formula of benzene? C6H6 18. Ammonia is prepared by reacting nitrogen and hydrogen gases at high temperature according to the unbalanced chemical equation below. __ N2(g) + __ ...
... 17. An organic solvent has the empirical formula CH. If the molar mass of this solvent is 78.11 g/mol, what is the molecular formula of benzene? C6H6 18. Ammonia is prepared by reacting nitrogen and hydrogen gases at high temperature according to the unbalanced chemical equation below. __ N2(g) + __ ...
Thermochemistry 2 Matching Match each item with the correct
... Matching Match each item with the correct statement below. a. heat of reaction d. heat of fusion b. heat of formation e. heat of solution c. Hess's law of heat summation ____ ...
... Matching Match each item with the correct statement below. a. heat of reaction d. heat of fusion b. heat of formation e. heat of solution c. Hess's law of heat summation ____ ...
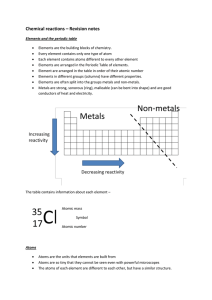
Chemical reactions revision
... oxygen. ‘Oxygen’ does not show up in the name; the ‘ate’ is the only clue it is there You should be able to give the name of the compound formed when different elements combine and tell which elements are present in any simple compound ...
... oxygen. ‘Oxygen’ does not show up in the name; the ‘ate’ is the only clue it is there You should be able to give the name of the compound formed when different elements combine and tell which elements are present in any simple compound ...
Unit 6 – Chemical Reactions: Particles and Energy
... rearrangement process of a chemical reaction requires that all atoms from the reactant molecules MUST become part of one of the products. The conservation of mass we observed at the beginning of the course is evident during chemical reactions; coefficients describe how many whole particles of each ...
... rearrangement process of a chemical reaction requires that all atoms from the reactant molecules MUST become part of one of the products. The conservation of mass we observed at the beginning of the course is evident during chemical reactions; coefficients describe how many whole particles of each ...
Stoichiometry

Stoichiometry /ˌstɔɪkiˈɒmɨtri/ is the calculation of relative quantities of reactants and products in chemical reactions.Stoichiometry is founded on the law of conservation of mass where the total mass of the reactants equals the total mass of the products leading to the insight that the relations among quantities of reactants and products typically form a ratio of positive integers. This means that if the amounts of the separate reactants are known, then the amount of the product can be calculated. Conversely, if one reactant has a known quantity and the quantity of product can be empirically determined, then the amount of the other reactants can also be calculated.As seen in the image to the right, where the balanced equation is:CH4 + 2 O2 → CO2 + 2 H2O.Here, one molecule of methane reacts with two molecules of oxygen gas to yield one molecule of carbon dioxide and two molecules of water. Stoichiometry measures these quantitative relationships, and is used to determine the amount of products/reactants that are produced/needed in a given reaction. Describing the quantitative relationships among substances as they participate in chemical reactions is known as reaction stoichiometry. In the example above, reaction stoichiometry measures the relationship between the methane and oxygen as they react to form carbon dioxide and water.Because of the well known relationship of moles to atomic weights, the ratios that are arrived at by stoichiometry can be used to determine quantities by weight in a reaction described by a balanced equation. This is called composition stoichiometry.Gas stoichiometry deals with reactions involving gases, where the gases are at a known temperature, pressure, and volume and can be assumed to be ideal gases. For gases, the volume ratio is ideally the same by the ideal gas law, but the mass ratio of a single reaction has to be calculated from the molecular masses of the reactants and products. In practice, due to the existence of isotopes, molar masses are used instead when calculating the mass ratio.