Atom is a basic unit of matter that consists of a nucleus
... Describe the modern view of atomic structure ...
... Describe the modern view of atomic structure ...
THE ATOM - Montgomery College
... Tungsten is used to construct the anode and cathode because it has a high atomic number A high atomic number means more energy is required to ionize the material Recall that ionization causes eventual breakdown of material ...
... Tungsten is used to construct the anode and cathode because it has a high atomic number A high atomic number means more energy is required to ionize the material Recall that ionization causes eventual breakdown of material ...
There is a theory which states that if ever for... Universe is for and why it is here it will...
... d) The difference between states of matter is due to __________________________________________ (skip Piza, click arrow at bottom right to move on to next page) II. Atomic Structure a) Atoms are made of 3 different particles (include description): i) ii) iii) Draw a picture of an atom with the thre ...
... d) The difference between states of matter is due to __________________________________________ (skip Piza, click arrow at bottom right to move on to next page) II. Atomic Structure a) Atoms are made of 3 different particles (include description): i) ii) iii) Draw a picture of an atom with the thre ...
ELECTRONIC STRUCTURE OF ATOMS
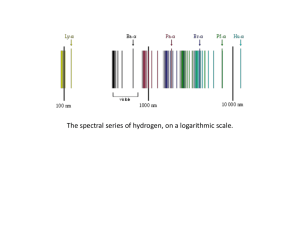
... The ground state is the lowest energy state . An electron in a higher state is said to be in an excited state. Colors from excited gases(street lights) arise because electrons move between energy states. Since energy states are quantized, the light emitted must be quantized and appear as a line ...
... The ground state is the lowest energy state . An electron in a higher state is said to be in an excited state. Colors from excited gases(street lights) arise because electrons move between energy states. Since energy states are quantized, the light emitted must be quantized and appear as a line ...
Historical Introduction to the Elementary Particles
... • 1. Thomson knew That cathode rays emitted by a hot filament could be deflected by a magnet. This suggested that they carried electric charge; • 2. in fact, the direction of the curvature required that the charge be negative. • 3. It seemed, therefore, that these were not rays at all, but rather st ...
... • 1. Thomson knew That cathode rays emitted by a hot filament could be deflected by a magnet. This suggested that they carried electric charge; • 2. in fact, the direction of the curvature required that the charge be negative. • 3. It seemed, therefore, that these were not rays at all, but rather st ...
chapter 7 – cyu
... cathode end. The shadow on the one end (anode) allowed him to see where the electrons were coming from. Crookes also had another experiment using a pinwheel in which electric currents, when switched on, would cause the wheel to spin. This allowed them to conclude that they had mass as well as motion ...
... cathode end. The shadow on the one end (anode) allowed him to see where the electrons were coming from. Crookes also had another experiment using a pinwheel in which electric currents, when switched on, would cause the wheel to spin. This allowed them to conclude that they had mass as well as motion ...
1-7-
... It didn’t explain such things as why elements give off light of specific colors when heated: Example: fireworks, iron ...
... It didn’t explain such things as why elements give off light of specific colors when heated: Example: fireworks, iron ...
Development of the Model of the Atom
... to photon emission. Electrons can only circle the nucleus only in allowed paths or orbits; the further the obit, the higher the energy. His model could explain emission spectrum observed for the hydrogen atom. He calculated the allowed energy levels for the hydrogen atom and related the possible ene ...
... to photon emission. Electrons can only circle the nucleus only in allowed paths or orbits; the further the obit, the higher the energy. His model could explain emission spectrum observed for the hydrogen atom. He calculated the allowed energy levels for the hydrogen atom and related the possible ene ...
Things to Know, Understand and Do
... deBroglie’s X-ray diffraction experiment and how it showed wave-particle duality How the quantum model is similar and different than Bohr’s model Heisenberg’s Uncertainty Principle Understand how an orbital for an electron in an atom corresponds to both the allowed energy of that electron. Understan ...
... deBroglie’s X-ray diffraction experiment and how it showed wave-particle duality How the quantum model is similar and different than Bohr’s model Heisenberg’s Uncertainty Principle Understand how an orbital for an electron in an atom corresponds to both the allowed energy of that electron. Understan ...
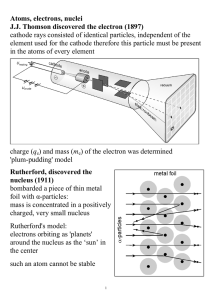
Atoms, electrons, nuclei J.J. Thomson discovered the electron (1897
... de Broglie (1923) described the discrete energy levels of electrons within an atom as results of a wave phenomenon momentum of an electron p = mev = h/p where λ is the wavelength of the matter wave corresponding to the electron, and h is the Planck constant. Davisson and Germer (1927) used electro ...
... de Broglie (1923) described the discrete energy levels of electrons within an atom as results of a wave phenomenon momentum of an electron p = mev = h/p where λ is the wavelength of the matter wave corresponding to the electron, and h is the Planck constant. Davisson and Germer (1927) used electro ...
7.4 The Wave Nature of Matter * 7.5 Quantum Mechanics and the Atom
... Electrons (Particles or Waves?) • Electrons exhibit both particle and wave nature (wave-particle duality). • Heisenberg’s uncertainty principle: we are unable to identify a particles position and velocity at the same time. • Since we can not determine the exact location and velocity of an electron ...
... Electrons (Particles or Waves?) • Electrons exhibit both particle and wave nature (wave-particle duality). • Heisenberg’s uncertainty principle: we are unable to identify a particles position and velocity at the same time. • Since we can not determine the exact location and velocity of an electron ...
Rutherford–Bohr model
... In 1914, James Franck and Gustav Hertz performed an experiment which demonstrated the existence of excited states in mercury atoms, helping to confirm the quantum theory which predicted that electrons occupied only discrete, quantized energy states. Electrons were accelerated by a voltage toward a p ...
... In 1914, James Franck and Gustav Hertz performed an experiment which demonstrated the existence of excited states in mercury atoms, helping to confirm the quantum theory which predicted that electrons occupied only discrete, quantized energy states. Electrons were accelerated by a voltage toward a p ...
Bohr Model and Quantum Model
... Principle Heisenberg stated that you may know the location of an electron or the velocity of electron but you may not know both simultaneously ...
... Principle Heisenberg stated that you may know the location of an electron or the velocity of electron but you may not know both simultaneously ...
Easy explanation
... • Proposed ‘plum-pudding’ model of atom, in which electrons move throughout a solid atom with evenly-distributed positive charge. ...
... • Proposed ‘plum-pudding’ model of atom, in which electrons move throughout a solid atom with evenly-distributed positive charge. ...
Non-KAM dynamical chaos in semiconductor superlattices Arkadii Krokhin, UNT
... I will present our new results concerning electron dynamics in semiconductor superlattices in the presence of non-parallel electric and magnetic field. In this geometry the electrons in the superlattice miniband turn out to form a non-KAM dynamical system that exhibits a non-traditional chaotic beha ...
... I will present our new results concerning electron dynamics in semiconductor superlattices in the presence of non-parallel electric and magnetic field. In this geometry the electrons in the superlattice miniband turn out to form a non-KAM dynamical system that exhibits a non-traditional chaotic beha ...
Schrödinger and Matter Waves
... – e.g., gravitational potential energy, electric potential energy ...
... – e.g., gravitational potential energy, electric potential energy ...
Electron

The electron is a subatomic particle, symbol e− or β−, with a negative elementary electric charge. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. The electron has a mass that is approximately 1/1836 that of the proton. Quantum mechanical properties of the electron include an intrinsic angular momentum (spin) of a half-integer value in units of ħ, which means that it is a fermion. Being fermions, no two electrons can occupy the same quantum state, in accordance with the Pauli exclusion principle. Like all matter, electrons have properties of both particles and waves, and so can collide with other particles and can be diffracted like light. The wave properties of electrons are easier to observe with experiments than those of other particles like neutrons and protons because electrons have a lower mass and hence a higher De Broglie wavelength for typical energies.Many physical phenomena involve electrons in an essential role, such as electricity, magnetism, and thermal conductivity, and they also participate in gravitational, electromagnetic and weak interactions. An electron generates an electric field surrounding it. An electron moving relative to an observer generates a magnetic field. External magnetic fields deflect an electron. Electrons radiate or absorb energy in the form of photons when accelerated. Laboratory instruments are capable of containing and observing individual electrons as well as electron plasma using electromagnetic fields, whereas dedicated telescopes can detect electron plasma in outer space. Electrons have many applications, including electronics, welding, cathode ray tubes, electron microscopes, radiation therapy, lasers, gaseous ionization detectors and particle accelerators.Interactions involving electrons and other subatomic particles are of interest in fields such as chemistry and nuclear physics. The Coulomb force interaction between positive protons inside atomic nuclei and negative electrons composes atoms. Ionization or changes in the proportions of particles changes the binding energy of the system. The exchange or sharing of the electrons between two or more atoms is the main cause of chemical bonding. British natural philosopher Richard Laming first hypothesized the concept of an indivisible quantity of electric charge to explain the chemical properties of atoms in 1838; Irish physicist George Johnstone Stoney named this charge 'electron' in 1891, and J. J. Thomson and his team of British physicists identified it as a particle in 1897. Electrons can also participate in nuclear reactions, such as nucleosynthesis in stars, where they are known as beta particles. Electrons may be created through beta decay of radioactive isotopes and in high-energy collisions, for instance when cosmic rays enter the atmosphere. The antiparticle of the electron is called the positron; it is identical to the electron except that it carries electrical and other charges of the opposite sign. When an electron collides with a positron, both particles may be totally annihilated, producing gamma ray photons.