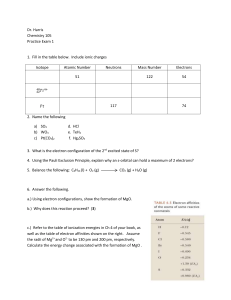
Dr. Harris Chemistry 105 Practice Exam 1 Isotope Atomic Number
... 13. A sphere of gold has a radius of 1.5 inches. The volume of a sphere is 4.19r3. Given that the mass of this sphere is 4469 g, calculate the density of gold in g/cm3 to the correct number of significant figures. V = 4.19(1.5 in)3 =113.13 in3 x (2.54 cm/ in)3 = 231.73 cm3 ρ = 4469 g/ 231.73 cm3 = 1 ...
... 13. A sphere of gold has a radius of 1.5 inches. The volume of a sphere is 4.19r3. Given that the mass of this sphere is 4469 g, calculate the density of gold in g/cm3 to the correct number of significant figures. V = 4.19(1.5 in)3 =113.13 in3 x (2.54 cm/ in)3 = 231.73 cm3 ρ = 4469 g/ 231.73 cm3 = 1 ...
Chem 101A Exam 4 Concepts Chapter 7 – Modern Atomic Theory
... Chem 101A Exam 4 Concepts Chapter 7 – Modern Atomic Theory Use formulas that relate energy of photon, frequency, wavelength, speed of light, and the Rydberg Equation Notable scientists and their contributions: Rutherford, Bohr, Planc, de Broglie, Heisenberg, Schrödinger. The four Quantum ...
... Chem 101A Exam 4 Concepts Chapter 7 – Modern Atomic Theory Use formulas that relate energy of photon, frequency, wavelength, speed of light, and the Rydberg Equation Notable scientists and their contributions: Rutherford, Bohr, Planc, de Broglie, Heisenberg, Schrödinger. The four Quantum ...
MIDTERM REVIEW GAME 16-17
... 5) The product of the frequency and the wavelength is the: 1. Speed of the wave 2. Number of waves passing a point per second 3. Distance between waves 4. Time for a wave to ...
... 5) The product of the frequency and the wavelength is the: 1. Speed of the wave 2. Number of waves passing a point per second 3. Distance between waves 4. Time for a wave to ...
PPT - Lawless Teaching : Home
... This is also the smallest amount of charge in nature and all other charges are integer multiples of this (we will later see quarks as the exception to this). Named by an Irish guy, George J. Stoney (cause that’s important). ...
... This is also the smallest amount of charge in nature and all other charges are integer multiples of this (we will later see quarks as the exception to this). Named by an Irish guy, George J. Stoney (cause that’s important). ...
MiniQuiz 3
... One of the major factors in determining the energy of an electron is its electrostatic attraction to the positive nucleus. Shielding refers to the: a) the number of electrons in the outer shell. b) the electron’s angular momentum quantum number. c•) the presence of other electrons between the electr ...
... One of the major factors in determining the energy of an electron is its electrostatic attraction to the positive nucleus. Shielding refers to the: a) the number of electrons in the outer shell. b) the electron’s angular momentum quantum number. c•) the presence of other electrons between the electr ...
Quantum Theory and Electrons as Waves
... If light could have particle-like behavior, then could matter have wave-like behavior? ...
... If light could have particle-like behavior, then could matter have wave-like behavior? ...
Exam 2-1
... Einstein’s explanation of the photoelectric effect. Rutherford’s experiment with a beam of α particles passing through gold foil. Boer’s model of the atom. Faraday’s experiment of the electroplating of metals. Binnig and Rohrer’s demonstration of the scanning tunneling microscope. ...
... Einstein’s explanation of the photoelectric effect. Rutherford’s experiment with a beam of α particles passing through gold foil. Boer’s model of the atom. Faraday’s experiment of the electroplating of metals. Binnig and Rohrer’s demonstration of the scanning tunneling microscope. ...
Physics 105 - Multiple Choice Questions Ch 16
... move an electron 1 meter C) the energy gained by an electron in moving through a potential difference of 1 volt D) the energy needed to move an electron through 1 meter in any electric field E) the work done when 1 coulomb of charge is moved through a potential difference of 1 volt ...
... move an electron 1 meter C) the energy gained by an electron in moving through a potential difference of 1 volt D) the energy needed to move an electron through 1 meter in any electric field E) the work done when 1 coulomb of charge is moved through a potential difference of 1 volt ...
Dr. Harris Chemistry 105 Practice Exam 1 Isotope Atomic Number
... 14. A laser emits 200mJ of energy per hour. Given that the wavelength of the photons in the beam is 300 nm, and assuming that the emission rate is constant, how many photons are emitted per minute? ...
... 14. A laser emits 200mJ of energy per hour. Given that the wavelength of the photons in the beam is 300 nm, and assuming that the emission rate is constant, how many photons are emitted per minute? ...
Prentice Hall Chemistry Worksheets
... 5. the amount of energy required to move an electron from its present energy level to the next higher one ...
... 5. the amount of energy required to move an electron from its present energy level to the next higher one ...
Introduction to Chemistry
... Learning Objectives (from Zumdahl Resource Guide): (3-4 days lecture/discussion) To characterize electromagnetic radiation in terms of wavelength, frequency, and speed. To introduce the concept of quantized energy. To show that light has both wave and particulate properties. To describe how diffract ...
... Learning Objectives (from Zumdahl Resource Guide): (3-4 days lecture/discussion) To characterize electromagnetic radiation in terms of wavelength, frequency, and speed. To introduce the concept of quantized energy. To show that light has both wave and particulate properties. To describe how diffract ...
AP Chemistry Chapter 6 Outline for Concepts to Know 6.1 Wave
... order of categories of electromagnetic radiation order and approximate range of visible radiation 4 – 8(x10-7) m 6.2 Quantized Energy and Photons Concept of smallest unit of light energy as photon – having properties of both particles and waves Quantum as smallest possible packets or quantit ...
... order of categories of electromagnetic radiation order and approximate range of visible radiation 4 – 8(x10-7) m 6.2 Quantized Energy and Photons Concept of smallest unit of light energy as photon – having properties of both particles and waves Quantum as smallest possible packets or quantit ...
Ch.5 VocabReview
... Choose the term from the following list that best matches each description. quantum photons hertz Pauli exclusion principle wavelength ...
... Choose the term from the following list that best matches each description. quantum photons hertz Pauli exclusion principle wavelength ...
The Electron - Student Moodle
... Because an electron has mass (though it’s very small—about 1/1836 of the mass of a proton or neutron), this means electrons are particles, and all of the equations that apply to motion of solid particles also apply to electrons. However, an electromagnetic wave is a wave of electricity, and electric ...
... Because an electron has mass (though it’s very small—about 1/1836 of the mass of a proton or neutron), this means electrons are particles, and all of the equations that apply to motion of solid particles also apply to electrons. However, an electromagnetic wave is a wave of electricity, and electric ...
2·QUIZLET VOCABULARY: Quantum Numbers Study online at
... 5. Magnetic (orbital) quantum Number: ml Indicates orientation of orbital in space S- 1 orbital P- 3 orbitals D- 5 orbitals F- 7 orbitals 6. orbital: A 3-D space around the nucleus where an electron is likely (high probability) to be found. 7. pauli exclusion principle: An atomic orbital may describ ...
... 5. Magnetic (orbital) quantum Number: ml Indicates orientation of orbital in space S- 1 orbital P- 3 orbitals D- 5 orbitals F- 7 orbitals 6. orbital: A 3-D space around the nucleus where an electron is likely (high probability) to be found. 7. pauli exclusion principle: An atomic orbital may describ ...
Electron

The electron is a subatomic particle, symbol e− or β−, with a negative elementary electric charge. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. The electron has a mass that is approximately 1/1836 that of the proton. Quantum mechanical properties of the electron include an intrinsic angular momentum (spin) of a half-integer value in units of ħ, which means that it is a fermion. Being fermions, no two electrons can occupy the same quantum state, in accordance with the Pauli exclusion principle. Like all matter, electrons have properties of both particles and waves, and so can collide with other particles and can be diffracted like light. The wave properties of electrons are easier to observe with experiments than those of other particles like neutrons and protons because electrons have a lower mass and hence a higher De Broglie wavelength for typical energies.Many physical phenomena involve electrons in an essential role, such as electricity, magnetism, and thermal conductivity, and they also participate in gravitational, electromagnetic and weak interactions. An electron generates an electric field surrounding it. An electron moving relative to an observer generates a magnetic field. External magnetic fields deflect an electron. Electrons radiate or absorb energy in the form of photons when accelerated. Laboratory instruments are capable of containing and observing individual electrons as well as electron plasma using electromagnetic fields, whereas dedicated telescopes can detect electron plasma in outer space. Electrons have many applications, including electronics, welding, cathode ray tubes, electron microscopes, radiation therapy, lasers, gaseous ionization detectors and particle accelerators.Interactions involving electrons and other subatomic particles are of interest in fields such as chemistry and nuclear physics. The Coulomb force interaction between positive protons inside atomic nuclei and negative electrons composes atoms. Ionization or changes in the proportions of particles changes the binding energy of the system. The exchange or sharing of the electrons between two or more atoms is the main cause of chemical bonding. British natural philosopher Richard Laming first hypothesized the concept of an indivisible quantity of electric charge to explain the chemical properties of atoms in 1838; Irish physicist George Johnstone Stoney named this charge 'electron' in 1891, and J. J. Thomson and his team of British physicists identified it as a particle in 1897. Electrons can also participate in nuclear reactions, such as nucleosynthesis in stars, where they are known as beta particles. Electrons may be created through beta decay of radioactive isotopes and in high-energy collisions, for instance when cosmic rays enter the atmosphere. The antiparticle of the electron is called the positron; it is identical to the electron except that it carries electrical and other charges of the opposite sign. When an electron collides with a positron, both particles may be totally annihilated, producing gamma ray photons.