Chapter 2 Study Guide
... 3. The atomic number of an element whose atoms have 12 protons and 11 neutrons is _____. 4. The mass number of an element whose atoms have 15 protons and 17 neutrons is _________. 5. One isotope of carbon has 6 protons and 6 neutrons. The number of protons and neutrons of a second isotope of carbon ...
... 3. The atomic number of an element whose atoms have 12 protons and 11 neutrons is _____. 4. The mass number of an element whose atoms have 15 protons and 17 neutrons is _________. 5. One isotope of carbon has 6 protons and 6 neutrons. The number of protons and neutrons of a second isotope of carbon ...
Chemistry 1 Concept 5 “Electrons in Atoms” Study Guide
... 18. The spin quantum number indicates that the number of possible spin states for an electron in an orbital is __________ 19. The angular momentum quantum number indicates the ________________________ 20. What is the energy of a photon whose frequency is 5.0 x 1020 Hz? ______________ 21. What state ...
... 18. The spin quantum number indicates that the number of possible spin states for an electron in an orbital is __________ 19. The angular momentum quantum number indicates the ________________________ 20. What is the energy of a photon whose frequency is 5.0 x 1020 Hz? ______________ 21. What state ...
Quantum Mechanical Model of the Atom and Electronic Structure 1
... When electrons one, energy is This change in of the emitted ...
... When electrons one, energy is This change in of the emitted ...
Chapter 2 Learning Objectives
... Electromagnetic radiation The photoelectric effect Atomic orbitals and quantum numbers Atomic line spectra Multielectron atoms and electron configurations ...
... Electromagnetic radiation The photoelectric effect Atomic orbitals and quantum numbers Atomic line spectra Multielectron atoms and electron configurations ...
WAVE MECHANICS AND QUANTUM NUMBERS
... A. Electrons as Waves 1. Louis de Broglie 1924- electrons are considered waves confined to the space around a nucleus. 2. supported by the facts that electrons undergo diffraction and interference 3. Werner Heisenberg 1927- Heisenberg Uncertainty Principle: it is impossible to simultaneously identif ...
... A. Electrons as Waves 1. Louis de Broglie 1924- electrons are considered waves confined to the space around a nucleus. 2. supported by the facts that electrons undergo diffraction and interference 3. Werner Heisenberg 1927- Heisenberg Uncertainty Principle: it is impossible to simultaneously identif ...
Aufbau Diagram Directions
... AUFBAU DIAGRAM/ ORBITAL NOTATIONDIRECTIONS Why do we use the Aufbau Diagram? To figure out the electron configuration In doing so, we demonstrate the 3 main principles: Aufbau: Electrons enter orbitals of lowest energy first Pauli Exclusion: an atomic orbital may describe at most 2 electrons (each e ...
... AUFBAU DIAGRAM/ ORBITAL NOTATIONDIRECTIONS Why do we use the Aufbau Diagram? To figure out the electron configuration In doing so, we demonstrate the 3 main principles: Aufbau: Electrons enter orbitals of lowest energy first Pauli Exclusion: an atomic orbital may describe at most 2 electrons (each e ...
Exam sample
... 7. “No two electrons in the same atom may have the same values for all four quantum numbers” is a statement of: a. Hund’s Rule. b. deBroglie’s Hypothesis. c. the Pauli Exclusion Principle. d. the Heisenberg Uncertainty Principle. 8. All s orbitals are: a. shaped like four-leaf clovers. b. dumbbell- ...
... 7. “No two electrons in the same atom may have the same values for all four quantum numbers” is a statement of: a. Hund’s Rule. b. deBroglie’s Hypothesis. c. the Pauli Exclusion Principle. d. the Heisenberg Uncertainty Principle. 8. All s orbitals are: a. shaped like four-leaf clovers. b. dumbbell- ...
Development of Quantum Mechanics Waves
... If light is a wave with particle properties, are electrons particles with wave properties? C. J. Davisson and L. H. Germier at Bell labs proved that electrons produce diffraction patterns and verified deBroglies hypothesis ...
... If light is a wave with particle properties, are electrons particles with wave properties? C. J. Davisson and L. H. Germier at Bell labs proved that electrons produce diffraction patterns and verified deBroglies hypothesis ...
Electron configuration Jeopardy
... Continuous spectrum is like the rainbow. You could look out the window to see that. Line spectrum is certain colors show up in lines. You could pass electricity through an element for that. Who said that? 100 – He came up with the idea of quantum in 1900. Max Planck 200 – Electrons will make the max ...
... Continuous spectrum is like the rainbow. You could look out the window to see that. Line spectrum is certain colors show up in lines. You could pass electricity through an element for that. Who said that? 100 – He came up with the idea of quantum in 1900. Max Planck 200 – Electrons will make the max ...
Homework for the electron microscopy class
... Planck’s constant and c is the speed of light (in the medium). For electrons, the equivalent expression is = h/p where p is the electron momentum: p = mv. In classical mechanics the energy of a particle is given by E = ½ mv2 where m is its mass and v is its velocity. 1. The wavelength of visible l ...
... Planck’s constant and c is the speed of light (in the medium). For electrons, the equivalent expression is = h/p where p is the electron momentum: p = mv. In classical mechanics the energy of a particle is given by E = ½ mv2 where m is its mass and v is its velocity. 1. The wavelength of visible l ...
Ch. 13 Worksheet blank
... 1. Which orbital is the first to be filled in any atom? __________ 2. How many electrons can each orbital hold? s = _______, p = _______, d = ______, f = ______ 3. The third energy level (n=3) can hold a maximum of _______ electrons and is divided into a total of _______ sublevels. 4. According to B ...
... 1. Which orbital is the first to be filled in any atom? __________ 2. How many electrons can each orbital hold? s = _______, p = _______, d = ______, f = ______ 3. The third energy level (n=3) can hold a maximum of _______ electrons and is divided into a total of _______ sublevels. 4. According to B ...
107 chem Assement Q
... c. fundamental state. d. original state. 5. The hydrogen emission spectrum includes light with a wavelength of 434 nanometers. This is caused by an electron moving from: a. the n = 3 state to the n = 2 state. b. the n = 4 state to the n = 2 state. c. the n = 5 state to the n = 2 state. d. the n = 6 ...
... c. fundamental state. d. original state. 5. The hydrogen emission spectrum includes light with a wavelength of 434 nanometers. This is caused by an electron moving from: a. the n = 3 state to the n = 2 state. b. the n = 4 state to the n = 2 state. c. the n = 5 state to the n = 2 state. d. the n = 6 ...
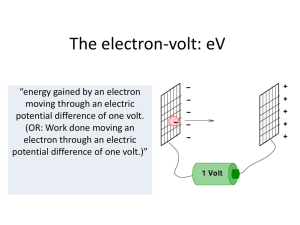
The electron-volt - Hockerill Students
... The electron-volt: eV “energy gained by an electron moving through an electric potential difference of one volt. (OR: Work done moving an electron through an electric potential difference of one volt.)” ...
... The electron-volt: eV “energy gained by an electron moving through an electric potential difference of one volt. (OR: Work done moving an electron through an electric potential difference of one volt.)” ...
introduction [Kompatibilitätsmodus]
... Millikan‘s experiment (1911, Nobel Price in 1923): Oil droplets are sprayed in a chamber between two electrodes and charged by ionizing radiation. Their motion is observed by a microscope. ...
... Millikan‘s experiment (1911, Nobel Price in 1923): Oil droplets are sprayed in a chamber between two electrodes and charged by ionizing radiation. Their motion is observed by a microscope. ...
The Shroedinger/Modern Model of the Atom
... photon – quantum of energy that behaves in some ways like a particle ...
... photon – quantum of energy that behaves in some ways like a particle ...
Slide 1
... What would the wavelength of an electron be (9.11 x 10-28 grams) whizzing about it’s orbit in the Bohr model traveling at a speed of 2.0 million meters per second? 3.6 x 10-10 m ...
... What would the wavelength of an electron be (9.11 x 10-28 grams) whizzing about it’s orbit in the Bohr model traveling at a speed of 2.0 million meters per second? 3.6 x 10-10 m ...
Electron

The electron is a subatomic particle, symbol e− or β−, with a negative elementary electric charge. Electrons belong to the first generation of the lepton particle family, and are generally thought to be elementary particles because they have no known components or substructure. The electron has a mass that is approximately 1/1836 that of the proton. Quantum mechanical properties of the electron include an intrinsic angular momentum (spin) of a half-integer value in units of ħ, which means that it is a fermion. Being fermions, no two electrons can occupy the same quantum state, in accordance with the Pauli exclusion principle. Like all matter, electrons have properties of both particles and waves, and so can collide with other particles and can be diffracted like light. The wave properties of electrons are easier to observe with experiments than those of other particles like neutrons and protons because electrons have a lower mass and hence a higher De Broglie wavelength for typical energies.Many physical phenomena involve electrons in an essential role, such as electricity, magnetism, and thermal conductivity, and they also participate in gravitational, electromagnetic and weak interactions. An electron generates an electric field surrounding it. An electron moving relative to an observer generates a magnetic field. External magnetic fields deflect an electron. Electrons radiate or absorb energy in the form of photons when accelerated. Laboratory instruments are capable of containing and observing individual electrons as well as electron plasma using electromagnetic fields, whereas dedicated telescopes can detect electron plasma in outer space. Electrons have many applications, including electronics, welding, cathode ray tubes, electron microscopes, radiation therapy, lasers, gaseous ionization detectors and particle accelerators.Interactions involving electrons and other subatomic particles are of interest in fields such as chemistry and nuclear physics. The Coulomb force interaction between positive protons inside atomic nuclei and negative electrons composes atoms. Ionization or changes in the proportions of particles changes the binding energy of the system. The exchange or sharing of the electrons between two or more atoms is the main cause of chemical bonding. British natural philosopher Richard Laming first hypothesized the concept of an indivisible quantity of electric charge to explain the chemical properties of atoms in 1838; Irish physicist George Johnstone Stoney named this charge 'electron' in 1891, and J. J. Thomson and his team of British physicists identified it as a particle in 1897. Electrons can also participate in nuclear reactions, such as nucleosynthesis in stars, where they are known as beta particles. Electrons may be created through beta decay of radioactive isotopes and in high-energy collisions, for instance when cosmic rays enter the atmosphere. The antiparticle of the electron is called the positron; it is identical to the electron except that it carries electrical and other charges of the opposite sign. When an electron collides with a positron, both particles may be totally annihilated, producing gamma ray photons.














![introduction [Kompatibilitätsmodus]](http://s1.studyres.com/store/data/017596641_1-03cad833ad630350a78c42d7d7aa10e3-300x300.png)