Modelling glycolysis with Cellware
... in which models are constructed to cover whole biological networks is known as systems biology. The term “glycolysis” is derived form Greek words glyk meaning sweet and lysis for dissolution. In consistent with this logic, glycolysis is the sequence of reactions that metabolizes one molecule of gluc ...
... in which models are constructed to cover whole biological networks is known as systems biology. The term “glycolysis” is derived form Greek words glyk meaning sweet and lysis for dissolution. In consistent with this logic, glycolysis is the sequence of reactions that metabolizes one molecule of gluc ...
Discovery of Enzymes
... Inhibitors Enzyme inhibitors are substances which alter the catalytic action of the enzyme and consequently slow down or stop catalysis. There are three common types of enzyme inhibition 1. Competitive inhibitors 2. Non-competitive inhibitors ...
... Inhibitors Enzyme inhibitors are substances which alter the catalytic action of the enzyme and consequently slow down or stop catalysis. There are three common types of enzyme inhibition 1. Competitive inhibitors 2. Non-competitive inhibitors ...
The Cardiovascular System and Exercise
... substantial energy reservoir. Fat is less accessible for cellular metabolism as it must first be reduced from its complex form, triglyceride, to the simpler components of glycerol and free fatty acids. So although fat acts as a vast stockpile of fuel, energy release is too slow for very intense acti ...
... substantial energy reservoir. Fat is less accessible for cellular metabolism as it must first be reduced from its complex form, triglyceride, to the simpler components of glycerol and free fatty acids. So although fat acts as a vast stockpile of fuel, energy release is too slow for very intense acti ...
Biochem-EnzymesL
... Inhibitors Enzyme inhibitors are substances which alter the catalytic action of the enzyme and consequently slow down or stop catalysis. There are three common types of enzyme inhibition 1. Competitive inhibitors 2. Non-competitive inhibitors ...
... Inhibitors Enzyme inhibitors are substances which alter the catalytic action of the enzyme and consequently slow down or stop catalysis. There are three common types of enzyme inhibition 1. Competitive inhibitors 2. Non-competitive inhibitors ...
CHAPTER 2 ATOMS, MOLECULES, AND IONS Questions
... neutrons, and protons are the key smaller parts of an atom. b. All atoms of hydrogen have 1 proton in the nucleus. Different isotopes of hydrogen have 0, 1, or 2 neutrons in the nucleus. Because we are talking about atoms, this implies a neutral charge, which dictates 1 electron present for all hydr ...
... neutrons, and protons are the key smaller parts of an atom. b. All atoms of hydrogen have 1 proton in the nucleus. Different isotopes of hydrogen have 0, 1, or 2 neutrons in the nucleus. Because we are talking about atoms, this implies a neutral charge, which dictates 1 electron present for all hydr ...
CHAPTER 2 ATOMS, MOLECULES, AND IONS Questions
... neutrons, and protons are the key smaller parts of an atom. b. All atoms of hydrogen have 1 proton in the nucleus. Different isotopes of hydrogen have 0, 1, or 2 neutrons in the nucleus. Because we are talking about atoms, this implies a neutral charge, which dictates 1 electron present for all hydr ...
... neutrons, and protons are the key smaller parts of an atom. b. All atoms of hydrogen have 1 proton in the nucleus. Different isotopes of hydrogen have 0, 1, or 2 neutrons in the nucleus. Because we are talking about atoms, this implies a neutral charge, which dictates 1 electron present for all hydr ...
answer key - chem.uwec.edu
... • Describe the cofactor used in the aconitase reaction. • Compare the a-ketoglutarate dehydrogenase reactions with the pyruvate dehydrogenase reaction. • Describe the strategy used to produce GTP in the succinyl-CoA synthetase reaction. (Question 6) • Describe the overall stoichiometry for the citri ...
... • Describe the cofactor used in the aconitase reaction. • Compare the a-ketoglutarate dehydrogenase reactions with the pyruvate dehydrogenase reaction. • Describe the strategy used to produce GTP in the succinyl-CoA synthetase reaction. (Question 6) • Describe the overall stoichiometry for the citri ...
Chem*3560 Lecture 22: Fatty acid desaturation Relationship of
... Fatty acid desaturase is a multienzyme complex containing the following components: The 9-desaturase is the substrate specific component that accepts a saturated 16 or 18 carbon acyl-CoA, and removes 2 H atoms to create a cis-double bond between C-9 and C-10 (i.e. bond 9 in the chain). The H atoms f ...
... Fatty acid desaturase is a multienzyme complex containing the following components: The 9-desaturase is the substrate specific component that accepts a saturated 16 or 18 carbon acyl-CoA, and removes 2 H atoms to create a cis-double bond between C-9 and C-10 (i.e. bond 9 in the chain). The H atoms f ...
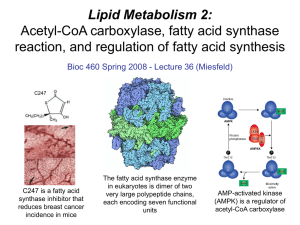
Lecture 36 - Lipid Metabolism 2
... priming group only in the first cycle, after that, only malonyl-CoA is added to the ACP carrier protein each time. There are four reaction steps required each cycle to result in the net addition two carbons to the growing fatty acid chain. ...
... priming group only in the first cycle, after that, only malonyl-CoA is added to the ACP carrier protein each time. There are four reaction steps required each cycle to result in the net addition two carbons to the growing fatty acid chain. ...
Nitrogen Metabolism, Ammonia Degradation and Urea Formation
... Terrestrial vertebrates synthesize urea (excreted by the kidneys) - ureotelic organisms Birds, reptiles synthesize uric acid Urea formation takes place in the liver ...
... Terrestrial vertebrates synthesize urea (excreted by the kidneys) - ureotelic organisms Birds, reptiles synthesize uric acid Urea formation takes place in the liver ...
Calvin Cycle
... Glyceraldehyde-3-P Dehydrogenase catalyzes reduction of the carboxyl of 1,3-bisphosphoglycerate to an aldehyde, with release of Pi, yielding glyceraldehyde-3-P. This is like the Glycolysis enzyme running backward, but the chloroplast Glyceraldehyde-3-P Dehydrogenase uses NADPH as e donor, while the ...
... Glyceraldehyde-3-P Dehydrogenase catalyzes reduction of the carboxyl of 1,3-bisphosphoglycerate to an aldehyde, with release of Pi, yielding glyceraldehyde-3-P. This is like the Glycolysis enzyme running backward, but the chloroplast Glyceraldehyde-3-P Dehydrogenase uses NADPH as e donor, while the ...
Export To Word
... glycolysis undergoes oxidation to produce Acetyl CoA. The video then explains what occurs when Acetyl CoA enters the Kreb's cycle and how NADH and FADH2 are produced. This Khan Academy video explains how the NADH And FADH2 that were made during glycolysis and the Kreb's Cycle are used to generate AT ...
... glycolysis undergoes oxidation to produce Acetyl CoA. The video then explains what occurs when Acetyl CoA enters the Kreb's cycle and how NADH and FADH2 are produced. This Khan Academy video explains how the NADH And FADH2 that were made during glycolysis and the Kreb's Cycle are used to generate AT ...
biology_knowledge_survey.v2 - the Biology Scholars Program
... 133. At the end of the citric acid cycle, what has happened to the carbon atoms from the original glucose molecule? 134. At the end of the citric acid cycle, what has happened to the energy in the glucose molecule? 135. How is the energy in NADH released, and why is it harvested in steps, rather tha ...
... 133. At the end of the citric acid cycle, what has happened to the carbon atoms from the original glucose molecule? 134. At the end of the citric acid cycle, what has happened to the energy in the glucose molecule? 135. How is the energy in NADH released, and why is it harvested in steps, rather tha ...
Engineering Tyrosine-Based Electron Flow Pathways in Proteins
... radical location on the basis of EPR hyperfine structure. Mutation to tyrosine decreases the lipid peroxidase activity of this myoglobin in the presence of low concentrations of reductant, and the possibility of decreasing the intrinsic toxicity of hemoglobin by introduction of these pathways is disc ...
... radical location on the basis of EPR hyperfine structure. Mutation to tyrosine decreases the lipid peroxidase activity of this myoglobin in the presence of low concentrations of reductant, and the possibility of decreasing the intrinsic toxicity of hemoglobin by introduction of these pathways is disc ...
Chapter 5: chemical reactions in the living cell
... and algae, and ultimately converted into other types of energy, most prominently into the formation of new covalent bonds of sugar molecules during photosynthesis (see Chapter 7) - the importance of sugar molecules produced by agricultural plants for human nutrition and food supply proves the prime ...
... and algae, and ultimately converted into other types of energy, most prominently into the formation of new covalent bonds of sugar molecules during photosynthesis (see Chapter 7) - the importance of sugar molecules produced by agricultural plants for human nutrition and food supply proves the prime ...
Krebs Cycle
... ultimately cycles to itself in a series of reactions—this is in contrast to a pathway in which a substrate undergoes conversion to a chemically-distinct product! - Krebs cycle is comprised of a total of eight enzymatic steps—excluding Step 0 for the synthesis of acetyl-CoA—and occurs within the mito ...
... ultimately cycles to itself in a series of reactions—this is in contrast to a pathway in which a substrate undergoes conversion to a chemically-distinct product! - Krebs cycle is comprised of a total of eight enzymatic steps—excluding Step 0 for the synthesis of acetyl-CoA—and occurs within the mito ...
Digestive Enzymes - Emerson Ecologics
... broad spectrum of digestive enzymes to provide activity for overall digestive u support It does this by first providing the fiber digesting enzymes that the human body does not produce, with ProCerelase®, an exclusive combination of hemicellulose, beta-glucanase, cellulase, and phytase. This proprie ...
... broad spectrum of digestive enzymes to provide activity for overall digestive u support It does this by first providing the fiber digesting enzymes that the human body does not produce, with ProCerelase®, an exclusive combination of hemicellulose, beta-glucanase, cellulase, and phytase. This proprie ...
Cells and Energy
... your body. And, when fats are broken down, they yield the most ATP. For example, a typical triglyceride can be broken down to make about 146 molecules of ATP. Proteins store about the same amount of energy as carbohydrates, but they are less likely to be broken down to make ATP. The amino acids that ...
... your body. And, when fats are broken down, they yield the most ATP. For example, a typical triglyceride can be broken down to make about 146 molecules of ATP. Proteins store about the same amount of energy as carbohydrates, but they are less likely to be broken down to make ATP. The amino acids that ...
Concepts of Biology
... quantified and expressed? How can the energy released from one reaction be compared to that of another reaction? A measurement of free energy is used to quantify these energy transfers. Recall that according to the second law of thermodynamics, all energy transfers involve the loss of some amount of ...
... quantified and expressed? How can the energy released from one reaction be compared to that of another reaction? A measurement of free energy is used to quantify these energy transfers. Recall that according to the second law of thermodynamics, all energy transfers involve the loss of some amount of ...
4|HOW CELLS OBTAIN ENERGY
... quantified and expressed? How can the energy released from one reaction be compared to that of another reaction? A measurement of free energy is used to quantify these energy transfers. Recall that according to the second law of thermodynamics, all energy transfers involve the loss of some amount of ...
... quantified and expressed? How can the energy released from one reaction be compared to that of another reaction? A measurement of free energy is used to quantify these energy transfers. Recall that according to the second law of thermodynamics, all energy transfers involve the loss of some amount of ...
Figure 4.5 - Amazon S3
... quantified and expressed? How can the energy released from one reaction be compared to that of another reaction? A measurement of free energy is used to quantify these energy transfers. Recall that according to the second law of thermodynamics, all energy transfers involve the loss of some amount of ...
... quantified and expressed? How can the energy released from one reaction be compared to that of another reaction? A measurement of free energy is used to quantify these energy transfers. Recall that according to the second law of thermodynamics, all energy transfers involve the loss of some amount of ...
Imposed Oscillations of Kinetic Barriers Can Cause an Enzyme To
... reaction. The sign of AG defines the direction of spontaneous reaction, and the condition AG = 0 defines the point of chemical equilibrium. This is strictly true for elementary reactions-reactions that pass through only one local maximum (the transition state) along the reaction coordinate connectin ...
... reaction. The sign of AG defines the direction of spontaneous reaction, and the condition AG = 0 defines the point of chemical equilibrium. This is strictly true for elementary reactions-reactions that pass through only one local maximum (the transition state) along the reaction coordinate connectin ...
structure and function of procaryotic cells
... Fimbriae are shorter and stiffer than flagella, and slightly smaller in diameter. Generally, fimbriae have nothing to do with bacterial movement (there are exceptions). Fimbriae are very common in Gram-negative bacteria, but occur in some archaea and Gram-positive bacteria as well. Fimbriae are most ...
... Fimbriae are shorter and stiffer than flagella, and slightly smaller in diameter. Generally, fimbriae have nothing to do with bacterial movement (there are exceptions). Fimbriae are very common in Gram-negative bacteria, but occur in some archaea and Gram-positive bacteria as well. Fimbriae are most ...
Raven/Johnson Biology 8e Chapter 7 – Answers 1. An autotroph is
... D. Answer d is incorrect. Cellular respiration involves a complex series of redox reactions. The use of multiple small reactions allows the cell to capture the energy stored in the glucose molecules. 6. The majority of the ATP produced during aerobic respiration is made by— a. the electrons carried ...
... D. Answer d is incorrect. Cellular respiration involves a complex series of redox reactions. The use of multiple small reactions allows the cell to capture the energy stored in the glucose molecules. 6. The majority of the ATP produced during aerobic respiration is made by— a. the electrons carried ...
Raven/Johnson Biology 8e
... D. Answer d is incorrect. Cellular respiration involves a complex series of redox reactions. The use of multiple small reactions allows the cell to capture the energy stored in the glucose molecules. 6. The majority of the ATP produced during aerobic respiration is made by— a. the electrons carried ...
... D. Answer d is incorrect. Cellular respiration involves a complex series of redox reactions. The use of multiple small reactions allows the cell to capture the energy stored in the glucose molecules. 6. The majority of the ATP produced during aerobic respiration is made by— a. the electrons carried ...
Oxidative phosphorylation
Oxidative phosphorylation (or OXPHOS in short) is the metabolic pathway in which the mitochondria in cells use their structure, enzymes, and energy released by the oxidation of nutrients to reform ATP. Although the many forms of life on earth use a range of different nutrients, ATP is the molecule that supplies energy to metabolism. Almost all aerobic organisms carry out oxidative phosphorylation. This pathway is probably so pervasive because it is a highly efficient way of releasing energy, compared to alternative fermentation processes such as anaerobic glycolysis.During oxidative phosphorylation, electrons are transferred from electron donors to electron acceptors such as oxygen, in redox reactions. These redox reactions release energy, which is used to form ATP. In eukaryotes, these redox reactions are carried out by a series of protein complexes within the inner membrane of the cell's mitochondria, whereas, in prokaryotes, these proteins are located in the cells' intermembrane space. These linked sets of proteins are called electron transport chains. In eukaryotes, five main protein complexes are involved, whereas in prokaryotes many different enzymes are present, using a variety of electron donors and acceptors.The energy released by electrons flowing through this electron transport chain is used to transport protons across the inner mitochondrial membrane, in a process called electron transport. This generates potential energy in the form of a pH gradient and an electrical potential across this membrane. This store of energy is tapped by allowing protons to flow back across the membrane and down this gradient, through a large enzyme called ATP synthase; this process is known as chemiosmosis. This enzyme uses this energy to generate ATP from adenosine diphosphate (ADP), in a phosphorylation reaction. This reaction is driven by the proton flow, which forces the rotation of a part of the enzyme; the ATP synthase is a rotary mechanical motor.Although oxidative phosphorylation is a vital part of metabolism, it produces reactive oxygen species such as superoxide and hydrogen peroxide, which lead to propagation of free radicals, damaging cells and contributing to disease and, possibly, aging (senescence). The enzymes carrying out this metabolic pathway are also the target of many drugs and poisons that inhibit their activities.