Anaerobic glycolysis
... • Generates 2 molecules of ATP (substrate-level phosphorylation) and 2 NADH • Pyruvate can enter mitochondria for complete oxidation to CO2 in TCA + electron transport chain • Anaerobic glycolysis reduces pyruvate to lactate, and recycles (wastes) NADH -> NAD+ • Key enzymes of glycolysis are regulat ...
... • Generates 2 molecules of ATP (substrate-level phosphorylation) and 2 NADH • Pyruvate can enter mitochondria for complete oxidation to CO2 in TCA + electron transport chain • Anaerobic glycolysis reduces pyruvate to lactate, and recycles (wastes) NADH -> NAD+ • Key enzymes of glycolysis are regulat ...
Potential energy - Madeira City Schools
... 3. Endergonic reaction – requires a net input of energy a. yield products rich in potential energy – stored in covalent bonds of the products b. photosynthesis (CO2 + H2O + sunlight glucose + O2) 4. Exergonic reaction – chemical reaction that releases energy a. wood burning b. cellular respiration ...
... 3. Endergonic reaction – requires a net input of energy a. yield products rich in potential energy – stored in covalent bonds of the products b. photosynthesis (CO2 + H2O + sunlight glucose + O2) 4. Exergonic reaction – chemical reaction that releases energy a. wood burning b. cellular respiration ...
File - Ms. Perez`s Science
... o Globular proteins that act as catalysts (activators) for metabolic reactions o Catalysts speed up reactions by lowering the activation energy needed for the reaction to take place. o Substrate: the substance(s) that enzymes act on o Enzymes are substrate-specific. (Selective – only interact w/spec ...
... o Globular proteins that act as catalysts (activators) for metabolic reactions o Catalysts speed up reactions by lowering the activation energy needed for the reaction to take place. o Substrate: the substance(s) that enzymes act on o Enzymes are substrate-specific. (Selective – only interact w/spec ...
emboj7600663-sup
... using DM (Cowtan and Main, 1996). The NMR structure of L2 (PDB code: 1FYC) (Howard et al., 1998) was fitted in the improved density and re-modeled manually using the program O (Jones et al., 1991). During subsequent refinements, a lipoyl acid, ADP or ATP, a magnesium ion, potassium ions, and water m ...
... using DM (Cowtan and Main, 1996). The NMR structure of L2 (PDB code: 1FYC) (Howard et al., 1998) was fitted in the improved density and re-modeled manually using the program O (Jones et al., 1991). During subsequent refinements, a lipoyl acid, ADP or ATP, a magnesium ion, potassium ions, and water m ...
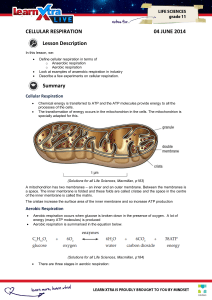
CELLULAR RESPIRATION 04 JUNE 2014 Lesson Description
... Oxidative phosphorylation: takes the energy from the energy-rich hydrogens to make ATP. The energy depleted hydrogens combine with oxygen to make water. This is either breathed out as water vapour or excreted via the kidneys. ...
... Oxidative phosphorylation: takes the energy from the energy-rich hydrogens to make ATP. The energy depleted hydrogens combine with oxygen to make water. This is either breathed out as water vapour or excreted via the kidneys. ...
Chapter 9
... transport chain • Unlike an uncontrolled reaction, the electron transport chain passes electrons in a series of steps instead of one explosive reaction • Oxygen pulls electrons down the chain in an energy-yielding tumble • The energy yielded is used to regenerate ATP Copyright © 2005 Pearson Educati ...
... transport chain • Unlike an uncontrolled reaction, the electron transport chain passes electrons in a series of steps instead of one explosive reaction • Oxygen pulls electrons down the chain in an energy-yielding tumble • The energy yielded is used to regenerate ATP Copyright © 2005 Pearson Educati ...
video slide - Somers Public Schools
... transport chain • Unlike an uncontrolled reaction, the electron transport chain passes electrons in a series of steps instead of one explosive reaction • Oxygen pulls electrons down the chain in an energy-yielding tumble • The energy yielded is used to regenerate ATP Copyright © 2005 Pearson Educati ...
... transport chain • Unlike an uncontrolled reaction, the electron transport chain passes electrons in a series of steps instead of one explosive reaction • Oxygen pulls electrons down the chain in an energy-yielding tumble • The energy yielded is used to regenerate ATP Copyright © 2005 Pearson Educati ...
The laws of cell energetics
... Lipmann's rule, assuming that not only ATP but also A & + and ADNa + can couple energy-releasing and energy-consuming processes. In the great majority of bioenergetic processes, such a coupling is carried out with the use of one of the three abovementioned energy currencies. I t does not mean, howev ...
... Lipmann's rule, assuming that not only ATP but also A & + and ADNa + can couple energy-releasing and energy-consuming processes. In the great majority of bioenergetic processes, such a coupling is carried out with the use of one of the three abovementioned energy currencies. I t does not mean, howev ...
No Slide Title - Kinver High School
... • PC is a very easy compound to break down. As it is stored in the muscle cells, it is readily available and does not need oxygen. • ATP can be made quickly with no fatiguing waste products ...
... • PC is a very easy compound to break down. As it is stored in the muscle cells, it is readily available and does not need oxygen. • ATP can be made quickly with no fatiguing waste products ...
Chapter 34 HEIN
... • This polymer breaks down to glucose, which is oxidized to replenish the ATP supply. • Because glucose oxidation is a complex process, muscle contraction must proceed at a slower rate. • This energy supply is only useful for about 2 minutes of work; muscles rapidly deplete their glycogen stores and ...
... • This polymer breaks down to glucose, which is oxidized to replenish the ATP supply. • Because glucose oxidation is a complex process, muscle contraction must proceed at a slower rate. • This energy supply is only useful for about 2 minutes of work; muscles rapidly deplete their glycogen stores and ...
NVC Bio 120 lect 9 cell respiration
... Electron transfer in the electron transport chain causes proteins to pump H+ from the mitochondrial matrix to the intermembrane space H+ then moves back across the membrane, passing through the proton, ATP synthase ...
... Electron transfer in the electron transport chain causes proteins to pump H+ from the mitochondrial matrix to the intermembrane space H+ then moves back across the membrane, passing through the proton, ATP synthase ...
Enzymes
... place in cells. 2. Enzymes are very specific, generally catalyzing only one chemical reaction. 3. For this reason, part of an enzyme’s name is usually derived from the reaction it catalyzes. Enzymes usually end in the suffix “–ase”. Ex. Alcohol dehydrogenase catalyzes the reaction that removes water ...
... place in cells. 2. Enzymes are very specific, generally catalyzing only one chemical reaction. 3. For this reason, part of an enzyme’s name is usually derived from the reaction it catalyzes. Enzymes usually end in the suffix “–ase”. Ex. Alcohol dehydrogenase catalyzes the reaction that removes water ...
Transmembrane Transport of Ions and Small Molecules
... • The animal cell plasma membrane resting potential is generated by the ATP-powered Na+/K+ pump and nongated K+ channels. • The structure and chemical nature of a channel pore lowers the activation energy for passage of a specific ion over other ions, which may be even smaller. • Patch-clamping tech ...
... • The animal cell plasma membrane resting potential is generated by the ATP-powered Na+/K+ pump and nongated K+ channels. • The structure and chemical nature of a channel pore lowers the activation energy for passage of a specific ion over other ions, which may be even smaller. • Patch-clamping tech ...
Chapters 4 and 5 Mrs. Svencer CP Biology 4.1 Life Requires About
... All amino acids consist of a central carbon bonded to an amino group, a carboxyl group, and a hydrogen atom. The fourth bond is with a unique side group. The differences in side groups convey different properties to each amino acid. ...
... All amino acids consist of a central carbon bonded to an amino group, a carboxyl group, and a hydrogen atom. The fourth bond is with a unique side group. The differences in side groups convey different properties to each amino acid. ...
HCC Learning Web
... Chemical Cycling between Photosynthesis and Cellular Respiration • The ingredients for photosynthesis are carbon dioxide and water. – CO2 is obtained from the air by a plant’s leaves. – H2O is obtained from the damp soil by a plant’s roots. ...
... Chemical Cycling between Photosynthesis and Cellular Respiration • The ingredients for photosynthesis are carbon dioxide and water. – CO2 is obtained from the air by a plant’s leaves. – H2O is obtained from the damp soil by a plant’s roots. ...
Researchers Are First To Simulate The Binding Of Molecules To A
... flip into an orientation that allowed it to bind to the carrier. The identified binding pocket for ADP explained a lot of known experimental data, and revealed an unusual feature of the carrier protein: Its binding site and the entryway leading to it had an extremely positive electrical charge. It h ...
... flip into an orientation that allowed it to bind to the carrier. The identified binding pocket for ADP explained a lot of known experimental data, and revealed an unusual feature of the carrier protein: Its binding site and the entryway leading to it had an extremely positive electrical charge. It h ...
fermentation
... 3. The reactions of fermentation occur completely in the cytosol. Because of its increased efficiency, aerobic respiration is generally the preferred path for cells to take when they need to produce energy. However, in environments where oxygen is scarce, and sugar is plentiful, many organisms thri ...
... 3. The reactions of fermentation occur completely in the cytosol. Because of its increased efficiency, aerobic respiration is generally the preferred path for cells to take when they need to produce energy. However, in environments where oxygen is scarce, and sugar is plentiful, many organisms thri ...
Unit 2 - Biochemistry Notes
... Compound – when different elements combine. CO2 and H2O are molecules, but they are also compounds because they are molecules containing more than one element. ...
... Compound – when different elements combine. CO2 and H2O are molecules, but they are also compounds because they are molecules containing more than one element. ...
File
... • Electrons are transferred from NADH or FADH2 to the electron transport chain • Electrons are passed through a number of proteins including cytochromes (each with an iron atom) to O2 • The electron transport chain generates no ATP • The chain’s function is to break the large freeenergy drop from f ...
... • Electrons are transferred from NADH or FADH2 to the electron transport chain • Electrons are passed through a number of proteins including cytochromes (each with an iron atom) to O2 • The electron transport chain generates no ATP • The chain’s function is to break the large freeenergy drop from f ...
CLEP Biology - Problem Drill 06: Metabolism and Cellular
... Oxidative Phosphorylation NADH and FADH2 carry electrons to protein machines. NADH and FADH2 pass electrons to machines, which pass them to the final acceptor: oxygen. As they pass electrons, machines pump hydrogen ions to create a concentration gradient. Hydrogen ions flow back due to concentration ...
... Oxidative Phosphorylation NADH and FADH2 carry electrons to protein machines. NADH and FADH2 pass electrons to machines, which pass them to the final acceptor: oxygen. As they pass electrons, machines pump hydrogen ions to create a concentration gradient. Hydrogen ions flow back due to concentration ...
U2-D3-03 – PO and Kreb
... The two molecules of acetyl-CoA enter the Krebs cycle where additional free energy transfers occur. The two molecules of NADH proceed to stage 4 (electron transport and chemiosmosis) to produce ATP by oxidative phosphorylation. The two CO 2 molecules produced during pyruvate oxidation diffuse out of ...
... The two molecules of acetyl-CoA enter the Krebs cycle where additional free energy transfers occur. The two molecules of NADH proceed to stage 4 (electron transport and chemiosmosis) to produce ATP by oxidative phosphorylation. The two CO 2 molecules produced during pyruvate oxidation diffuse out of ...
Oxidative phosphorylation
Oxidative phosphorylation (or OXPHOS in short) is the metabolic pathway in which the mitochondria in cells use their structure, enzymes, and energy released by the oxidation of nutrients to reform ATP. Although the many forms of life on earth use a range of different nutrients, ATP is the molecule that supplies energy to metabolism. Almost all aerobic organisms carry out oxidative phosphorylation. This pathway is probably so pervasive because it is a highly efficient way of releasing energy, compared to alternative fermentation processes such as anaerobic glycolysis.During oxidative phosphorylation, electrons are transferred from electron donors to electron acceptors such as oxygen, in redox reactions. These redox reactions release energy, which is used to form ATP. In eukaryotes, these redox reactions are carried out by a series of protein complexes within the inner membrane of the cell's mitochondria, whereas, in prokaryotes, these proteins are located in the cells' intermembrane space. These linked sets of proteins are called electron transport chains. In eukaryotes, five main protein complexes are involved, whereas in prokaryotes many different enzymes are present, using a variety of electron donors and acceptors.The energy released by electrons flowing through this electron transport chain is used to transport protons across the inner mitochondrial membrane, in a process called electron transport. This generates potential energy in the form of a pH gradient and an electrical potential across this membrane. This store of energy is tapped by allowing protons to flow back across the membrane and down this gradient, through a large enzyme called ATP synthase; this process is known as chemiosmosis. This enzyme uses this energy to generate ATP from adenosine diphosphate (ADP), in a phosphorylation reaction. This reaction is driven by the proton flow, which forces the rotation of a part of the enzyme; the ATP synthase is a rotary mechanical motor.Although oxidative phosphorylation is a vital part of metabolism, it produces reactive oxygen species such as superoxide and hydrogen peroxide, which lead to propagation of free radicals, damaging cells and contributing to disease and, possibly, aging (senescence). The enzymes carrying out this metabolic pathway are also the target of many drugs and poisons that inhibit their activities.