* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Cellular Respiration - Seattle Central College
Fatty acid synthesis wikipedia , lookup
Amino acid synthesis wikipedia , lookup
Basal metabolic rate wikipedia , lookup
Butyric acid wikipedia , lookup
Fatty acid metabolism wikipedia , lookup
Lactate dehydrogenase wikipedia , lookup
Metalloprotein wikipedia , lookup
Photosynthesis wikipedia , lookup
Light-dependent reactions wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
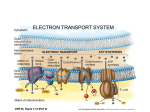
Electron transport chain wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
NADH:ubiquinone oxidoreductase (H+-translocating) wikipedia , lookup
Adenosine triphosphate wikipedia , lookup
Microbial metabolism wikipedia , lookup
Oxidative phosphorylation wikipedia , lookup
Biochemistry wikipedia , lookup
Citric acid cycle wikipedia , lookup
Cellular Respiration • Glycolysis – each glucose molecule is split and converted to pyruvate (2-three carbon units. • Stages of glycolysis Stage 1: glucose is phosphorylated and cleaved to form to 2 molecules of glyceraldyde-3-phosphate (G-3-P). The two ATP molecules consumed during this stage are later used in ATP synthesis. Stage 2: G-3-P is converted to pyruvate. Four ATP molecules and two NADH are produced. ATP are consumed in the formation of Glucose-6-phosphate from glucose and the conversion of Fructose-6-phosphate to Fructose-1,6-diphosphate. The net production of ATP per glucose is 2. • The energy captured during the process is stored temporarily in 2 molecules each of ATP and NADH. • The pyruvate can be oxidized to CO2 and H2O in aerobic respiration . • In anaerobic respiration the pyruvate is converted to either lactic acid or ethanol. • In the presence of oxygen, the energy rich pyruvate produce substantial amount of ATP through the TCA cycle. • In the absence of oxygen, additional ATP cannot be generated from pyruvate. • Under anaerobic conditions, alcoholic fermentation occurs in yeast. Pyruvate is decarboxyated to form acetaldehyde, which then reduced by NADH to form ethanol, • Yeast also contain the enzyme lactic acid dehydrogenase which catalyzes the reduction of pyruvic acid to lactic acid. NADH is oxidized to NAD+ in the process. Image from Purves et al., Life: The Science of Biology, 4th Edition, by Sinauer Associates and WH Freeman NADH • The transfer of electrons from biomolecules is initiated by a compound that is abbreviated as NAD+, Nicotinamide Adenine Dinucleotide. • This is an oxidation reaction where 2 hydrogen atoms (or 2 hydrogen ions and 2 electrons) are removed from the organic metabolite. When electrons are transferred from a metabolite by NAD+, NAD+ additionally removes two protons, hydrogen ions, H+ MH2 + NAD+ → NADH + H+ + M + energy This converts NAD+ into NADH + H+ In such reactions NAD+ serves as the electron acceptor and the metabolite from which the electrons are stripped serves as the electron donor. • NAD+ is reduced to form NADH + H+ and the electron-donating metabolite is similarly oxidized Procedure: Part 1 • • Fermentation: Measure the volume of CO2 produced during fermentation. #4. Heat water on a hotplate and use the hotwater to keep the waterbath constant. After setting up the experiment proceed to Part 2. Part 2 Glycolysis: Methylene blue (MB) is used in place of NAD+ and is blue in the oxidized state and is colorless when in the reduced state (MBH2) mimicking NADH. #3. Place the test tubes marked with you initial in the 37o C incubator. Keep the glass door closed to examine the color. Heading for Table 2: Write the Initial Color on the 0 minute column Column 4 Final Color