* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download From Natural Product to clinical trial
Development of analogs of thalidomide wikipedia , lookup
Discovery and development of angiotensin receptor blockers wikipedia , lookup
Drug discovery wikipedia , lookup
NK1 receptor antagonist wikipedia , lookup
Metalloprotease inhibitor wikipedia , lookup
Theralizumab wikipedia , lookup
HIV vaccine wikipedia , lookup
Discovery and development of proton pump inhibitors wikipedia , lookup
Discovery and development of cephalosporins wikipedia , lookup
Discovery and development of HIV-protease inhibitors wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
CCR5 receptor antagonist wikipedia , lookup
Discovery and development of ACE inhibitors wikipedia , lookup
Discovery and development of non-nucleoside reverse-transcriptase inhibitors wikipedia , lookup
Discovery and development of neuraminidase inhibitors wikipedia , lookup
Discovery and development of integrase inhibitors wikipedia , lookup
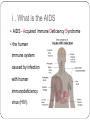
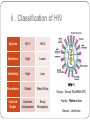
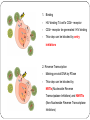
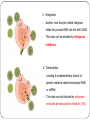
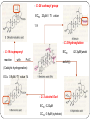
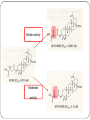
From Natural Product to Clinical Trials: Bevirimat, a Plant-Derived Anti-AIDS Drug 천연물약품화학연구실 석 사 1 기 장 성 완 Index 0. AIDS 1. Introduction 2. Bioactivity-directed Fractionation and Isolation 3. Lead Identification 4. Lead Optimisation and SAR Study 4.1 Modification of the BA Triterpene Skeletion 4.2 Modification on C-3 Position of BA 4.3 Introduction of C-28 Side Chain into BA 4.4 Bifunctional BA Analouges – Potential for Maturation Inhibitor Development 5. Mechanism of Action Studies of Bevirimat 6. Preclinical Studies of Bevirimat 7. Clinical Trials and Current Status of Bevirimat 8. Conclusions 0. AIDS ⅰ. What is the AIDS ⅱ. Classification of HIV ⅲ. HIV Life Cycle ⅳ. Current status ; Anti-HIV Drug ⅴ. Domestic research trend ⅰ. What is the AIDS AIDS - Acquired Immune Deficiency Syndrome the human immune system caused by infection with human immunodeficiency virus (HIV). ⅱ. Classification of HIV Species HIV-1 HIV-2 Virulence High Lower Infectivity High Low HIV-1 Prevalence Global West Africa Group : Group Ⅵ(ssRNA-RT) Inferred Origin Common chimpanzee Sooty Mangabey Family : Retrociridae Genus : Lentivirus ⅲ. HIV Life Cycle 1 2 1. Binding 2. Reverse Transcription 3 4 3. Integration 4. Transcription 5. Translation 5 6. Viral Assembly and 6 Maturation 1. Binding - HIV binding T4-cell’s CD4+ receptor - CD4+ receptor be generated HIV binding - This step can be blocked by entry inhibitors 2. Reverse Transcription - Making proviral DNA by RTase - This step can be blocked by NRTIs(Nucleoside Reverse Transcripatase Inhibitors) and NNRTIs (Non-Nucleoside Reverse Transcriptase Inhibitors) 3. Integration - another viral enzyme called integrase hides the proviral DNA into the cell's DNA - This step can be blocked by intergrase inhibitors 4. Transcription - creating a complementary strand of genetic material called messenger RNA or mRNA - This step can be blocked by antisense antivirals or transcription inhibitors (TIs), 5. Translation - The mRNA carries instructions for making new viral proteins from the nucleus 6. Viral Assembly and Maturation -Final step virus infection -Viral Assembly step can be blocked by Protease Inhibitors (PIs) -Maturation step can be blocked by Maturation Inhibitors(Mis) ⅳ. Current status ; Anti-HIV Drug ⅳ. Current status ; Anti-HIV Drug ⅴ. Domestic research trend Patent - 태림제약 anti-virus drug Raw matrial supply - 유한양행 GWM사와 계약 Technology Licensing - 화학硏 - Gilead 1. Introduction 29 approved drugs marketed to treat HIV-1 infection ex) Fuzeon [enfuvirtide,ENF,T-20] – inhibit viral entry Selzentry[maraviroc,MVC] - CCR5 coreceptor antagonist HAART(Highly active antiretroviral therapy) combination of drugs with different targets HAART improved patient’s life quality and length But HAART made drug-resistant virus up to 78% if HIV-1-infected individuals harbour drug-resistance virus with a rapidly growing subgroup Therefore, novel potent antiviral agents need 2. Bioactivity-directed Fractionation and Isolation QSAR(Qunatitative Structure-activity relationship) ADMET (absorption, distribution, metabolism, excretion, toxicity) 3. Lead Identification - Triterpene have diverse structure and pharmacological activities - Several naturally occurring triterpenes have been reported to show anti-HIV activity - Bevirimat, the first in a new class of compound termed HIV maturation inhibitor(Mis) - betulinic acid and platanic acid can inhibit HIV-1ⅢB replication (EC50/bet 1.4 μM / EC50/pla 6.5 μM) - betulinic acid contains many plant species ( e.g Tryphyllum peltatum, Ancistrocladus heyneanus, Ziziphi fructus, Tetracera boloviana ) - betulin can modify to betulinic acid (betulin in birch bark) 4.Lead Optimisation and SAR Study 4.1 Modification of the BA Triterpene Skeletion - Betulinic acid have a readily chemical modification site C–3 alcohol C–19 isopropenyl - C–3 alcohol C–28 carboxyl group - C–19 isopropenyl - C–28 carboxyl group This functional group This functional group This functional group can undergoes typical reaction be derivatised providing with allylic group ester and amide can be eliminated or oxidised - C–28 carboxyl group EC50 23μM / TI value 1.9 - C-2 Hydroxylation EC50 - C–19 isopropenyl reaction with Pd-C activity) (Catalytic hydrogenation) EC50 0.9μM / TI value 14 - C–3 alcohol Oxd EC50 0.22μM CC50 0.9μM (cytotoxic) 42.3μM(weak 4.2 Modification on C-3 Position of BA Com 8, 9 ) Extremely potent anti-HIV - C-3 hydroxyl group readily acylated with variety of anhydrides and acid chlorides 1 activity Com 15, 16) similar potencties compare com 8,9 Com 10, 17 ) reduction in antiviral 2 activity Com 13, 20~22 ) lack or no antiviral activity Com 7, 11, 12, 14, 18 ) partial antiviral activity - C-3 modification’s result indicates that the two C-3’ methyl group in the bevirimat side chain contribute anti-HIV activity and terminal carboxyl acid substitution has antiviral activity potent Similar activity Moderate activity - C-3 position of BA and betulin, geminal dimethyl substitution of the side chain C-3’ position coupled with a terminal carboxylic acid are important to the enhanced antiviral activity for both of templates these triterpene C-3 stereo C-3 stereo convert eliminate Two compound anti-viral activity dramatically reduced, indicating the importance of the 3-C stereo R S - C-3 oxygen exchange with nitrogen Only com 42, 45 weak antiviral activity - com 46 have similar activity compare buletin(com 4) but have cytotoxic (EC50 0.22μM / CC50 0.9μM) - Similary com 47 has antiviral activity and cytotoxic (EC50 0.22μM / CC50 0.9μM) - com 48~50 has similar antiviral activity but decrease cytotoxic (EC50 0.57~4.57μM / CC50 >15μM) 4.3 Introduction of C-28 Side Chain into BA - These type functions by blocking the viral entry into the host cells - Increasing chain length influence the anti-HIV potency - compound with amide side chains between aminooctanoic acid and aminododecanoic acid tend to increase antiviral potency - com 51 inhibits the infectivity of several HIV-1 strains (EC50 0.05~2μM) - com 52 equipotent antiviral activity - com 53 have equipotent against HIV-1 virus when compare with com 51, com 52 - com 51 show potent antiviral activity in vitro but its clinical development was stopped due to ‘pharmacodynamic properties’ poor 4.4 Bifunctional BA Analouges – Potential for Maturation Inhibitor Development - Two functionalities are on the opposite position of the BA skeleton - a design combining both modifications led to the development of bi-functional BA analogues - EC50 0.0026μM higher potent than C-3 and C-28 parent compound - This category preserves both the anti HIV-1 entry and maturation inhibit ability 5. Mechanism of Action Studies of Bevirimat - Bevirimat affect virus replication at a time point after the completion of viral DNA integration and Tat expression - inhibited the processing of the viral Gag polyprotein at a specific step - Bevirimat does not affect viral RT, reverse protease, virus particle budding - Bevirimat blocks processing of CA- SP1(p25) to CA (p24) which necessary for final capsid condensation and formation of infectious, bevirimat cause abnormal morphology to virus - consequently virus become non-infectious - Bevirimat has no effect on processing at other site of Gag Gag region relate virion structural proteins protase - Bevirimat-resistant isolates were selected by serial passage of HIV-1 - resistant virus found several point mutations proximal to the cleavage site of CA-SP1 CA H226Y, L231M, L231F, SP1 A1V and A3V - no mutation observed in other region of Gag or in the PR coding region - Bevirimat does not affect SIV / HIV-2 6. Preclinical Studies of Bevirimat - EC50 10.3nM against subtype B virus (similar activity compare withAZT,nevirapine) -Bevirimat retains nanomolar inhibitory activity against drug resistant HIV strains(NRTIs, NNRTIs, PIs resistant) - no activity against HIV-2, SIV and other similar envelop virus type Mouse type : SCID-hu Thy/Liv >2log10 in HIV-1 RNA Does : Twice daily / Oral ≥ 90% p24 conc. Virus : HIV-1 infected Gag-p24+ thymocytes at 100mg/kg per day Dose-dependent manner Observe in the mice at plasma conc. 7. Clinical Trials and Current Status of Bevirimat - Phase Ⅰ(In 2004 completed) PanocosPharm dose single and multiple phase clinical trials of bevirimat in healthy volunteers / Oral solution It well tolreated half-life of ~ 2.5-3 days / supporting daily dosing regimen - Phase Ⅱa (In August 2005, completed) random double-blind, bevirimat monotherapy for ten days resulted in statistically significant reduction in viral load compare with placebo (individual decrease up to 1.7log10, 100 and 200mg dose) No evidence develop drug resistant virus 7. Clinical Trials and Current Status of Bevirimat - Phase Ⅱb (In 2008 completed) patients failing HIV therapy due to drug resistance Good activity in 369, 370, 371 on Gag amino acid sequence mutant, less activity in polymorphism(variants) 20μg/mL bevirimat is required for a robust response the mean viral load reduction was -1.18log10copies/mL after 14days in the patients who free of key baseline Gag polymorphism and bevirimat conc. Minimum target of 20μg/mL 91% of patients with these two response predictors han at least a 0.5log10 viral load reduction by week 2 with a maximum treatment response of 2.03log10 7. Clinical Trials and Current Status of Bevirimat - Phase Ⅱb (In 2008 completed) bevirimat well tolerated, with a safety profile comparable to placebo through the 14days of treatment Bevirimat tablet formulation dosed twice daily achieved target plasma levels After 14 days of bevirimat treatment given twice daily at doses of 200mg or 300mg(using the 50 mg tablet) 32% patient (treatment naïve and experienced) had plasma concentrations well above the previously identified minimum target of 20μg/mL PhaseⅢ are being planned 8. Conclusions - HAART has led to a significant improvement in the health and life span HIV-1-infected patients - But, major problem is the increasing prevalence of virus strains that are resistant to approved drug - Plants are a major source of biologically active compounds and can provide good leads that are structurally unique and/or have new mechanism of action Thank You for Your Attention