* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download The Origin of the Elements - Indiana University Astronomy
Survey
Document related concepts
Theoretical astronomy wikipedia , lookup
Non-standard cosmology wikipedia , lookup
Extraterrestrial life wikipedia , lookup
Corvus (constellation) wikipedia , lookup
Physical cosmology wikipedia , lookup
Stellar classification wikipedia , lookup
Timeline of astronomy wikipedia , lookup
H II region wikipedia , lookup
Big Bang nucleosynthesis wikipedia , lookup
Stellar kinematics wikipedia , lookup
Astronomical spectroscopy wikipedia , lookup
Chronology of the universe wikipedia , lookup
Star formation wikipedia , lookup
Transcript
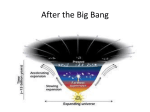
H205 Cosmic Origins APOD Tom Lehrer's "The Elements". A Flash animation by Mike Stanfill, Private Hand Our last class! Origin of the Elements Hand in EP7 Abundance of Elements in the Galaxy How are the chemical elements created in the Early Universe? in Stars? in Supernovae? How is the Galaxy enriched in chemical elements? Big Bang Nucleosynthesis Within first three minutes, hydrogen & helium formed At t =1 s, T=10,000,000,000 K: soup of particles: photons, electrons, positrons, protons, neutrons. Particles created & destroyed At t =3 min, T=1,000,000,000 K: p+n => D D + D => He Primordial Nucleosynthesis Hydrogen and helium were created during the Big Bang while the Universe was cooling from its initial hot, dense state. About 10% of the lithium in the Universe today was also created in the Big Bang. We’re still not sure where the rest comes from. The first stars formed from this material. The Creation of Elements… “Just between you and me, where does it get enriched?” STARS! The Composition of Stars 90% hydrogen atoms 10% helium atoms Less than 1% everything else (and everything else is made in stars!) everything else The Evolution of Stars Making Elements in Stars Some elements are created in the cores of low mass stars like the Sun Small Stars: Fusion of light elements Fusion: (at 15 million degrees !) 4 (1H) => 4He + 2 e+ + 2 neutrinos + energy Where does the energy come from ? Mass of four 1H > Mass of one 4He E= 2 mc Hydrogen Burning Stars burn hydrogen in their interiors to produce helium. Hydrogen burning also rearranges carbon, nitrogen, and oxygen. Small Stars to Red Giants After Hydrogen is exhausted in core, energy released from nuclear fusion no longer counteracts inward force of gravity. • Core collapses, Kinetic energy of collapse converted into heat. This heat expands the outer layers. • Meanwhile, as core collapses, Increasing temperature and pressure ... A Red Giant You Know Beginning of Heavier Elements At 100 million degrees Celsius, helium fuses: 3 (4He) => 12C + energy After helium exhausted, a small star is not large enough to reach temperatures necessary to fuse carbon to heavier elements Helium Burning Three helium atoms combine to form carbon The end for small stars After helium exhausted, outer layers of star expelled Planetary Nebulae Heavy Elements from Massive Stars Large stars also fuse hydrogen into helium, and helium into carbon But their larger masses lead to higher temperatures, which allow fusion of carbon into magnesium, etc. “Alpha” Elements The “Odd-Even” Effect Carbon, oxygen, neon, magnesium, silicon, sulfur, arcon, calcium are **high** Nitrogen, fluorine, sodium, aluminum, phosphorus, chlorine, potassium are **low** Stellar Senior Citizens Many elements are made in supernovae when massive stars explode When stars finally deplete their nuclear fuel, they become white dwarfs, neutron stars, or black holes. In the process, much of the stellar material is returned to interstellar space Supernova Fusion of iron takes energy, rather than releases energy So fusion stops at iron No nuclear fusion energy to balance inward force of gravity Nothing to stop gravity Massive star ends its life in supernova explosion The Iron Peak Metals In the cores of massive stars just before supernova explosions, atomic nuclei exchange protons and neutrons to form the iron peak metals. The “Iron Peak” The “transition metals” vanadium through zinc, including copper, iron, and nickel are produced in supernove explosions Most of the SN production is through Type IA supernovae (exploding white dwarfs) Supernovae Explosive power of supernovae: Creates new elements Disperses elements created in large stars All X-ray Energies Calcium Silicon Iron • Hydrogen – from big bang nucleosynthesis • Helium – from big bang and from hydrogen burning via the p-p chain and CNO cycle • Nitrogen – from CNO cycle • Carbon, Oxygen – from helium burning • Light elements (Neon, Magnesium, Calcium – from carbon and oxygen burning • Iron metals – from the final burning Making Elements Up to Iron Elements Heavier than Iron … • Once iron is formed, it is no longer possible to create energy via fusion. Elements heavier than iron require a different process (Iron is atomic number 26.) • The heaviest naturally occurring nucleus is uranium (atomic number 92). How do we get to uranium then? •Elements heavier than iron are created by neutron capture •The neutron is converted into a proton and added to the nucleus, increasing the atomic number to make the next element in the periodic table. Making Heavy Metals in Stars • In low mass stars like the Sun, heavy metals are created when the star is a giant • Massive stars make heavy metals when they become supernovae Isotopes built by n-capture syntheses The valley of beta-stability Rolfs & Rodney (1988) How to Make Heavy Metals: neutron-capture processes Supernovae High neutron flux Type II Supernovae (massive stars) No time for b-decay Makes Eu, Gd, Dy Plus some Sr, Y, Zr, Ba, La… Red Giants Low neutron flux Time for b-decay before next neutron capture Makes Sr, Y, Zr, Ba, La, Ce But no Eu, Gd, Dy Heavy Metals All heavier elements are formed when iron peak elements capture neutrons Nucleosynthesis from Cosmic Rays Lithium, beryllium, and boron are difficult to produce in stars Li, Be, and B are formed in the fusion chains, but they are unstable at high temperatures, and tend to break up into residues of He, which are very stable So what is the origin of these rare elements? Collisions of Cosmic Rays with hydrogen & helium in interstellar space Cosmic Rays Collisions with ISM Cosmic ray Light nucleus Interstellar matter (~1 hydrogen atom per cm3) Light nucleus Lithium, beryllium, and boron and sub-iron enhancements attributed to nuclear fragmentation of carbon, nitrogen, oxygen, and iron with interstellar matter (primarily hydrogen and helium). (CNO or Fe) + (H & He)ISM (LiBeB or sub-Fe) Cosmic Elements White - Big Bang Pink - Cosmic Rays Yellow - Small Stars Green - Large Stars Blue - Supernovae Nucleosynthesis in Stars p3 Betelgeuse steadily making He. Future C, N Red Giant making Ca and beyond. Future supernova. Orion Nebula New stars getting heavy elements. Future Earths? Rigel - Blue Supergiant making, He, C, N. Future heavy elements. Matching Our Universe What would the composition of our universe be if only very large stars formed? Only very small stars? Composition of the Universe (Actually, this is just the solar system) Composition varies from place to place in universe, and between different objects Chemical Evolution Elements are created in stars and mixed back out into the Galaxy The Galaxy’s composition changes as stars form, evolve, and die Chemical Evolution Nucleosynthesis in stars leads to chemical enrichment of the Galaxy Rate of enrichment depends on sites and Primordial mechanisms of nucleosynthesis nucleosynthesis Hydrogen burning The variables are: Star formation rate Initial mass function Yields Stellar evolution time Proton-capture chains Helium Burning Alpha Process Equilibrium process Neutron-capture processes Odd-ball stuff Nucleosynthesis since the beginning of time By studying stars of different ages, formed at different times in the Galaxy’s history, we can trace the history of the Milky Way The Galaxy (and the universe) is gradually enriched in heavy elements Despite all the nucleosynthesis that has occurred since the creation of the universe, only 2% of the ordinary matter in the universe is now in the form of heavy elements. Most is still hydrogen and helium Your Cosmic Origin We’re Done! • Final Reflection due by 4:45 PM on Friday, May 8 • Earlier submission is welcome!