* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Anaphylaxis_01 (Pathophysiology)
Complement system wikipedia , lookup
Hygiene hypothesis wikipedia , lookup
Adaptive immune system wikipedia , lookup
Molecular mimicry wikipedia , lookup
Psychoneuroimmunology wikipedia , lookup
Polyclonal B cell response wikipedia , lookup
Cancer immunotherapy wikipedia , lookup
Sjögren syndrome wikipedia , lookup
Adoptive cell transfer wikipedia , lookup
Innate immune system wikipedia , lookup
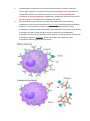
Anaphylaxis (Pathophysiology) INTRODUCTION 1. 2. Anaphylaxis is acute, potentially lethal, multisystem syndrome resulting from sudden release of mast cell- and basophil-derived mediators into circulation. It most often results from immunologic reactions to foods, medications, and insect stings, although it can also be induced through non-immunologic mechanisms by any agent capable of producing sudden, systemic degranulation of mast cells or basophils. The phenomenon of anaphylaxis was first described in modern medical literature in 1902 in a study involving protocols for immunizing dogs with jellyfish toxin. The injection of small amounts of toxin in some animals rather than generating protection precipitated rapid onset of fatal or near-fatal symptoms. The authors named this response "l'anaphylaxie," which is derived from the Greek words a- (against) and phylaxis (immunity or protection). 3. The pathophysiology of anaphylaxis will be reviewed here. The clinical manifestations, diagnosis, and management of anaphylaxis, and the epidemiology and etiology of fatal anaphylaxis are discussed separately. PROPOSED MECHANISM 1. The mechanism responsible for most cases of human anaphylaxis involves IgE. Possible alternative mechanisms remain incompletely understood. Environmental exposures and complex genetic factors may also have important roles, although these are not explored in this review. 2. Terminology A. The term "anaphylaxis" has traditionally been reserved for IgE-dependent events, and term "anaphylactoid reaction" has been used to describe IgE-independent events, although two reactions are often clinically indistinguishable. The World Allergy Organization (WAO), international umbrella organization representing large number of regional and national professional societies dedicated to allergy and clinical immunology, has proposed discarding this nomenclature. WAO categorizes anaphylaxis as either immunologic or non-immunologic, and this is terminology used in this review. B. Immunologic anaphylaxis i. IgE-mediated reactions ii. IgG-mediated reactions (which have not been identified in humans, as discussed below) iii. Immune complex/complement-mediated reactions C. Non-immunologic anaphylaxis i. Non-immunologic anaphylaxis is caused by agents or events that induce sudden, massive mast cell or basophil degranulation in absence of immunoglobulins. 3. Immunologic anaphylaxis A. IgE-mediated i. ii. iii. iv. v. B. The classical mechanism associated with human-allergic disease is initiated by antigen (allergen) interacting with allergen-specific IgE bound to receptor Fc-epsilon-RI on mast cells and/or basophils. The events leading to allergen-specific IgE production in atopic individual are complex. In brief, B cells are driven to differentiate into IgE-producing cells via activity of type 2 subset of CD4-bearing helper T cells (Th2 cells). This process largely takes place in peripheral lymphoid tissues. IL-4 and its receptors (IL-4R-α/γ-c and IL-4R-α/IL-13R-α-1) and IL-13 and its receptor (IL-4R-α/IL-13R-α-1) contribute to IgE responses in humans. Once produced, allergen-specific IgE diffuses through tissues and vasculature and constitutively occupies high-affinity IgE receptors (Fc-epsilon-RI) on mast cells and basophils (figure 1). The generation of allergen-specific IgE is reviewed in more detail separately. When allergen diffuses into proximity of mast cell or basophil, it interacts with any surface-bound IgE that is specific for that allergen. Certain allergens are able to interact with IgE molecules on two or more receptors of cell surface to cause cross-linking, which in turn causes receptors to become aggregated and initiate intracellular signaling. Allergens that are capable of cross-linking are either multivalent (having multiple identical sites for IgE antibody binding) or univalent (having multiple different sites for IgE antibody binding). If signaling is sufficiently robust, mast cell (or basophil) becomes activated and degranulates, releasing preformed mediators, enzymes, and cytokines (such as histamine, tryptase, and TNF) and initiating additional mediator, cytokine, and enzyme production. Mast cell biology is discussed in more depth elsewhere. These mediators either act directly on tissues to cause allergic symptoms or recruit and activate additional inflammatory cells, particularly eosinophils. The recruited cells, in turn, release more mediators and propagate fulminant "chain reaction" of allergic inflammation. The various mediators and cytokines involved are reviewed below. IgG-mediated (in animal models) i. Animal models that appear analogous to human anaphylaxis have been established in mice, pigs, and dogs. Clinically, each has some distinctive signs and symptoms. As example, murine anaphylaxis is characterized by dramatic reductions in core body temperature and subtle cardiopulmonary differences, compared with human anaphylaxis. ii. In mouse models, at least two IgG-mediated pathways have been identified. 1. In one model, allergen interacts with allergen-specific IgG bound to Fc-γ-RIII on macrophages and basophils. This IgG-dependent pathway requires proportionately more antibody and antigen than murine IgE-mediated pathway, and macrophage activation results primarily in release of PAF, rather than histamine. PAF causes platelet aggregation and release of potent vasoconstrictors TxA2 and serotonin, and can act directly on vascular endothelial cells to increase vascular permeability. 2. 3. There is evidence in mice that pathways of IgG- and IgE-mediated anaphylaxis are interrelated. When low doses of allergen are administered, IgG antibody can block IgE-dependent anaphylaxis by intercepting antigen before it can cross-link mast cell- and basophil-associated IgE, and by activating inhibitory receptor, Fc-γ-RIIb. Low doses of IgG are insufficient to induce IgG-mediated anaphylaxis, presumably because Fc-γ-RIII has much lower affinity than Fc-ε-RI. In comparison, high doses of allergen can precipitate IgG-dependent anaphylaxis by forming complexes that activate macrophages and basophils through Fc-γ-RIII. Another mouse model found evidence of above mechanism in concert with activation of neutrophils resulting from interaction of allergen-specific IgG2 with Fc-γ-RIV on those cells. PAF was predominant mediator in this model also. iii. iv. IgG-dependent anaphylaxis has not been demonstrated in humans. However, human IgG receptors are capable of activating macrophages and neutrophils to secrete PAF and PAF can activate mast cells in vitro, so PAF potentially may contribute to human anaphylaxis. Additionally, anaphylaxis has been reported to be more severe in individuals who catabolize PAF slowly. Rare individuals have experienced anaphylaxis after receiving therapeutic preparations of IgG anti-IgE antibodies (omalizumab). Omalizumab blocks binding of IgE to Fc-ε-RI receptors and does not bind Fc-ε-RI-associated IgE. These anaphylactic reactions could conceivably be IgG-mediated, with patient's IgE acting as antigen, and IgG of drug acting as causative antibody. IgE-independent anaphylaxis has also been reported in some patients receiving another monoclonal antibody preparation, infliximab. More human data are needed to clarify mechanism underlying these clinical events. v. On basis of previous observations and preliminary studies, one group of investigators has hypothesized that decreased blood neutrophil Fc-γ-RIII expression without increased IL-4R-α expression by T lymphocytes might be used in humans to distinguish IgG- from IgE-dependent anaphylaxis. If observed, decreased neutrophil Fc-γ-RIII expression would be associated with IgG-dependent anaphylaxis, whereas increased IL-4R-α expression would be associated with IgE-dependent events. C. Immune complex/complement-mediated i. Several drugs have been implicated in immediate life-threatening reactions that are clinically similar to anaphylaxis except that drug-specific IgE could not be identified. Activation of complement by immune complexes composed of culprit drug and IgG or other isotypes has been proposed for some of these drugs, such as protamine. D. 4. Other proposed mechanism i. A number of non IgE-mediated mechanisms have been proposed to explain anaphylaxis caused by radiocontrast media (RCM). One of these involves the interaction of RCM with Fc portions of IgE or IgG already bound to mast cell or basophil surface, causing cross-linking and activation. Non-immunologic anaphylaxis A. Anaphylactic reactions to various drugs have revealed potential mechanisms by which mast cells and basophils could be activated without evidence of involvement of IgE, other antibodies, or immune complexes. i. Activation of complement, in absence of immune complex formation, has been ii. iii. proposed to account for reactions to drugs that were solubilized in diluent Cremophor EL, such as older preparations of propofol and paclitaxel. It has been proposed that under physiologic conditions, Cremophor formed large micelles with serum lipids and cholesterol, stimulating complement activation. Some human mast cells express receptors for "anaphylatoxins" C3a and C5a, and release histamine in response to exposure to these complement fragments. Macrophages and basophils also have C3a receptors and can produce PAF in response to their activation. This mechanism has been implicated in peanut-induced anaphylaxis in mice, although significance of this in human anaphylaxis has not been demonstrated. Direct activation of mast cells and/or basophils by vancomycin, leading to histamine release, has been implicated in "red man syndrome." This reaction can involve HoTN and present similarly to anaphylaxis in up to 15% of patients. The mechanism is unknown. Opiate, such as meperidine and codeine, can cause non-immunologic histamine release via direct mast cell degranulation. Mild reactions, such as urticaria, are common, although anaphylactic reactions are occasionally reported. In the past, some allergy specialists used opiates as positive controls in skin testing, because these agents induce characteristic wheal-and-flare response due to direct degranulation of mast cells in skin. iv. v. Cold urticaria is reproducible disorder that is characterized by rapid onset of erythema, pruritus, and edema after exposure to cold (water, air, food/beverage, or other source of cold temperature). In patients with this disorder, systemic cold exposure, as might occur with swimming or total body exposure to cold air, can cause massive release of histamine and other mediators, and lead to HoTN. Some episodes are characterized by presence of abnormal proteins (cryoglobulins or cryofibrinogens), which may agglutinate or precipitate at lower temperatures. However, most instances of cold urticaria/anaphylaxis are idiopathic and lack abnormal circulating protein. Oversulfated chondroitin sulfate (OSCS), compound contaminating worldwide heparin supplies in 2007 to 2008, caused anaphylaxis by directly activating kinin-kallikrein pathway, which generated bradykinin, C3a, and C5a. Anaphylactic reactions consisted of HoTN and abdominal pain, and variably included dyspnea, diarrhea, flushing, and angioedema. However, these reactions consistently lacked urticaria or pruritus. 5. Regulation of mast cell activation in anaphylaxis A. Multiple additional protein motifs, receptors, channels, and molecular signals act at various levels to modulate the reactivity and responsiveness of mast cells. These are discussed separately. CHEMICAL MEDIATORS OF ANAPHYLAXIS 1. The chemical mediators of IgE-mediated anaphylaxis in humans include biologically active 2. products of mast cells, basophils, and eosinophils, as well as serum components of complement, coagulation, and kallikrein-kinin pathways. In addition, cytokines that alter sensitivity of various target cells to these mediators are believed to influence severity of anaphylaxis. Mast cells and basophils A. The degranulation of mast cells and basophils results in systemic release of various biochemical mediators and chemotactic substances. i. Histamine, tryptase, chymase, and heparin, which are preformed substances associated with intracellular granules. ii. Histamine-releasing factor and other cytokines (TNF, IL-4, IL-13). iii. B. C. Newly-generated lipid-derived mediators such as prostaglandin D2, leukotriene B4, PAF, and cysteinyl leukotrienes, LTC4, LTD4, and LTE4. The functions of these mediators specifically in anaphylaxis have not been extensively studied, although available data are reviewed here. A more complete description of mediators, cytokines, and chemokines produced by mast cells is found elsewhere. A mutation of c-kit, tyrosine kinase receptor expressed on membrane surfaces of all mast cells has been associated with anaphylaxis. Subjects with D816V c-kit mutation present with normal numbers of mast cells in BM but abnormal expression of CD25 and symptoms of severe anaphylaxis. D. Histamine i. Localized histamine release in skin causes urticaria. Systemic release of histamine, ii. iii. iv. v. however, causes hemodynamic and CV changes, and is not associated with presence of urticaria. Serum histamine levels correlated with severity and persistence of cardiopulmonary manifestations in studies of human anaphylaxis. The systemic effects of histamine are dose-dependent. Histamine was administered to normal volunteers over 30 minutes at doses ranging from 0.05 to 1.0 μg/kg/min, to determine plasma levels required to elicit symptoms of anaphylaxis. 1. At low plasma levels, histamine was associated with 30% increase in HR. 2. At moderate plasma levels, histamine precipitated flushing and headache. 3. Higher plasma histamine levels elicited 30% increase in pulse pressure (systolic pressure minus diastolic pressure). The actions of histamine in anaphylaxis are mediated by binding to H1 and H2 receptors on target cells. In study above, pretreatment with H1 antihistamines, H2 antihistamines, or both, suggested that both H1 and H2 receptors mediated flushing, HoTN, and headache, whereas H1 receptors alone mediated tachycardia, pruritus, rhinorrhea, and bronchospasm. H3 receptors have been implicated in canine model of anaphylaxis and appear to influence CV responses to norepinephrine, although this has not been studied in human anaphylaxis. Murine models suggest H4 receptors might be involved in chemotaxis and mast cell cytokine release, and they might also help to mediate pruritus. Their role (if any) in human anaphylaxis has not been studied. vi. The specific effects of histamine on CV system are discussed below. E. Tryptase i. Tryptase is protease that is abundant in human mast cells. Tryptase is relatively specific for mast cells, although basophils and myeloid precursors contain small amount. There are different forms of tryptases. Beta tryptase is enzymatically active, concentrated in mast cell secretory granules, and released upon degranulation. ii. Tryptase can activate complement and coagulation pathways, as well as iii. kallikrein-kinin contact system. Potential clinical consequences include HoTN, angioedema, clotting, and clot lysis, with the latter two explaining variable development of DIC in severe anaphylaxis. The route of allergen exposure appears to influence resultant tryptase levels for reasons that have not been fully explained. Specifically, anaphylaxis triggered by ingested food may have minimal or no elevation in serum tryptase. iv. v. In analysis of anaphylaxis fatalities, parenterally-administered triggers (injected medications, insect venoms) were associated with higher serum levels of tryptase and lower levels of antigen-specific IgE, whereas orally-administered allergens were associated with low tryptase levels and comparatively high levels of antigen-specific IgE. This difference in tryptase levels may be related to subtype of mast cell first encountered by culprit antigen. Mast cells that predominate in mucosa of small intestine and lung contain much less tryptase per cell than those in connective tissues. Overall, tryptase levels generally correlate with clinical severity of anaphylaxis, with notable exception of food allergens previously described. Postmortem measurements of serum tryptase may be useful in establishing anaphylaxis as the cause of death. 1. vi. F. The measurement of tryptase in anaphylaxis and differential diagnosis of elevated tryptase level are presented in more detail elsewhere. 2. Technical aspects of collecting and measuring tryptase in postmortem setting are reviewed separately. There is mounting evidence to suggest that closer scrutiny to baseline total tryptase levels might be appropriate, especially in patients who experienced hypotension during anaphylaxis. Most studies have evaluated patients with severe anaphylaxis to insect stings. Higher baseline tryptase concentrations (> 11.4 mcg/L) might indicate mastocytosis or monoclonal mast cell disorder (c-kit mutation) and require bone marrow biopsy and cytogenetic analysis for further evaluation. Platelet-activating factor i. PAF-receptor antagonists are effective in rodent models of anaphylaxis. In contrast, roles of PAF and PAF acetylhydrolase, enzyme that inactivates PAF, are not well-defined in human anaphylaxis, although available data suggest that PAF may be important. PAF receptors have been identified in some subsets of human mast cells. In addition, in prospective study of 41 subjects (ages 15 to 74 years) and 23 non-allergic adult controls, serum PAF levels correlated directly and PAF acetylhydrolase levels correlated inversely with severity of anaphylaxis. In companion retrospective analysis, PAF acetylhydrolase activity was significantly lower in nine individuals who experienced fatal peanut-induced anaphylaxis compared with patients in 5 different control groups. Similarly, PAF levels in this study population of 41 subjects correlated better with severity of acute allergic reactions than did serum levels of histamine or tryptase. G. Nitric oxide i. NO acts as messenger molecule in most human organ systems. Within blood vessel walls it has potent vasodilator and accounts for bioactivity of EDRF. Under normal circumstances, NO participates in homeostatic control of vascular tone and regional BP. NO is also involved in complex interaction of regulatory and counter-regulatory mediators in mast cell activation, and has been implicated in HoTN of anaphylaxis. ii. iii. iv. NO promotes protective responses, such as bronchodilation, coronary artery vasodilation, and decreased histamine release, as evidenced by experiments with NO inhibitors in mice, rabbits, and dogs. However, its net effects in anaphylaxis appear to be detrimental through vascular smooth muscle relaxation and enhanced vascular permeability. The binding of histamine to H1 receptors initiates phospholipase C-dependent calcium mobilization, converting L-arginine to NO through activity of NOS. Various isoforms of NOS have been identified, depending on tissue in which they were first isolated. Constitutively expressed isoforms, ie, eNOS and nNOS, are presumed to produce low amounts of NO for physiologic and/or anti-inflammatory functions. In contrast, iNOS expression is associated with inflammation. Increased expression of iNOS results in overproduction of NO and activation of guanylate cyclase. This mechanism may be responsible for CV morbidity and mortality associated with septic shock and has widely been presumed also to apply to anaphylaxis. However, subsequent studies in knockout mice have demonstrated that anaphylaxis can occur in absence of iNOS, and that in PAF-associated anaphylaxis, eNOS is critical mediator. Similar data in humans are not available, although these murine findings suggest that NOS involvement in anaphylaxis may be more complex than previously thought. Seven case reports describe use of methylene blue for treatment of anaphylactic shock refractory to epinephrine, IVF, vasoconstrictors, and IABP, and one report describes its successful use in normotensive subject with refractory anaphylaxis. Methylene blue may exert its favorable effects by blocking NO-mediated vascular smooth muscle relaxation. However, methylene blue itself is capable of causing anaphylaxis in some subjects. H. Arachidonic acid metabolites i. Arachidonic acid is fatty acid derived from membrane phospholipids that can be metabolized via lipoxygenase and cyclooxygenase pathways to generate proinflammatory mediators, such as leukotrienes, prostaglandins, and PAF. Effects of these AA metabolites include bronchospasm, HoTN, and erythema. 1. LTB4 is chemotactic agent that theoretically may contribute to biphasic and 2. 3. 4. protracted reactions. Overproduction of LTC4 enhances mast cell degranulation. LTD4 and E4 increase microvascular permeability and are potent bronchoconstrictors. PGD2 causes vasodilation, increased vasopermeability, and airway smooth muscle bronchoconstriction in various experimental models. It is also chemotactic for neutrophils and activates eosinophils. I. 3. 4. 5. Modulatory mediator i. Other mediators may have anti-inflammatory and modulatory effects that limit anaphylaxis. As examples, chymase may facilitate conversion of angiotensin I to angiotensin II, theoretically helping to counteract HoTN during anaphylaxis. Heparin opposes complement activation, modulates tryptase activity, and inhibits clotting, plasmin, and kallikrein. Eosinophils A. Eosinophils may be proinflammatory (through release of cytotoxic granule-associated proteins) or anti-inflammatory (through metabolism of vasoactive mediators). A guinea pig anaphylaxis model suggests that eosinophils already present in chronically-inflamed airways may participate in immediate-phase response to allergen exposure, as well as their traditional role in late-phase allergic response. These mechanisms have not been studied in human anaphylaxis. Serum factors and other inflammatory pathways A. During severe episodes of anaphylaxis, there is concomitant activation of complement, coagulation pathways, and kallikrein-kinin contact system. Much of evidence for this was obtained during experimental insect sting challenges. Decreases in C4 and C3 and generation of C3a have been observed in anaphylaxis. Demonstrable evidence for coagulation pathway activation during severe anaphylaxis includes decreases in factor V, factor VIII, and fibrinogen, and fatal DIC in some instances. An analysis of 202 anaphylaxis fatalities over 10-year period in the UK determined that 8% of deaths were attributable to DIC. Successful treatment with tranexamic acid has been reported. B. Decreased high molecular weight kininogen and formation of kallikrein C1 inhibitor and factor XIIa C1 inhibitor complexes indicate contact system activation. Kallikrein activation not only generates bradykinin but also activates factor XII. Factor XII itself can cause clotting and clot lysis via plasmin formation, leading to complement activation. In mouse models of anaphylaxis, PAF appears to be important mediator in development of DIC. Changes in target cells A. The development and severity of anaphylaxis also depend upon responsiveness of cells targeted by these mediators. As example, IL-4 and IL-13 are cytokines important in initial generation of antibody and inflammatory cell responses to anaphylaxis in both mice and humans. In murine anaphylaxis, however, IL-4 also induces 3- to 6-fold increase in responsiveness of target cells to inflammatory and vasoactive mediators, including histamine, cysteinyl leukotrienes, serotonin, and PAF. This action of IL-4 appears to take place through alpha chain of IL-4 receptor, resulting in activation of transcription factor signal transducer and activator of transcription 6 (STAT-6). Comparable mechanisms have not been demonstrated in humans. TEMPORAL COURSE 1. Anaphylaxis is usually characterized by rapid onset of symptoms over period of minutes to 2. hours following exposure to trigger. Factors affecting time course A. The variables that determine temporal course of anaphylaxis are not entirely defined. However, several factors appear to be involved. i. The route through which allergen enters body is one factor in determining rapidity of onset of symptoms. Specifically, injected or intravenously-administered allergens tend to precipitate symptoms in seconds to minutes, while ingested allergens cause symptoms in minutes to 1 hour or two. However, these are generalizations to which exceptions are well-reported. ii. 3. The type of allergen responsible for reaction also affects timing of symptom onset. In IgE-mediated anaphylaxis triggered by protein allergens (the best-characterized type of allergen), symptoms usually begin within 2 hours of trigger exposure. In contrast, IgE-mediated anaphylaxis to carbohydrate allergens, such as those responsible for some anaphylaxis to mammalian meats and to monoclonal drug cetuximab, results in symptoms that typically appears 4 to 6 hours after exposure. B. These factors have been examined in cases of fatal and near-fatal anaphylaxis, and are reviewed in more detail elsewhere. Temporal pattern A. Anaphylaxis symptoms most commonly appear, build, peak, and subside in unimodal manner, although biphasic and protracted anaphylaxis are other recognized patterns of anaphylaxis. The other patterns are mentioned briefly here and reviewed in greater detail separately. B. Biphasic anaphylaxis i. Biphasic anaphylaxis is defined as recurrence of symptoms that develops following apparent resolution of initial anaphylactic event without additional exposure to trigger. Biphasic anaphylaxis occurs up to 20% of anaphylaxis cases and mechanisms underlying recurrence of symptoms is unclear. C. Protracted anaphylaxis i. Protracted anaphylaxis is defined as anaphylactic reaction that lasts for hours, days, or even weeks in extreme cases. ORGAN SYSTEMS IN ANAPHYLAXIS 1. Organ system involvement in anaphylaxis varies from species to species and determines clinical manifestations observed. Factors that determine specific "shock organ" include variations in immune response, location of smooth muscle, and distribution, rate of degradation, and responsiveness to chemical mediators. A. In human, predominant shock organs are heart, vasculature, and lungs, and fatalities are divided between circulatory collapse and respiratory arrest. B. Anaphylaxis in rabbits produces fatal pulmonary artery vasoconstriction with right ventricular failure. C. 2. 3. 4. In guinea pig, there is bronchial smooth muscle constriction, which leads to bronchospasm, hypoxemia, and death. D. The primary shock organ in dog is hepatic venous system, which contracts and produces severe hepatic congestion. Human anaphylaxis was traditionally considered form of distributive shock characterized by profound reduction in venous tone, with similarities to septic shock and toxic shock syndrome. An emerging view, however, is that anaphylaxis has features of hypovolemic shock also, with fluid extravasation causing reduced venous return, as well as depressed myocardial function. The clinical manifestations and diagnosis of anaphylaxis is discussed elsewhere. Cardiovascular system A. The human heart may be profoundly affected during anaphylaxis, independently of effects of pharmacologic agents administered during treatment. One report described two previously healthy patients without apparent underlying heart disease, who developed profound myocardial depression during anaphylaxis. Echocardiography, nuclear imaging, and hemodynamic measurements confirmed myocardial dysfunction. IABP was used to provide hemodynamic support, in addition to standard anaphylaxis treatment. This intervention was required for up to 72 hours because of persistent myocardial depression, even though other clinical signs of anaphylaxis had resolved. Both patients recovered with no subsequent evidence of myocardial dysfunction. B. Anaphylaxis has been associated clinically with myocardial ischemia, as well as conduction defects, including atrial and ventricular arrhythmias and T-wave abnormalities. C. It is unclear whether such changes are related to direct mediator effects on myocardium, exacerbation of preexisting myocardial insufficiency by hemodynamic stress of anaphylaxis, endogenous epinephrine released from adrenal medulla in response to stress, or exogenously-injected epinephrine. D. Effects of mediator i. Histamine, acting at H1 receptors, mediates coronary artery vasoconstriction and possibly vasospasm, and increases vascular permeability. H2 receptors increase ii. iii. atrial and ventricular inotropy, atrial chronotropy, and coronary artery vasodilation, as previously mentioned. The interaction of H1 and H2 receptor stimulation results in decreased diastolic pressure and increased pulse pressure. PAF decreases coronary blood flow, delays AV conduction, and has negative inotropic effects on heart. CGRP, sensory neurotransmitter widely distributed in CV tissues and released during anaphylaxis, may help to counteract coronary artery vasoconstriction during anaphylaxis. CGRP relaxed vascular smooth muscle and had cardioprotective effects in animal models of anaphylaxis. iv. Levels of enzymes involved in bradykinin metabolism, serum ACE, and aminopeptidase P (APP) were measured in 122 patients with peanut and tree nut allergy who presented to regional allergy center with acute allergic reactions after ingestion of these agents. Of these 122, 46 had moderate-to-severe pharyngeal edema, 36 had moderate-to-severe bronchospasm, and the rest lacked these symptoms. Patients clinically deemed to have severe pharyngeal edema had significantly lower serum ACE levels than those with no pharyngeal edema. Multivariate analysis indicated that patients with serum ACE concentrations in the lowest quartile were almost 10 times more likely to have severe pharyngeal edema than those with higher ACE concentrations. However, patients with serum ACE levels in the lowest quartile were no more likely than others to have reduced consciousness, bronchospasm, or urticaria. Serum APP levels did not correlate with clinical severity or show any statistical trends. More studies are needed, but these findings suggest clinical scenario in which some patients who experience angioedema during anaphylaxis might be more resistant to treatment with epinephrine and 2nd-line therapeutic agents (antihistamines, glucocorticoids) commonly recommended for use after epinephrine. E. Responses to fluid shift i. Massive fluid shifts occur during anaphylaxis due to increased vascular permeability. Up to 35% of intravascular volume can shift to extravascular space within 10 minutes during anaphylaxis. ii. F. Compensatory responses include release of endogenous catecholamines, angiotensin II, and endothelins. When adequate, these responses may be life-saving, independent of any medical intervention. Some patients, however, experience abnormal elevations of peripheral vascular resistance (maximal vasoconstriction), yet shock persists due to reduced intravascular volume. Others have decreased SVR despite elevated levels of catecholamines. These differences have important clinical implications, since the latter scenario may respond to treatment with vasoconstrictor agents, while the former is vasoconstrictor-unresponsive and requires large-volume fluid resuscitation. Body posture i. The patient's posture during anaphylaxis may impact clinical outcome. In retrospective review of 10 prehospital anaphylactic fatalities in the UK, 4 of the 10 fatalities were associated with assumption of upright or sitting posture. Postmortem findings were consistent with PEA and "empty ventricle," attributed to inadequate venous return secondary to vasodilation and loss of intravascular volume. This is discussed further elsewhere. G. Bradycardia i. Tachycardia is the most common arrhythmia observed during anaphylaxis and is ii. iii. iv. v. believed to develop in response to HoTN, intravascular depletion, and endogenous catecholamines, as in other forms of shock. However, some patients present with bradycardia or with relative bradycardia (initial tachycardia followed by reduction in HR despite worsening HoTN). This has been reported in setting of experimentally-induced insect sting anaphylaxis, as well as in trauma patients. The etiology of this bradycardia has been studied in animal models of hypovolemia. Two distinct phases of physiologic response are apparent. 1. The initial response to hypovolemia is baroreceptor-mediated increase in cardiac sympathetic drive and concomitant withdrawal of resting vagal drive, which together produce tachycardia and peripheral vasoconstriction. 2. A second phase follows when EAV falls by 20 to 30%, which is characterized by withdrawal of vasoconstrictor drive, relative or absolute bradycardia, increased vasopressin, further catecholamine release as adrenal axis becomes more active, and HoTN. HoTN in this setting is independent of bradycardia, since it persists even if bradycardia is reversed with atropine. Bradycardia has also been observed in porcine anaphylaxis precipitated experimentally by various liposomal medications. In this setting, release of anaphylatoxin, C5a, and adenosine acting via A1 adenosine receptors, are believed responsible. Conduction defects and sympatholytic medications, such as βB, may also produce bradycardia in patients with anaphylaxis. Excessive venous pooling with decreased venous return (also seen in vasodepressor reactions) may activate tension-sensitive sensory receptors in inferoposterior portions of LV, thus resulting in cardioinhibitory (Bezold-Jarisch) reflex that stimulates vagus nerve and causes bradycardia. The implications of relative or absolute bradycardia in human anaphylaxis and hypovolemic shock have not been studied, although one retrospective review of approximately 11,000 trauma patients found that 29% of hypotensive patients were bradycardic and mortality was lower in this group compared with those who were tachycardic, after adjustment for other mortality factors. Thus, there may be a specific compensatory role of bradycardia in these settings. H. Exacerbation of underlying cardiac disease i. The concurrence of acute coronary events and anaphylaxis has been noted, although causal relationship between them is unclear. Mast cells accumulate at sites of coronary atherosclerotic plaques, and mast cell degranulation may promote plaque rupture during both acute myocardial events and anaphylaxis. ii. 5. Coronary artery vasoconstriction and decreased intravascular volume could conceivably also precipitate ACS in patient who already had atherosclerotic cardiovascular disease. PAF induction of platelet aggregation and activation of coagulation pathways might additionally predispose to coronary artery thrombosis. Respiratory system A. Anaphylaxis may have adverse effects on any part of respiratory tract. Upper airway symptoms include sneezing, rhinorrhea, dysphonia, laryngeal edema, laryngeal obstruction, or oropharyngeal angioedema. Lower airway manifestations of anaphylaxis include cough, wheeze, pulmonary hyperinflation, edema, hemorrhage, petechiae, mucus plugging, respiratory failure, or respiratory arrest (table 1). B. In retrospective series of acute nonfatal anaphylaxis, respiratory signs and symptoms were observed in 40 to 60% of subjects, with rhinitis, dyspnea/wheeze, and upper airway angioedema in up to 20, 50, and 60%. C. Similar observations have been made in cases of fatal anaphylaxis. i. One report examined 214 anaphylactic fatalities, among which mode of death could be surmised in 196. Asphyxia was cause of death in approximately 50% (98 cases), with involvement of lower airways (bronchospasm) in 49, upper airway angioedema in 23, and both upper and lower airway involvement in 26. The fatalities from acute bronchospasm during anaphylaxis occurred almost exclusively in those with preexisting asthma. ii. Another postmortem analysis of 23 unselected cases of fatal anaphylaxis 6. determined that 16 of 20 "immediate" deaths (deaths occurring within one hour of symptom onset) were due to upper airway edema. Anaerobic metabolism A. Anaerobic metabolism complicates anaphylaxis. Blood flow to periphery is decreased, to preserve perfusion of central organs, such as brain, heart, and kidneys. B. Preliminary evidence suggests that anaerobic metabolism occurs within peripheral tissues during anaphylaxis, similar to other forms of distributive shock, although mechanism may be distinct. In septic shock, paradigm of distributive shock, HoTN results from decreased SVR. Oxygen consumption by skeletal muscle is impaired despite increased partial pressure of oxygen, leading to anaerobic metabolism. This impairment C. in cellular respiration has been attributed to unregulated inflammatory process called "cytopathic hypoxia". One study compared rats with ovalbumin-induced anaphylaxis to parallel group with severe HoTN induced experimentally by nicardipine. The time course and magnitude of HoTN were similar, and both groups experienced decreased perfusion of skeletal muscle. There were metabolic differences. i. ii. iii. The anaphylactic animals showed greater activation of sympathetic nervous system, with higher plasma catecholamine levels beginning at 20 minutes, which were maintained throughout 60-minute protocol. Plasma epinephrine increased 15-fold and norepinephrine levels increased 10-fold over baseline values in anaphylactic animals. Skeletal muscle blood flow was decreased in both nicardipine- and anaphylaxis-induced hypotensive rats initially, which was followed by further decrease in anaphylaxis group beginning at 20 minutes and persisting for duration of observation period. A higher gradient between plasma and interstitial epinephrine indicated more impaired skeletal muscle blood flow in anaphylactic animals, possibly due to greater skeletal muscle vasoconstriction. iv. The anaphylactic animals experienced more rapid increase in interstitial lactate levels and corresponding decrease in interstitial pyruvate levels, indicating depletion of cellular energy stores. This latter finding was not observed in the rats with nicardipine-induced hypotension. D. These findings suggest that skeletal muscle maintains high rates of oxygen utilization during anaphylaxis compared with other forms of distributive shock, and that this, combined with decreased perfusion, leads rapidly to anaerobic metabolism. This may partly explain why end-organ injury and irreversible shock can develop so quickly. AUTOPSY FINDING 1. 2. Victims of fatal anaphylaxis may show no distinguishing gross pathologic features at autopsy, possibly because anaphylaxis can progress to death so rapidly. A retrospective review included 56 cases of fatal anaphylaxis in which autopsy information was available. Death occurred within 1 hour in 39 cases. This is in keeping with clinical observation that in patients in whom shock develops rapidly, there may be essentially no other physical signs or symptoms. When present however, other findings include upper airway edema and petechial hemorrhages in airway mucosa, mucus plugging and hyperinflation of lungs, and cerebral edema. Cutaneous findings, such as urticaria or angioedema, are present in only a minority of fatal cases. Autopsy findings are described in more detail elsewhere. SUMMARY 1. Anaphylaxis is an acute, potentially lethal, multisystem syndrome resulting from the sudden release of mast cell-, basophil- and macrophage-derived mediators into the circulation. 2. Anaphylaxis can be classified as "immunologic" or "nonimmunologic." Immunologic anaphylaxis includes both IgE-mediated and IgG-mediated reactions (which have not been identified in humans), as well as immune complex/complement-mediated mechanisms. Non-immunologic anaphylaxis is caused by agents or events that induce sudden, massive mast cell or basophil degranulation, without involvement of antibodies. 3. 4. 5. 6. In IgE-mediated anaphylaxis, the activation of mast cells, basophils, and eosinophils results in the release of preformed inflammatory mediators, including histamine, tryptase, chymase, heparin, histamine-releasing factor, and PAF. Cellular activation also stimulates the production of lipid-derived mediators such as prostaglandins and cysteinyl leukotrienes. In humans, predominant shock organs are heart, lung, and vasculature, and fatalities are divided between circulatory collapse and respiratory arrest. A. Anaphylaxis is associated with myocardial depression, arrhythmias, and myocardial ischemia. Contributing factors include direct mediator effects on the myocardium, exacerbation of preexisting myocardial insufficiency by the hemodynamic stress of anaphylaxis, and exogenous or endogenous epinephrine. B. Anaphylaxis may affect any part of the respiratory tract, causing bronchospasm and mucus plugging in the smaller airways, and laryngeal edema and asphyxiation in the upper airway. Asphyxiation typically occurs rapidly after allergen exposure, with death occurring within one hour in many cases. Severe bronchospasm during anaphylaxis characteristically develops in individuals with preexisting asthma. Preliminary evidence suggests that during anaphylaxis, peripheral tissues continue to consume oxygen at relatively high rates, and that this, in combination with peripheral vasoconstriction and decreased perfusion, leads rapidly to anaerobic metabolism and end-organ damage. Victims of fatal anaphylaxis may show no distinguishing gross pathologic features at autopsy, possibly because death can ensue so rapidly. However, when present, findings may include upper airway edema and petechial hemorrhages in airway mucosa, mucus plugging and hyperinflation of lungs, and cerebral edema. Cutaneous findings, such as urticaria or angioedema, are uncommon.