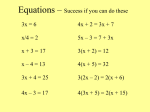* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download NSCC Chem 121 chapter5
Organic chemistry wikipedia , lookup
Acid–base reaction wikipedia , lookup
Inorganic chemistry wikipedia , lookup
Hypervalent molecule wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Determination of equilibrium constants wikipedia , lookup
Nanofluidic circuitry wikipedia , lookup
Asymmetric induction wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Photosynthetic reaction centre wikipedia , lookup
Process chemistry wikipedia , lookup
Metalloprotein wikipedia , lookup
Multi-state modeling of biomolecules wikipedia , lookup
Photoredox catalysis wikipedia , lookup
Marcus theory wikipedia , lookup
Chemical equilibrium wikipedia , lookup
George S. Hammond wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Hydrogen-bond catalysis wikipedia , lookup
Rate equation wikipedia , lookup
Bioorthogonal chemistry wikipedia , lookup
Electrochemistry wikipedia , lookup
Click chemistry wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
Lewis acid catalysis wikipedia , lookup
Transition state theory wikipedia , lookup
Chemical reaction wikipedia , lookup
Spencer L. Seager Michael R. Slabaugh Chapter 5: Chemical Reactions CHEMICAL EQUATIONS • Chemical equations are a convenient way to represent chemical reactions. Chemical equations are written in terms of reactants and products. • A symbol is written in parentheses to the right of each reactant and product to indicate the state or form in which the substance exists. Gases are indicated by (g), liquids by (l), solids by (s), and substances dissolved in water by (aq). solid sugar = C12H22O11 (s) and liquid water = H2O (l) sugar dissolved in water = C12H22O11 (aq) CHEMICAL EQUATIONS (continued) • REACTANTS OF A CHEMICAL EQUATION • The reactants in a chemical equation are the substances written on the left side of the arrow that points toward the products. When two or more reactants are involved in an equation, they are separated by a plus sign (+). Reactants 2 H2 (g) + O2 (g) → 2 H2O (l) Products • PRODUCTS OF A CHEMICAL EQUATION • The products in a chemical equation are the substances written on the right side of the arrow. When two or more products are involved in an equation, they are separated by a plus sign (+). BALANCED CHEMICAL EQUATIONS • A balanced chemical equation is one in which the number of atoms of each element in the reactants is equal to the number of atoms of that same element in the products. • A reaction can be balanced by applying the law of conservation of matter. • Coefficients (in red below) are written to the left of each reactant or product in order to achieve balance. 2 H2 (g) + O2 (g) → 2 H2O (l) EXAMPLES OF UNBALANCED & BALANCED EQUATIONS • Methane, CH4,is the main ingredient in natural gas. It combines with oxygen, O2, when it burns to form carbon dioxide, CO2, and water, H2O. This information written in the form of an unbalanced equation is: CH4(g) + O2 (g) CO2 (g) + H2O(g) • The equation is unbalanced as it is written because the number of H atoms on the left is 4 and the number on the right is 2. Also, the number of O atoms on the left is 2 and the number on the right is 3. EXAMPLES OF UNBALANCED & BALANCED EQUATIONS (continued) • The equation is balanced by inserting appropriate coefficients on the left of each reactant and product to give the following balanced equation: CH4(g) + 2O2 (g) CO2 (g) + 2H2O • When atoms are counted, 1C, 4H and 4O atoms are on the left and the same number are on the right. BALANCED EQUATION PRACTICE • Determine if the following equations are balanced. If not, then add coefficients to balance the equations. • NaOH (aq) + HCl (aq) → NaCl (aq) + H2O (l) • Mg (s) + O2 (g) → MgO (s) BALANCED EQUATION PRACTICE • Determine if the following equations are balanced. If not, then add coefficients to balance the equations. • NaOH (aq) + HCl (aq) → NaCl (aq) + H2O (l) • Balanced (1 Na, 1 O, 2 H, and 1 Cl both sides) • Mg (s) + O2 (g) → MgO (s) • Unbalanced (2 O vs. 1 O) • 2 Mg (s) + O2 (g) → 2 MgO (s) BALANCED EQUATION PRACTICE (continued) • Determine if the following equations are balanced. If not, then add coefficients to balance the equations. • Na2CO3 (s) → Na2O (s) + CO2 (g) • Mg (s) + HCl (aq) → MgCl2 (aq) + H2 (g) BALANCED EQUATION PRACTICE (continued) • Determine if the following equations are balanced. If not, then add coefficients to balance the equations. • Na2CO3 (s) → Na2O (s) + CO2 (g) • Balanced (2 Na, 1 C, and 3 O both sides) • Mg (s) + HCl (aq) → MgCl2 (aq) + H2 (g) • Unbalanced (1 H vs. 2 H and 1 Cl vs. 2 Cl) • Mg (s) + 2 HCl (aq) → MgCl2 (aq) + H2 (g) Problems 5.2, 5.8 TYPES OF CHEMICAL REACTIONS • Chemical reactions are often classified into categories according to characteristics of the reactions. The following is a useful classification scheme: REDOX REACTIONS • The word redox is a combination of two words, reduction and oxidation. These two words have multiple meanings when applied to chemical reactions. • The concept of oxidation numbers provides a convenient way to work with redox reactions. OXIDATION NUMBERS • Oxidation numbers (also called oxidation states) are positive or negative numbers assigned to elements in chemical formulas according to a set of rules. The term oxidation number is abbreviated O.N. • Rule 1: The O.N. of any uncombined element is 0. For example: Fe (0), Cl2 (0), and Ca(0) • Rule 2: The O.N. of a simple ion is equal to the charge on the ion. For example: Mg2+(+2), O2-(-2), and Cl-(-1). OXIDATION NUMBERS (continued) • Rule 3: The O.N. of group IA and IIA elements when they are in compounds are always +1 and +2, respectively. For example: Na2S (Na = +1) and Ca(NO3)2 (Ca = +2) • Rule 4: The O.N. of hydrogen is +1. For example: HBr (H = +1) and H2SO4(H = +1) • Rule 5: The O.N. of oxygen is -2 except in peroxides, where it is -1. For example: MgO (O = -2), HBrO3 (O = -2), and H2O2 (O = -1) A closer look at H2O2 : 2(O.N. of H) + 2(O.N. of O) = 0 2(+1) + 2(O.N. of O) = 0 2(O.N. of O) = -2 (O.N. of O) = -1 A closer look at MgO : (O.N. of Mg) + (O.N. of O) = 0 +2 + (O.N. of O) = 0 (O.N. of O) = -2 OXIDATION NUMBERS (continued) • Rule 6: The algebraic sum of the oxidation numbers of all atoms in a complete compound equals zero. For example: MgSO4 (O.N. of Mg = +2 by rule 2, O.N. of O = -2 by rule 5, and O.N. of S = +6 by algebra and rule 6) A closer look at MgSO4 : (O.N. of Mg) + (O.N. of S) + 4(O.N. of O) = 0 +2 + (O.N. of S) + 4(-2) = 0 +2 + (O.N. of S) + -8 = 0 (O.N. of S) = +6 Because there is only one Mg and one S, the total positive oxidation number is +8. The four O atoms, with an O.N. of -2 each, give a total negative O.N. of -8. Thus, the total positive and the total negative O.N. values add up to zero. OXIDATION NUMBERS (continued) • Rule 7: The algebraic sum of the O.N. of all the atoms in a polyatomic ion is equal to the charge on the ion. For example: HCO3- (O.N. of H is +1 by rule 4, O.N. of O is -2 by rule 5, and O.N. of C is +4 by algebra and rule 7) A closer look at HCO3- : (O.N. of H) + (O.N. of C) + 3(O.N. of O) = -1 +1 + (O.N. of C) + 3(-2) = -1 +1 + (O.N. of C) + -6 = -1 (O.N. of S) = +4 Because there is only one H and one C, the total positive oxidation number is +5. The three O atoms, with an O.N. of -2 each, give a total negative O.N. of -6. Thus, the total positive and the total negative O.N. values add up to -1, which is the charge on the bicarbonate ion. Problems 5.9, 5.12 OXIDIZING & REDUCING AGENTS • In a redox reaction, the substance that contains an element that is oxidized during the reaction is called the reducing agent. • In a redox reaction, the substance that contains an element that is reduced during the reaction is called the oxidizing agent. OXIDIZING & REDUCING AGENTS EXAMPLE • Determine the oxidizing and reducing agents in the reaction: 2Na(s) +2H2O(l) H2 (g) + 2NaOH(aq) OXIDIZING & REDUCING AGENTS EXAMPLE • Determine the oxidizing and reducing agents in the reaction: 2Na(s) +2H2O(l) H2 (g) + 2NaOH(aq) • Solution: First, assign oxidation numbers. OXIDIZING & REDUCING AGENTS EXAMPLE (continued) 2Na(s) +2H2O(l) H2 (g) + 2NaOH(aq) OXIDIZING & REDUCING AGENTS EXAMPLE (continued) 2Na(s) +2H2O(l) H2 (g) + 2NaOH(aq) • Now, determine the oxidizing and reducing agents. • Na is oxidized; therefore, Na is the reducing agent. • H is reduced; therefore, H2O is the oxidizing agent. Problems 5.13, 5.14, 5.16, 5.18 DECOMPOSITION REACTIONS • In decomposition reactions, one substance is broken down into two or more simpler substances. Decomposition reactions may be either redox or nonredox reactions. • The general form of the equation for a decomposition reaction is: A B + C. • An example of a redox decomposition reaction is: 2HI(g) H2 (g) + I2 (g) • An example of a nonredox decomposition reaction is: H2CO3(aq) CO2(g) + H2O(l) DECOMPOSITION REACTIONS (continued) • A pictorial representation of a decomposition reaction: 2 H2O (l) → 2 H2 (g) + O2 (g) COMBINATION REACTIONS • In combination reactions two or more substances react to form a single substance. Combination reactions may be either redox or nonredox reactions. • The general form of the equation for a combination reaction is: A+B C • An example of a redox combination reaction is: S(s) + O2 (g) SO2 (g) • An example of a nonredox combination reaction is: N2O5(g) + H2O(l) 2HNO3(aq) COMBINATION REACTIONS (continued) • A pictorial representation of a combination reaction: 2 Al (s) + 3 Br2 (l) → 2 AlBr3 (s) SINGLE-REPLACEMENT REACTIONS • Single-replacement reactions are always redox reactions because they occur when one element reacts with a compound, displaces one of the elements from the compound, and becomes part of a new compound. • The general form of the equation for a single replacement reaction is: A + BX B + AX In this equation, A and B represent elements and AX and BX are compounds. • An example of a single replacement reaction is: Zn(s) + CuSO4(aq) Cu(s) + ZnSO4(aq) SINGLE-REPLACEMENT REACTIONS (continued) • A pictorial representation of a single replacement reaction: Cu (s) + 2 AgNO3 (aq)→ 2 Ag (s) + Cu(NO3)2 (aq) DOUBLE-REPLACEMENT REACTIONS • Double-replacement reactions are never redox reactions. These reactions often take place between substances dissolved in water. In typical reactions, two dissolved compounds react and exchange partners to form two new compounds. • The following general form of the equation for double replacement reactions shows the partner-swapping characteristic of the reactions: AX + BY BX + AY • An example of a double-replacement reaction is: Ba(NO3)2(aq) + Na2S(aq) BaS(s) +2NaNO3(aq) DOUBLE-REPLACEMENT REACTIONS (continued) • A pictorial representation of a double-replacement reaction: NaCl (aq) + AgNO3 (aq)→NaNO3 (aq) + AgCl (s) REACTION CLASSIFICATION • Classify each of the following equations as redox or nonredox reactions and as combination, decomposition, single-replacement, or double-replacement reactions. • NaOH (aq) + HCl (aq) → NaCl (aq) + H2O (l) • 2 Mg (s) + O2 (g) → 2 MgO (s) REACTION CLASSIFICATION • Classify each of the following equations as redox or nonredox reactions and as combination, decomposition, single-replacement, or double-replacement reactions. • NaOH (aq) + HCl (aq) → NaCl (aq) + H2O (l) • Nonredox reaction; double-replacement reaction • 2 Mg (s) + O2 (g) → 2 MgO (s) • Redox reaction; combination REACTION CLASSIFICATION (continued) • Classify each of the following equations as redox or nonredox reactions and as combination, decomposition, single-replacement, or double-replacement reactions. • Na2CO3 (s)→ Na2O (s) + CO2 (g) • Mg (s) + 2 HCl (aq) → MgCl2 (aq) + H2 (g) REACTION CLASSIFICATION (continued) • Classify each of the following equations as redox or nonredox reactions and as combination, decomposition, single-replacement, or double-replacement reactions. • Na2CO3 (s)→ Na2O (s) + CO2 (g) • Nonredox reaction; decomposition • Mg (s) + 2 HCl (aq) → MgCl2 (aq) + H2 (g) • Redox reaction; single-replacement reaction Problems 5.20, 5.22 IONIC EQUATIONS • Many reactions take place between compounds or elements that are dissolved in water. Ionic compounds and some polar covalent compounds break apart (dissociate) when they dissolve in water and form ions. • The equations for reactions that occur between dissolved materials can be written in three ways called molecular equations, total ionic equations, and net ionic equations. IONIC EQUATIONS (continued) • MOLECULAR EQUATIONS • In a molecular equation, each compound is represented by its formula. • TOTAL IONIC EQUATIONS • In a total ionic equation, all soluble ionic substances are represented by the ions they form in solution. Substances that do not dissolve or that dissolve but do not dissociate into ions are represented by their formulas. NaCl (aq) = Na+ (aq) + Cl− (aq) Na2S (aq) = 2 Na+ (aq) + S2− (aq) Na 3PO4 (aq) = 3 Na+ (aq) + PO43− (aq) IONIC EQUATIONS (continued) • NET IONIC EQUATIONS • In a net ionic equation, only unionized or insoluble materials and ions that undergo changes as the reaction proceeds are represented. • Any ions that appeared on both the left and right side of the total ionic equation are called spectator ions and are not included in the net ionic equation. N aCl (aq) + AgN O3 (aq) → AgCl (s) + N aN O3 (aq) N a+ (aq) + Cl− (aq) + Ag + (aq) + N O3− (aq) → AgCl (s) + N a+ (aq) + N O3− (aq) Cl− (aq) + Ag + (aq) → AgCl (s) IONIC EQUATIONS EXAMPLE • Write the following molecular equation in total ionic and net ionic forms. Soluble substances are indicated by (aq) after their formulas and insoluble solids are indicated by (s) after their formulas. BaCl2 (aq) + Na2S(aq) BaS(s) + 2NaCl(aq) • In total ionic form, all substances except the insoluble BaS will be written in the form of the ions they form: Ba2+(aq) + 2Cl-(aq) BaS(s) + 2Na+(aq) + S2-(aq) + 2Na+(aq) + 2Cl-(aq) • In net ionic form, all spectator ions are dropped. Both the Na+ and Cl- ions are spectator ions because they appear on both sides of the equation. The net ionic equation is: Ba2+(aq) + S2-(aq) BaS(s) Problems 5.28, 5.30, 5.33 ENERGY AND REACTIONS • In addition to changes in chemical composition, all chemical reactions are also accompanied by changes in energy. That is, all reactions either absorb or give up energy as they proceed. • The energy involved in chemical reactions can take numerous forms such as the electrical energy released by the chemical reactions of an ordinary cell phone battery. Often, all or most of the energy takes the form of heat. ENERGY AND REACTIONS • Chemical reactions that release heat as a product are called exothermic reactions. Ordinary combustion of a log in a fireplace is an example of an exothermic reaction. • While it is a physical process and not chemical, a familiar example of an endothermic process is the melting of ordinary ice. As the ice melts, heat is absorbed from the air surrounding the ice. Problems 5.34 THE MOLE AND CHEMICAL EQUATIONS • The mole concept can be applied to balanced chemical equations and used to calculate mass relationships in chemical reactions. • Balanced equations can be interpreted in terms of the mole concept and the results used to provide factors for use in factorunit solutions to numerical problems. THE MOLE AND CHEMICAL EQUATIONS EXAMPLE • Consider the following balanced reaction equation: 2H2S(g) + 3O2(g) 2SO2 (g) + 2H2O(l) • The mole concept can be used to write useful statements that will be the source of factors needed to solve numerical problems. The following are three of the possible statements: 2 mol H2 S + 3 mol O2 → 2 mol SO2 + 2 mol H2 O 2(6.02 × 1023 ) molecs H2 S+ 3(6.022 × 1023 ) molecs O2 68.2 g H2S + 96.0 g O2 2(6.02 × 1023 ) molecs SO2 + 2(6.022 × 1023 ) molecs H2 O 128.2 g SO2 + 36.0 g H2O FACTOR-UNIT METHOD REVIEW • Step 1: Write down the known or given quantity. Include both the numerical value and units of the quantity. • Step 2: Leave some working space and set the known quantity equal to the units of the unknown quantity. • Step 3: Multiply the known quantity by one or more factors, such that the units of the factor cancel the units of the known quantity and generate the units of the unknown quantity. • Step 4: After you generate the desired units, do the necessary arithmetic to produce the final answer. FACTOR-UNIT METHOD EXAMPLE • Calculate the number of moles of H2S that would need to react with excess O2 in order to produce 115 g of SO2. • Solution: Note that the factor used was obtained from the two statements given earlier. • Note that the “g SO2 ” units in the denominator of the factor cancel the “g SO2 ” units of the known quantity, and the “mol H2S” units of the numerator of the factor generate the needed “mol H2S” units of the answer. Problems 5.42, 5.47 THE LIMITING REACTANT • The limiting reactant present in a mixture of reactants is the reactant that will run out first, and thus, it determines the amount of product that can be produced. • A useful approach to solving limiting reactant problems is to calculate the amount of product that could be produced by each of the quantities of reactant that are available. The reactant that gives the least amount of product is then the limiting reactant. THE LIMITING REACTANT EXAMPLE • Calculate the maximum amount of SO2 that could be produced by reacting 55.2 g of O2 with 50.8 g of H2S. • Solution: • The amounts of SO2 that could be produced from 55.2 g of O2 reacting with excess H2S as well as from 50.8 g of H2S reacting with excess O2 will be calculated. • The reactant giving the least amount of SO2 will be the limiting reactant. • The amount of SO2 produced by the limiting reactant is the amount the reaction would produce. THE LIMITING REACTANT EXAMPLE • Calculation for SO2 produced from 55.2 g O2 and excess H2S: • Calculation for SO2 produced from 50.8 g H2S and excess O2: • According to the calculations, the 55.2 g of O2 gives the smallest amount of SO2, so O2 is the limiting reactant and the reaction will produce 73.7 g of SO2. REACTION YIELDS • The amounts of product calculated in the last three examples are not the amounts that would be produced if the reactions were actually done in the laboratory. • In each case, less product would be obtained than was calculated. There are numerous causes. Some materials are lost during transfers from one container to another and side reactions take place that are different from the one that is intended to take place. • The amount of product calculated in the examples is called the theoretical yield. The amount of product actually produced is called the actual yield. These two quantities are used to calculate the percentage yield using the following equation: REACTION YIELDS EXAMPLE • Suppose the mixture of reactants calculated earlier to give 73.7 g SO2 was done in the laboratory and only 42.7 g of SO2 was collected. What is the percentage yield of the reaction? • Solution: Problems 5.52, 5.56, 5.60