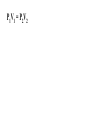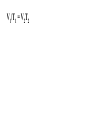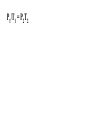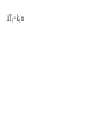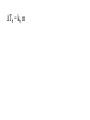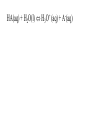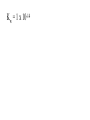* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chapter one
Nanofluidic circuitry wikipedia , lookup
Marcus theory wikipedia , lookup
Degenerate matter wikipedia , lookup
Reaction progress kinetic analysis wikipedia , lookup
Ionic compound wikipedia , lookup
Vapor–liquid equilibrium wikipedia , lookup
Van der Waals equation wikipedia , lookup
Thermodynamics wikipedia , lookup
George S. Hammond wikipedia , lookup
Spinodal decomposition wikipedia , lookup
History of electrochemistry wikipedia , lookup
Electrochemistry wikipedia , lookup
Equation of state wikipedia , lookup
Atomic theory wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
Rate equation wikipedia , lookup
Enzyme catalysis wikipedia , lookup
Ultraviolet–visible spectroscopy wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Electrolysis of water wikipedia , lookup
Chemical thermodynamics wikipedia , lookup
Determination of equilibrium constants wikipedia , lookup
Stability constants of complexes wikipedia , lookup
Acid dissociation constant wikipedia , lookup
Transition state theory wikipedia , lookup
Acid–base reaction wikipedia , lookup
Chemical equilibrium wikipedia , lookup
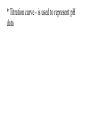
Chemistry - Final Review Guide REMEMBER YOUR CALCULATOR on the day of the exam. Also bring a sharpened pencils and eraser. Review topics and readings for the exam: Chemical Reactions Types of reactions Composition Decomposition Single- replacement Double- replacement Neutralization Balancing reactions Solubility Rules: Ions of metals from group IA and ammonium ions are soluble Chloride ions are soluble with the exception of silver, mercury and lead Sulfate are soluble with the exception of metals from group IIA and lead Chlorate are soluble All others are insoluble (ppt) Activity series rules: 1. Metals from the top of the series would displace any metal below it 2. Metals from group IA and IIA (Ca, Sr and Ba) would react with water, Mg reacts with steam 3. Metals above hydrogen would displace it from hydrochloric and sulfuric acid Chemical composition 1. Mole Calculations: Mol = mass/Mw Mol = (# particles) / (6.02 x 10^23) Mol = V in L/ 22.4 at STP Mol = M x V in L 2 Calculations for molecular mass Example: CaCO3 Mw = 40 + 12 + 3(16) =100g/mol Percent composition Example: K2SO4 Find % K %K = (2(Aw K)/ Mw) x100 Empirical formula and Molecular formula Stoichiometry Balancing Chemical equations Interpreting the coefficient in the balance equation Mole-mole calculations Mole-mass calculations Mass-mass calculations Limiting reactants *Limiting reactant: the amount of reactant that will limit the amount of product *Theoretical Yield: the amount of product calculated from the balance equation *Actual (expected) Yield: the amount of product produce in the experiment * % yield= actual yield/theoretical yield x100 IV. Solids, liquids and gases 1. KMT - five postulates a) Gas particles are far apart and can be compressed, because there are a lot of empty spaces b) Gas particles are constantly moving and exerting pressure on the side of the container c) Vgas = Vcontainer (particles occupy only small portion of the volume) d) Gas particles don’t stick to each other when they collide e) KE = 3/2RT 2. Distribution change / temperature graph 3. Physical properties of gases - Expansion, Fluidity, Low density, Diffusion and effusion 4. Gas pressure (Force/ area) 5. Measuring the gas pressure- barometer and manometer 6 Physical equation for water - heating curve KMT: shape, volume, distance between the particles and forces between the molecules (dipol-dipol, London-dispersion and hydrogen bonding) 8. Vapor pressure a) Difference between boiling and evaporation b) factors affecting ability of a liquid to evaporate (increases of surface area, temperature and decrease of pressure) c) Factors affecting the rate of evaporation of two different liquids (particle attraction and mass of particles) V. Gas laws: 1. Boyles’s Law - T = const P1V1 = P2V2 2. Charles’s Law - p = const V1/T1 = V2T2 3. Gay-Lussac’s Law - V = const P1/T1 = P2T2 4. Combined Law: P1V1/T1 = P2V2/T2 5. Avogadro’s Law: V1/n1 = V2/n2 6. Ideal gas Law: PV = nRT (R = 0.08205 atmL/molK) 7. Dalton’s Law: P total = Pgas + Pwater vapor for collecting gas by water displacement 8. Calculations for molecular weight: Mw = DRT/P 9. Real gases - at high p and lower temperature a) Molecules occupy volume b) There are attractive forces between the molecules c) Van der Waal’s equation: (P + n2a /V2) ( V-nb) = nRT 10. Phase diagram: triple point, determining boiling and freezing point VI. Solutions 1. Solution - a homogeneous mixture of two or more substances in a single physical state. a) Solute - substance that is dissolved in a solution (the less quantity). b) Solvent- substance that does the dissolving in a solution (the more quantity). 2. Types of solution - gaseous, liquids and solids (review the table from the notes). * Explore 15-1: Solutions of Gases, liquids and solids - in this activity you investigated how substances of different physical states can be mixed to produce a solution. * The nature of solutions 3. Solubility. Factors affecting the rate of solution. * Lab: Preparing solutions (heating, stirring and grinding) * Factors effecting the process of dissolving of liquids (“Like dissolve like” rule), solids and gases (temperature and pressure) * Henry’s Law - The bends (15-3 activity worksheet) 4. Saturated, unsaturated and supersaturated solutions a) Saturated- contains as much solute as can possibly be dissolved under existing conditions ( T and p). b) Supersaturated - a solution that contains more solute particles then are needed to form a saturated solution. c) Unsaturated - a solution that has less then the maximum amount of solute that can be dissolved. 5. Concentration units: a) Mass % = mass solute/mass solution x 100 b) Molarity (M) = mol of solute / volume of solution in L c) Molality (m) = mol of solute / kg of solvent d) Mol fraction = mola/moltotal * See practice problems 15-2 * 15-2 Cooking divinity worksheet 6. Colligative properties a) Freezing point depression Tf = kf m b) Boiling point elevation Tb = kb m * See: Practice problems 15-4 * 15-4 Explore - Colder then ice water - in this activity you have discovered what happens to the freezing point of water when a substance is dissolved VII. Equilibrium 1. The concept of equilibrium In chemistry - most chemical reactions are reversible processes. (Activity with the beads and Equal rate - !6.1 explore - investigate a reversible reaction.) a. When the rate of forward reaction is = to the rate of the reverse reaction - chemical equilibrium is established. b. Under the same condition, at equilibrium the concentration of both reactants and products remain constant. c. At the same temperature the equilibrium constant is constant d. Equilibrium may be approached from different starting points. e. At other temperature the value of the equilibrium constant differs. 2. The law of equilibrium (16.2 Review and reinforcement - worksheet) a) Equilibrium constant Keq and the reaction quotient Qeq. b) Collision theory -the molecules must collide in order to react. In successful collision the existing bonds in a molecule are broken and new bonds are formed. c) Factors affecting the rate of a chemical reaction * Collision frequency (depends of concentration of the molecules and temperature). * Collision energy * Orientation of the molecules 3. Reaction profile- shows the energy of reactants and products during reaction * Transition state - is the highest point on the reaction profile where reactant and products have the same potential energy. * Activation energy - the energy require for the reaction to achieve the transition state a) Energy profile for exothermic reaction b) Energy profile for endothermic reaction 4. Types of chemical equilibrium a) Homogeneous b) Heterogeneous 5. Le Chatelier’s principle - when a dynamic equilibrium is upset by disturbance, the equilibrium will shift in a direction to minimize the effect of disturbance (See 16-3 Review and reinforcement). a. Effect of concentration b. Effect of temperature c. Effect of pressure d. Effect of catalyst e. Shifting the equilibrium - the Haber process 6. Solubility equilibrium - Ksp ( see 17.1 Problems) sp concentration of its ions in a saturated solution, each raised to the power that is the coefficient of that ion in the chemical equation. VIII. Acids and bases 1. Properties of acid and bases * Acid -is any substance that produces hydrogen ions in water - changes blue litmus paper to red - Sour taste - pH 7 * Base- is any substance that produces OHions in water solution - changes red litmus paper to blue - Bitter taste - pH 7 - feel slippery or soapy to the touch Both acids and bases can undergo a neutralization reaction. Neutralization reaction is a reaction between acid and base where the products are salt and water. 2. Arrhenius definition * Acid - is a substance that ionizes in water to produce H+ * Base - is a substance that dissociate in water to release OH- ions 3. Bronsted - Lowry definition * Acid - is a substance that donates hydrogen ions to any other substance (proton donor). * Base - is any substance that accept a hydrogen ion (proton acceptor). 4. Acids and bases strength * Strong acid will ionize completely. (HCl, H2SO4, HNO3, HClO4) * Weak acid will ionize slightly. (CH3COOH, HNO2, HCN, H3PO4, H2CO3) HA(aq) + H2O(l) H3O+ (aq) + A-(aq) Ka = H3O+ A- HA * Strong base will dissociate completely ( MeIAOH ). * Weak base will provide relatively few ions in solution B (aq) + H2O (l) BH+ (aq) + OH- (aq) Kb = BH+OH-B 5. Acid- base titration a) Principles of titration’s measurement of the amount of a solution of known concentration that is required to react completely with a measured amount of a solution of unknown concentration. * Standard solution - a solution that contains a precisely known concentration of a solute * Titration curve - is used to represent pH data - Strong acid with the strong base (upward graph - end point at pH =7). - Weak acid with a strong base (upward graph - end point at pH = 8.9) - Weak base with strong acid (downward graph - end point at pH = 5.8) * Equivalent point - is the point at which exactly enough standard solution is added to neutralize the unknown solution. * End point - the point at which the indicator changes color. b) Indicators - are weak acids or bases dyes whose colors are sensitive to pH or hydronium ion concentration. 6. Ionization of water - ionization constant Kw = 1 x 10-14 if H3O+OH-- neutral if H3O+OH- - acidic if H3O+OH- - basic pH concept pH =- log H- pOH = - log OH- 8. Buffers - are solutions that resist changes in pH when an acid or a base is added * It is composed of an aqueous solution of a weak acid and one of its salts