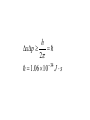* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Bohr´s Third Postulate
Interpretations of quantum mechanics wikipedia , lookup
Quantum state wikipedia , lookup
Symmetry in quantum mechanics wikipedia , lookup
Bremsstrahlung wikipedia , lookup
Renormalization wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Tight binding wikipedia , lookup
Hydrogen atom wikipedia , lookup
Coherent states wikipedia , lookup
Ensemble interpretation wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Delayed choice quantum eraser wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Hidden variable theory wikipedia , lookup
EPR paradox wikipedia , lookup
Identical particles wikipedia , lookup
Introduction to gauge theory wikipedia , lookup
Particle in a box wikipedia , lookup
Atomic orbital wikipedia , lookup
Electron configuration wikipedia , lookup
Elementary particle wikipedia , lookup
Probability amplitude wikipedia , lookup
Wave function wikipedia , lookup
Copenhagen interpretation wikipedia , lookup
Wheeler's delayed choice experiment wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Bohr–Einstein debates wikipedia , lookup
Double-slit experiment wikipedia , lookup
Atomic theory wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
Bohr’s Third Postulate A single photon is emitted whenever an electron jumps down from one orbit to another. hf Eupper Elower 1. Why do astronomers often use the terms color and temperature interchangeably when referring to stars? 2. Why did Bohr assume that the electrons do not radiate when they are in the allowed orbits? Wave Nature of Matter De Broglie Wavelength h mv Wave-particle duality apply also to matter. De Broglie’s Hypothesis Applied to Atoms Quantum Mechanics of Atoms The Wave Function and Its Interpolation In quantum mechanics the amplitude of a particle wave is called the wave function and is given the symbol Y. If we are dealing only with one photon: At any point the square of the electric field strength is a measure of the probability that a photon will be at that location. For a single particle: Y2 at a certain point in space and time represents the probability of finding the electron at the given position and time. Important: there is no way to predict where one electron would hit the screen. We could predict only probabilities. The main point of this discussion is this: if we treat electrons as if they were waves, then Y represents the wave amplitude. If we treat them as particles, then we must treat them on probabilistic basis. The Heisenberg Uncertainty Principle The act of observing produces a significant uncertainty in either the position or the momentum of electron. h xp 2 34 1.06 10 J s Position uncertainty of a baseball What is the uncertainty in position, imposed by the uncertainty principle, of a 150-g baseball thrown at 42+-1 m/s? Should the umpire be concerned? Can he use Heisenberg as an excuse? Particle in a Box Baby-Quiz 1. If all objects emit radiation, why don’t we see most of them in the dark? 2. Suppose you were a nineteenth-century scientist who had just discovered a new phenomenon known as Zeta rays. What experiment could you perform to define if Zeta rays are charged particles or e/m waves? Could this experiment distinguish between neutral particles and an e/m wave? 3. If a metal surface is illuminated by light at a single frequency, why don’t all the photoelectrons have the same kinetic energy when they leave the metal’s surface? 4. What property of the emitted electrons depends on the intensity of incident light?What property of the emitted photoelectrons depends on the frequency of incident light?