* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Document
Magnetoreception wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Particle in a box wikipedia , lookup
Matter wave wikipedia , lookup
Nitrogen-vacancy center wikipedia , lookup
Tight binding wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Electron scattering wikipedia , lookup
Wave–particle duality wikipedia , lookup
Symmetry in quantum mechanics wikipedia , lookup
Spin (physics) wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Chemical bond wikipedia , lookup
Atomic orbital wikipedia , lookup
Population inversion wikipedia , lookup
Hydrogen atom wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
Electron configuration wikipedia , lookup
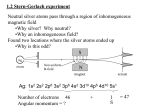
Chapter 40 All About Atoms In this chapter we continue with a primary goal of physics―discovering and understanding the properties of atoms. 100 years ago researchers struggled to find experiments that would prove the existence of atoms. Today, thanks to scientific and technological progress, we can manipulate atoms in amazing ways: we can image individual atoms using scanning tunneling microscopy; we can drag them on surfaces to make quantum corrals, and even hold an individual atom indefinitely in a trap in order to study its properties when isolated. (40-1) 40-2 Some Properties of Atoms Basic Properties Atoms are stable. Essentially all atoms have remained unchanged for billions of years. Atoms combine with each other. Atoms stick together to form molecules and stack up to form rigid solids. Even though atoms are mostly empty space, their interactions allow you to stand on a floor without falling through! These basic properties can be explained by quantum mechanics. (40-2) Some Properties of Atoms Subtler Properties Atoms Are Put Together Systematically. There are repetitive (periodic) patterns in the properties of different atoms that allow them to be organized into a periodic table. Six periods with 2, 8, 8, 18, 18, and 32 atoms in each period, respectively. These numbers are predicted by quantum mechanics. Fig. 40-2 Ionization energy vs. atomic number (number of protons in nucleus) (40-3) Some Properties of Atoms Subtler Properties, cont’d Atoms Emit and Absorb Light: hf Ehigh Elow Atoms Have Angular Momentum and Magnetism: “Orbit” of each electron (more correct to think in terms of angular momentum of electronic state) can produce a magnetic moment. Fig. 40-3 (40-4) Some Properties of Atoms Subtler Properties, cont’d Einstein-de Haas Experiment: Angular momentum and magnetic moment of atoms are coupled. Aligning magnetic moments of iron atoms using an external magnetic field causes the iron cylinder to rotate in a direction opposite to the now-aligned angular momenta of the iron atoms (conservation of angular momentum). Fig. 40-4 (40-5) 40-3 Electron Spin Trapped or free, electrons have intrinsic spin angular momentum S (spin). This is a basic characteristic like the electron’s mass or charge. This leads to two additional quantum numbers that are required to fully specify the electronic state: s (magnitude of the spin, which is always ½ for electrons) and ms (the component of spin along the z-axis). Table 40-1 Electron States for an Atom Quantum Number Symbol Allowed Value Related to Principal n 1, 2, 3, … Distance from nucleus Orbital l 0, 1, 2, …, (n-1) Orbital angular momentum Orbital magnetic ml -l, -(l-1), …+(l-1), +l Orb. ang. mom. (z-component) Spin s ½ Spin angular momentum Spin magnetic ms ±½ Spin ang. mom. (z-component) States with same n form a shell. States with same value for n and l form a subshell. (40-6) 40-4 Angular Momenta and Magnetic Dipole Moments Orbital Angular Momentum and Magnetism Orbital Angular Momentum: L Orbital Magnetic Dipole Moment: orb 1 e orb L 2m e 1 2m Neither orb nor L can be measured experimentally, but their components along a given axis can be measured. Applying a magnetic field B along the z axis allows the z components of orb and L to be measured. (40-7) Orbital Angular Momentum and Magnetic Dipole Moments orb,z m B Bohr magneton eh e B 4 m 2m 9.274 1024 J/T (Bohr magneton) Lz m : semi-classical angle between L and the z axis Fig. 40-5 Lz cos L (40-8) Spin Angular Momentum and Spin Magnetic Dipole Moment S, the magnitude of the spin angular momentum, has a single value for any electron, whether free or trapped: Fig. 40-6 S s s 1 12 12 1 0.866 where s (=½) is spin quantum number of the electron. The spin magnetic dipole moment s is related to S and is given by: e e s s s 1 s S m m Neither S nor s can be measured, but their components along a given axis (say the z -axis) can be measured. S z ms where ms 12 (spin up) or - 12 (spin down) s , z 2ms B (40-9) Orbital and Spin Angular Momentum Combined J represents the total angular momentum of atoms containing more than one electron. J is the vector sum of all the orbital and spin angular momenta of all the electrons. A neutrally charged atom with atomic number Z will have Z electrons and Z protons. J L1 L2 L3 LZ S1 S 2 S3 SZ Total magnetic dipole moment eff is: eff 1 2 3 Z Fig. 40-7 e e L1 L2 L3 LZ S1 S 2 S3 S Z 2m m since S is weighted more than L, eff is not parallel to J . (40-10) 40-5 Stern-Gerlach Experiment Magnetic Deflecting Force on Silver Atom U B U z B dU dB Fz z dz dz dB is the magnetic field dz gradient along the z -direction. Fig. 40-8 Stronger B z Fz Weaker B The z -projection of determines the direction and magnitude of the deflecting force. (40-11) Stern-Gerlach Experiment, cont’d Experimental surprise Silver atoms Meaning of Experiment: z is quantized with two possible values with opposite signs B when magnet ON Lz is also quantized the same way. The dipole moments of all the electrons in a silver atom vectorially cancel out except for the moment of a single electron. Fig. 40-9 s , z 2 12 B B and s , z 2 12 B B dB dB Fz B and Fz B dz dz (40-12) 40-6 Magnetic Resonance A proton has a spin magnetic moment that is associated with the proton's intrinsic spin angular momentum S . In a magnetic field B, the two spin states of the proton will lead to two orientations of , which in turn will have two different energies since U B. Fig. 40-10 E z B z B 2 z B photon absorption (nuclear magnetic resonance) at hf 2 z B spin flip f radio frequency (40-13) Magnetic Resonance, cont’d The net magnetic field that a proton experiences consists of the vector sum of the externally applied magnetic field Bext and internal fields Bint B Bext Bint absorption when hf 2 z Bext Bint magnetic dipole moments of atoms and nuclei near the proton→ Bint For fixed radio frequency light, when Bext = hf/2mz - Bint→ absorption occurs. Bint is different for protons in different molecules, so the resonance Bext will be different for protons in different molecules (local environment). Fig. 40-11 Resonances provide a fingerprint of what (and where in the case of Magnetic Resonance imaging) different proton-containing molecules are present in the material studied. (40-14) 40-7 Pauli Exclusion Principle No two electrons confined to the same trap (or atom) can have the same set of values for their quantum numbers. 40-8 Multiple Electrons in Rectangular Traps 1. One-dimensional trap. Two quantum numbers n=1, 2, 3… (wavefunction state along L) and ms= +½ or -½. 2. Rectangular corral. Three quantum numbers nx = 1, 2, 3… (wavefunction state along Lx) , ny = 1, 2, 3… (wavefunction state along Ly), and ms= +½ or -½. 3. Rectangular box. Four quantum numbers nx = 1, 2, 3… (wavefunction state along Lx) , ny = 1, 2, 3… (wavefunction state along Ly), nz = 1, 2, 3… (wavefunction state along Lz), and ms= +½ or -½. (40-15) Finding the Total Energy Adding electrons to a rectangular trap: Use energy level diagram. Start at lowest energy level and move up as lower levels become filled. Empty (unoccupied) level Partially filled level Filled levels Fig. 40-13 (40-16) 40-9 Building the Periodic Table Four quantum numbers n, l, ml, and ms identify the quantum states of individual electrons in a multi-electron atom. Subshells are labeled by letters: l= 0 1 2 3 4 5 . . . s p d f g h . . . Example: n = 3, l = 2→ 3d subshell Neon: Z = 10→10 electrons Energy n 3 2 1 l = 0 (s) l = 1 (p) ml = 0 -1 0 +1 __ __ __ __ __ __ __ __ __ l = 2 (d) -2 -1 0 +1 +2 __ __ __ __ __ 1s2 2s2 2p6 (40-17) Building the Periodic Table, cont’d Sodium: Z = 11→11 electrons Energy n 3 2 1 l =0 (s) ml = 0 __ __ __ l =1 (p)l =2 (d) -1 0 +1 -2 -1 0 __ __ __ __ __ __ +1 +2 __ __ __ __ __ 1s2 2s2 2p6 3s1 degenerate Chlorine: Z = 17→17 electrons Energy n 3 2 1 l =0 (s) ml = 0 __ __ __ l =1 (p)l =2 (d) -1 0 +1 -2 -1 0 __ __ __ __ __ __ +1 +2 __ __ __ __ __ 1s2 2s2 2p6 3s2 3p6 For smaller atoms such as these, one can assume that the energy only depends on n. (40-18) Building the Periodic Table , cont’d Iron: Z = 26→26 electrons For atoms with a larger number of electrons, the interactions among the electrons causes shells with the same n but different l to have different energies (degeneracy lifted). 1s2 2s2 2p6 3s2 3p6 3d6 4s2 Due to interactions, it takes less energy to start filling the 4s subshell before completing the filling of the 3d subshell, which can accommodate 10 electrons. (40-19) 40-10 X Rays and Ordering of Elements X rays are short-wavelength (10-10 m), high-energy (~keV ) photons. Photons in the visible range: ~ 10-6 m; ~eV. Useful for probing atoms Fig. 40-15 Fig. 40-14 K 0 hf Independent of target material hc min min hc K0 (cutoff wavelength) (40-20) Characteristic X-Ray Spectrum 1. Energetic electron strikes atom in target, knocks out deep-lying (low n value). If deep-lying electron in n = 1 (K-shell), it leaves a vacancy (hole) behind. 2. Another electron from a higher energy shell in the atom jumps down to the K-shell to fill this hole, emitting an x-ray photon in the process. If the electron that jumps into the hole starts from the n = 2 (L-shell), the emitted radiation is the Ka line. If it jumps from the n = 3 (M-shell), the emitted radiation is the Kb line. The hole left in the n = 2 or n = 3 shells is filled by still higher lying electrons, which relax by emitting lower energy photons (higher lying energy levels are more closely spaced). Fig. 40-16 (40-21) Ordering Elements Moseley (1913) bombarded different elements with x rays. Nuclear charge, not mass, is the critical parameter for ordering elements. Fig. 40-17 (40-22) Ordering Elements, cont’d Accounting for the Moseley Plot me4 1 13.60 eV Energy levels in hydrogen: En 2 2 2 = , for n 1, 2,3, 2 8 0 h n n Approximate effective energy levels in multi-electron atom with Z protons (replace e2 x e2 with e2 x (e(Z - 1))2: 2 En 13.60 eV Z 1 n2 13.60 eV Z 1 13.60 eV Z 1 Ka energy: E E2 E1 2 2 12 2 (10.2 eV) Z 1 2 2 (10.2 eV) Z 1 2 E 2 15 Ka frequency: f 2.46 10 Hz Z 1 15 h 4.14 10 eV s f CZ C where constant C 4.96 10 Hz 7 1 2 (40-23) 40-11 Lasers and Laser Light 1. Laser light is highly monochromatic: Its spread in wavelength is as small as 1 part in 1015. 2. Laser light is highly coherent: Single uninterrupted wave train up to 100 km long. Can interfere one part of beam, with another part that is very far away. 3. Laser light is highly directional: Beam spreads very little. Beam from Earth to Moon only spreads a few meters after traveling 4 x 108 m. 4. Laser light can be sharply focused: Can be focused into very small spot so that all the power is concentrated into a tiny area. Can reach intensities of 1017 W/cm2, compared to 103 W/cm2 for oxyacetylene torch. Lasers have many uses: Small: voice/data transmission over optic fibers, CDs, DVDs, scanners Medium: medical, cutting (from cloth to steel), welding Large: nuclear fusion research, astronomical measurements, military applications (40-24) 40-12 How Lasers Work hf Ex E0 Thermal distribution (Boltzmann): N x N0 e Ex E0 kT Fig. 40-19 To get more stimulated emission than absorption, Nx > N0 → population inversion → not in thermal equilibrium (40-25) Helium-Neon Gas Laser Thermal Equilibrium Population Inversion Fig. 40-20 Fig. 40-21 Fig. 40-22 (40-26)