* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download General Chemistry - Valdosta State University
Renormalization wikipedia , lookup
Quantum dot wikipedia , lookup
Quantum field theory wikipedia , lookup
Quantum fiction wikipedia , lookup
Quantum entanglement wikipedia , lookup
Molecular orbital wikipedia , lookup
Probability amplitude wikipedia , lookup
Path integral formulation wikipedia , lookup
Quantum computing wikipedia , lookup
Bell's theorem wikipedia , lookup
Many-worlds interpretation wikipedia , lookup
Coherent states wikipedia , lookup
Orchestrated objective reduction wikipedia , lookup
Double-slit experiment wikipedia , lookup
Quantum machine learning wikipedia , lookup
Quantum electrodynamics wikipedia , lookup
Quantum teleportation wikipedia , lookup
Quantum group wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Quantum key distribution wikipedia , lookup
History of quantum field theory wikipedia , lookup
Interpretations of quantum mechanics wikipedia , lookup
Tight binding wikipedia , lookup
Symmetry in quantum mechanics wikipedia , lookup
Matter wave wikipedia , lookup
Quantum state wikipedia , lookup
Particle in a box wikipedia , lookup
Copenhagen interpretation wikipedia , lookup
Canonical quantization wikipedia , lookup
EPR paradox wikipedia , lookup
Hidden variable theory wikipedia , lookup
Electron configuration wikipedia , lookup
Theoretical and experimental justification for the Schrödinger equation wikipedia , lookup
Wave–particle duality wikipedia , lookup
Bohr–Einstein debates wikipedia , lookup
Atomic orbital wikipedia , lookup
Atomic theory wikipedia , lookup
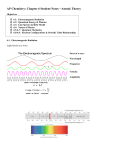
Atomic Structure Chapter 7 Chapter 7 1 Electromagnetic Radiation - Visible light is a small portion of the electromagnetic spectrum Chapter 7 2 Electromagnetic Radiation Frequency (v, nu) – The number of times per second that one complete wavelength passes a given point. Wavelength (l, lambda) – The distance between identical points on successive waves. lv=c c = speed of light, 2.997 x 108 m/s Chapter 7 3 Electromagnetic Radiation - When talking about atomic structure, a special type of wave is important: Standing Wave: A special type of wave with two or more stationary point with no amplitude. Chapter 7 4 Planck, Einstein, Energy and Photons Planck’s Equation - We can also say that light energy is quantized - This is used to explain the light given-off by hot objects. - Max Planck theorized that energy released or absorbed by an atom is in the form of “chunks” of light (quanta). E=hv h = planck’s constant, 6.63 x 10-34J/s - Energy must be in packets of (hv), 2(hv), 3(hv), etc. Chapter 7 5 Planck, Einstein, Energy and Photons The Photoelectric Effect Chapter 7 6 Planck, Einstein, Energy and Photons The Photoelectric Effect - The photoelectric effect provides evidence for the particle nature of light. - It also provides evidence for quantization. - If light shines on the surface of a metal, there is a point at which electrons are ejected from the metal. - The electrons will only be ejected once the threshold frequency is reached . - Below the threshold frequency, no electrons are ejected. - Above the threshold frequency, the number of electrons ejected depend on the intensity of the light. Chapter 7 7 Planck, Einstein, Energy and Photons The Photoelectric Effect - Einstein assumed that light traveled in energy packets called photons. - The energy of one photon, E = hn. Chapter 7 8 Bohr’s Model of the Hydrogen Atom Line Spectra Chapter 7 9 Bohr’s Model of the Hydrogen Atom Line Spectra Chapter 7 10 Bohr’s Model of the Hydrogen Atom Line Spectra Line spectra can be “explained” by the following equation: 1 1 2.179 x10 2 2 l n2 n1 1 18 - this is called the Rydberg equation for hydrogen. Chapter 7 11 Bohr’s Model of the Hydrogen Atom Bohr’s Model - Assumed that a single electron moves around the nucleus in a circular orbit. - The energy of a given electron is assumed to be restricted to a certain value which corresponds to a given orbit. kz E 2 n 2 k = 2.179 x 10-18J z = atomic number n = integer for the orbit Chapter 7 12 Bohr’s Model of the Hydrogen Atom Bohr’s Model - Assumed that a single electron moves around the nucleus in a circular orbit. - The energy of a given electron is assumed to be restricted to a certain value which corresponds to a given orbit. 2 n ao radius z n = integer for the orbit ao = 0.0529 angstroms z = atomic number Chapter 7 13 Bohr’s Model of the Hydrogen Atom Bohr’s Model – Important Features - Quantitized energy and angular momentum - The first orbit in the Bohr model has n = 1 and is closest to the nucleus. - The furthest orbit in the Bohr model has n close to infinity and corresponds to zero energy. - Electrons in the Bohr model can only move between orbits by absorbing and emitting energy in quanta (hn). Chapter 7 14 Bohr’s Model of the Hydrogen Atom Bohr’s Model – Line Spectra Ground State – When an electron is in its lowest energy orbit. Excited State – When an electron gains energy from an outside source and moves to a higher energy orbit. Chapter 7 15 Bohr’s Model of the Hydrogen Atom Bohr’s Model – Line Spectra E(light ) E2 E1 Chapter 7 16 Bohr’s Model of the Hydrogen Atom Bohr’s Model – Line Spectra E (light ) E2 E1 kz2 kz2 2 2 n1 n2 Chapter 7 17 Bohr’s Model of the Hydrogen Atom Bohr’s Model – Line Spectra E (light ) E2 E1 kz kz 2 2 n1 n2 1 2 1 kz 2 2 n2 n1 2 Chapter 7 2 18 Bohr’s Model of the Hydrogen Atom Bohr’s Model – Line Spectra E (light ) E2 E1 kz2 kz2 2 2 n1 n2 1 2 1 kz 2 2 n2 n1 1 1 2.179 x10 2 2 n2 n1 18 Chapter 7 19 Bohr’s Model of the Hydrogen Atom Bohr’s Model - Since the energy states are quantized, the light emitted from excited atoms must be quantized and appear as line spectra. Chapter 7 20 Quantum Mechanical View of the Atom - DeBroglie proposed that there is a wave/particle duality. - Knowing that light has a particle nature, it seems reasonable to assume that matter has a wave nature. - DeBroglie proposed the following equation to describe the relationship: h l mv - The momentum, mv, is a particle property, where as l is a wave property. Chapter 7 21 Quantum Mechanical View of the Atom The Uncertainty Principle Heisenberg’s Uncertainty Principle - on the mass scale of atomic particles, we cannot determine exactly the position, speed, and direction of motion simultaneously. - For electrons, we cannot determine their momentum and position simultaneously. Chapter 7 22 Quantum Mechanical View of the Atom - These theories (wave/particle duality and the uncertainty principle) mean that the Bohr model needs to be refined. Quantum Mechanics Chapter 7 23 Quantum Mechanics Schrödinger’s Model - The path of an electron can no longer be described exactly, now we use the wavefunction(y). Wavefunction (y) – A mathematical expression to describe the shape and energy of an electron in an orbit. - The probability of finding an electron at a point in space is determined by taking the square of the wavefunction: Probability density = y2 Chapter 7 24 Quantum Mechanics Quantum Numbers - The use of wavefunctions generates four quantum numbers. Principal Quantum Number (n) Angular Momentum Quantum Number (l) Magnetic Quantum Number (ml) Spin Quantum Number (ms) Chapter 7 25 Quantum Mechanics Quantum Numbers Principal Quantum Number (n) - This is the same as Bohr’s n - Allowed values: 1, 2, 3, 4, … (integers) - The energy of an orbital increases as n increases - A shell contains orbitals with the same value of n Chapter 7 26 Quantum Mechanics Quantum Numbers Angular Momentum Quantum Number (l) - Allowed values: 0, 1, 2, 3, 4, . , (n – 1) (integers) - Each l represents an orbital type l orbital 0 s 1 p 2 d 3 f Chapter 7 27 Quantum Mechanics Quantum Numbers Angular Momentum Quantum Number (l) - Allowed values: 0, 1, 2, 3, 4, . , (n – 1) (integers) - Each l represents an orbital type - Within a given value of n, types of orbitals have slightly different energy s<p<d<f Chapter 7 28 Quantum Mechanics Quantum Numbers Magnetic Quantum Number (ml). - This quantum number depends on l. - Allowed values: -l +l by integers. - Magnetic quantum number describes the orientation of the orbital in space. l 0 1 Orbital s p 2 d ml 0 -1, 0, +1 -2, -1, Chapter 7 0, +1, +2 29 Quantum Mechanics Quantum Numbers Magnetic Quantum Number (ml). - This quantum number depends on l. - Allowed values: -l +l by integers. - Magnetic quantum number describes the orientation of the orbital in space. - A subshell is a group of orbitals with the same value of n and l. Chapter 7 30 Quantum Mechanics Quantum Numbers Spin Quantum Number (ms) - Allowed values: -½ +½. - Electrons behave as if they are spinning about their own axis. - This spin can be either clockwise or counter clockwise. Chapter 7 31 Quantum Mechanics Quantum Numbers Chapter 7 32 Representation of Orbitals The s Orbitals - All s-orbitals are spherical. - As n increases, the s-orbitals get larger. - As n increases, the number of nodes increase. - A node is a region in space where the probability of finding an electron is zero. Chapter 7 33 Representation of Orbitals The s Orbitals Chapter 7 34 Representation of Orbitals The p Orbitals - There are three p-orbitals px, py, and pz. (The letters correspond to allowed values of ml of -1, 0, and +1.) - The orbitals are dumbbell shaped. Chapter 7 35 Representation of Orbitals The p Orbitals Chapter 7 36 Representation of Orbitals The d and f Orbitals - There are 5 d- and 7 f-orbitals. - Four of the d-orbitals have four lobes each. - One d-orbital has two lobes and a collar. Chapter 7 37 Representation of Orbitals The d and f Orbitals Chapter 7 38 Homework Problems 32, 34, 42 Chapter 7 39