* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download UV Spectroscopy
Homoaromaticity wikipedia , lookup
Vibrational analysis with scanning probe microscopy wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Auger electron spectroscopy wikipedia , lookup
Aromaticity wikipedia , lookup
Spectrum analyzer wikipedia , lookup
Metastable inner-shell molecular state wikipedia , lookup
Molecular Hamiltonian wikipedia , lookup
Gamma spectroscopy wikipedia , lookup
Atomic orbital wikipedia , lookup
Photoacoustic effect wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Photoelectric effect wikipedia , lookup
Transition state theory wikipedia , lookup
Chemical imaging wikipedia , lookup
Heat transfer physics wikipedia , lookup
Thermal radiation wikipedia , lookup
Electron configuration wikipedia , lookup
Chemical bond wikipedia , lookup
Ultrafast laser spectroscopy wikipedia , lookup
Two-dimensional nuclear magnetic resonance spectroscopy wikipedia , lookup
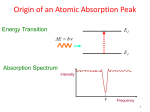
Atomic absorption spectroscopy wikipedia , lookup
Molecular orbital wikipedia , lookup
Mössbauer spectroscopy wikipedia , lookup
Physical organic chemistry wikipedia , lookup
X-ray fluorescence wikipedia , lookup
Franck–Condon principle wikipedia , lookup
Rotational spectroscopy wikipedia , lookup
Rotational–vibrational spectroscopy wikipedia , lookup
Magnetic circular dichroism wikipedia , lookup
Course: B.E & B.Tech Subject: Applied Chemistry Unit: IV SPECTROSCOPY Syllabus: • • • • • • • • Electromagnetic spectrum Absorption of radiation Rotational transition Vibrational transition Electronic transition Intensities of spectral lines Beer Lambert’s law Principle, theory, instrumentation applications of: • Colorimetric analysis • Visible and UV spectroscopy • IR spectroscopy • Flame photometry and Dr. K. SIVAKUMAR Department of Chemistry SCSVMV University [email protected] 1 Electromagnetic Waves - Terminologies Electromagnetic wave parameters: Wavelength (λ): Wavelength is the distance between the consecutive peaks or crests Wavelength is expressed in nanometers (nm) 1nm = 10-9 meters = 1/1000000000 meters 1A = 10-10 meters = 1/10000000000 meters 2 Electromagnetic Waves - Terminologies Electromagnetic wave parameters: Frequency (): Frequency is the number of waves passing through any point per second. Frequency is expressed in Hertz (Hz) 3 Electromagnetic Waves - Terminologies Electromagnetic wave parameters: Wave number ( ): Wave number is the number of waves per cm. Wavelength, Wave number and Frequency are interrelated as, 1 Where, = c is wave length is wave number is frequency c is velocity of light in vacuum. i.e., 3 x 108 m/s 4 Electromagnetic Spectral regions nm EM waves 10-4 to 10-2 g-rays 10-2 to 100 X-rays 100 to 102 102 to 103 103 to 105 UV Visible IR 105 to 107 107 to 109 Microwave Radio 5 Electromagnetic Spectrum E = h h – Planck’s constant 6 www.spectroscopyNOW.com The Electromagnetic wave lengths & Some examples 7 Electromagnetic radiation sources EM radiation Gamma rays X-rays Ultraviolet Visible Infrared Microwave Radio wave Spectral method Gamma spec. X-ray spec. UV spec. Visible spec. IR spec. ESR spec. NMR spec. Radiation source gamma-emitting nuclides Synchrotron Radiation Source (SRS), Betatron (cyclotron) Hydrogen discharge lamp tungsten filament lamp rare-earth oxides rod klystron valve magnet of stable field strength 8 Electromagnetic Spectrum – Type of radiation and Energy change involved 9 Electromagnetic Spectrum – Type of radiation and Energy change involved 10 Electromagnetic Spectrum – Type of radiation and Energy change involved 11 Effect of electromagnetic radiations on chemical substances The absorption spectrum of an atom often contains sharp and clear lines. Absorption spectrum of an atom; Hydrogen Energy levels in atom; Hydrogen 12 Effect of electromagnetic radiations on chemical substances But, the absorption spectrum of a molecule is highly complicated with closely packed lines This is due to the fact that molecules have large number of energy levels and certain amount of energy is required for transition between these energy levels. Energy levels in molecule Absorption spectrum of a molecule; Eg: H2O 13 Lambert’s law fraction of the monochromatic light absorbed by a homogeneous medium is independent of the intensity of the incident light and each successive unit layer absorbs an equal fraction of the light incident on it Lambert Beer’s law fraction of the incident light absorbed is proportional to the number of the absorbing molecules in the light-path and will increase with increasing concentration or sample thickness. Beer 14 Beer–Lambert law / Beer–Lambert– Bouguer law / Lambert – Beer law log (I0/I) = c l = A Where, I0 - the intensity of incident light I - the intensity of transmitted light - molar absorptivity / molar extinction coefficient in cm2 mol-1 or L mol-1 cm-1. c - concentration in mol L-1 l - path length in cm A - absorbance (unitless) Molar absorptivity 15 Absorption intensity max Intensity of absorption is directly proportional to the transition probability A fully allowed transition will have max > 10000 A low transition probability will have max < 1000 max wavelength of light corresponding to maximum absorption is designated as max and can be read directly from the horizontal axis of the spectrum Absorbance (A) is the vertical axis of the spectrum A = log (I0/I) I0 - intensity of the incident light; I - intensity of transmitted light max = 20000 16 Beer–Lambert law / Beer–Lambert– Bouguer law / Lambert – Beer law Bouguer Actually investigated the range of absorption Vs thickness of medium Lambert Extended the concepts developed by Bouguer Beer Applied Lambert’s concept to solutions of different concentrations ? Bernard Beer released the results of Lambert’s concept just prior to those of Bernard 17 Effect of electromagnetic radiations on chemical substances The radiation energies absorbed by molecules may produce Rotational, Vibrational and Electronic transitions. 18 Effect of electromagnetic radiations on chemical substances Rotational transition Microwave and far IR radiations bring about changes in the rotational energies of the molecule Example: Rotating HCl molecule 19 Effect of electromagnetic radiations on chemical substances Vibrational transition Infrared radiations bring about changes in the vibration modes (stretching, contracting and bending) of covalent bonds in a molecule Examples: Example: Vibrating HCl molecule 20 Effect of electromagnetic radiations on chemical substances Electronic transition UV and Visible radiations bring about changes in the electronic transition of a molecule Example: Cl2 in ground and excited states 21 Effect of electromagnetic radiations on chemical substances Cl2 in Ground state 22 Effect of electromagnetic radiations on chemical substances Cl2 in Excited state 23 Spectrometer The instrument used for recording the absorption spectra of a compound is called spectrometer. The different components present in various types of spectrometer are shown here. UV - hydrogen discharge lamp Visible - tungsten filament lamp Variable slit, rheostat, etc Prism, ilter, monochromator, grating Cuvette, test-tube, cell Photographic plate, photocell, photomultiplier, photoconductivity device etc Galvanometer; pen recorder; cathode ray oscillograph IR - electrically heated rod of rareearth oxides Microwave - klystron valve NMR - magnet of stable field strength. 24 Colorimetric analysis Principle: Colorimetry analysis method is useful in determining the concentration of coloured solutions using the visible region (400nm–750nm) of electromagnetic spectrum and Beer Lambert’s law. If the test solution is colourless then a suitable complexing agent can be added to test solution to get coloured which will absorb light. Example: For cuprous ions (Cu2+) estimation NH4OH can be added to get blue colour. Instrumentation: Tungsten filament lamp is used to generate visible region (400nm – 750nm) light. 25 Colorimetric analysis… The molecules in the cuvette absorb light and the remaining light is transmitted to the photocell. In photocell, Current generated Amount of light transmitted But the amount of light transmitted depends on the depth of colour of test solution. i.e., concentration of test solution. If high concentration solution is analysed then, more number of molecules will be in the path of light and more amount of light will be absorbed. So, the amount of light transmitted will be very less and generates only less current. 26 Colorimetric analysis: Applications useful in estimating the concentration of coloured solutions Example: Estimation of CuSO4 by colorimetry Series of CuSO4 solution with known concentration are prepared and ammonium hydroxide is added to each solution to get blue colour. Absorbance of each standard CuSO4 solution is measured with same filter and tabulated. Concentration (C) of CuSO4 Absorbance (A) 0.0001 A1 0.0002 A2 0.0003 . 0.0004 . 0.0005 . 0.0006 A6 test solution At A= C l 27 UV Visible Spectroscopy Principle: Visible and ultraviolet spectroscopy is a study of electronic spectra of organic molecules which are found in the wavelength region of 100nm-400nm (UV region) and 400nm-750nm (Visible region). UV and visible radiations absorbed by the molecules will bring transition of outer shell electrons(, and n electrons). According to molecular orbital theory when a organic molecule absorbs UV or visible radiations its electrons are promoted from a bonding to an antibonding orbital. 28 The Ultraviolet region [10 – 800nm] The Ultraviolet region may be divided as follows, 1. Far (or Vacuum) Ultraviolet region [10 – 200 nm] 2. Near (or Quartz) Ultraviolet region [200 – 380 nm] 3. Visible region [380 - 800 nm] 29 VISIBLE region in Electromagnetic Spectrum •Violet •Indigo •Blue •Green •Yellow •Orange •Red : 380 - 420 nm : 420 - 440 nm : 440 - 490 nm : 490 - 570 nm : 570 - 585 nm : 585 - 620 nm : 620 - 800 nm 30 UV - VISIBLE SPECTROSCOPY • In UV - Visible Spectroscopy, the sample is irradiated with the broad spectrum of the UV - Visible radiation • If a particular electronic transition matches the energy of a certain band of UV - Visible, it will be absorbed • The remaining UV - Visible light passes through the sample and is observed • From this residual radiation a spectrum is obtained with “gaps” at these discrete energies – this is called an absorption spectrum 31 Electronic Energy Levels • Absorption of UV - Visible radiation by an organic molecule leads to electronic excitation among various energy levels within the molecule. • Electron transitions generally occur in between a occupied bonding or lone pair orbital and an unoccupied non-bonding or antibonding orbital. • The energy difference between various energy levels, in most organic molecules, varies from 30 to 150 kcal/mole 32 Electronic Energy Levels * (anti-bonding) * (anti-bonding) Energy n (non-bonding) (bonding) (bonding) - orbitals are the lowest energy occupied molecular orbitals * - orbitals are the highest energy unoccupied molecular orbitals - orbitals are of somewhat higher energy occupied molecular orbitals * - orbitals are lower in energy (unoccupied molecular orbitals) than * n - orbitals; Unshared pairs (electrons) lie at the energy of the original atomic orbital. Most often n - orbitals energy is higher than and . since no bond is formed, there is no benefit in energy 33 Electronic Energy Levels Graphically, * Unoccupied levels * Energy Atomic orbital n Atomic orbital Occupied levels Molecular orbitals 34 Electronic Transitions • The valence electrons in organic molecules are involved in bonding as - bonds, - bonds or present in the non-bonding form (lone pair) • Due to the absorption of UV - Visible radiation by an organic molecule different electronic transitions within the molecule occurs depending upon the nature of bonding. • The wavelength of UV - Visible radiation causing an electronic transition depends on the energy of bonding and antibonding orbitals. • The lowest energy transition is typically that of an electron in the Highest Occupied Molecular Orbital (HOMO) to the Lowest Unoccupied Molecular Orbital (LUMO) * * Energy Atomic orbital n Molecular orbitals Unoccupied levels Atomic orbital Occupied levels 35 Types of Electronic Transitions Transition between bonding molecular orbitals and anti-bonding molecular orbitals They are of three types: * * * 36 Types of Electronic Transitions Transition between bonding molecular orbitals and anti-bonding molecular orbitals * (bonding to anti-bonding ) * (anti-bonding) • * transition requires large energies in far UV region in 120-200nm range. * (anti-bonding) • Molar absorptivity: Low max = 1000 - 10000 n (non-bonding) (bonding) (bonding) • Examples: Alkanes - transition @ 150nm Methane Cyclohexane Propane 37 Types of Electronic Transitions Transition between bonding molecular orbitals and anti-bonding molecular orbitals * (bonding to anti-bonding ) * (anti-bonding) • * occur in 200-700nm range. * (anti-bonding) • Molar absorptivity: High max = 1000 - 10000. n (non-bonding) Examples: •Unsaturated compounds •double or triple bonds •aromatic rings •Carbonyl groups •azo groups •Conjugated electrons Carbonyl Azo (bonding) (bonding) max is high because the and * orbitals are in same plane and consequently the probability of jump of an electron from * orbital is very high. 38 Types of Electronic Transitions Transition between bonding molecular orbitals and anti-bonding molecular orbitals * (bonding to anti-bonding ) • * occur only in <150 nm range. • Molar absorptivity: Low Examples: Carbonyl compounds * (anti-bonding) * (anti-bonding) n (non-bonding) (bonding) (bonding) • * and * transitions: high-energy, accessible in vacuum UV (max <150 nm). Not usually observed in molecular UV-Vis. 39 Types of Electronic Transitions Transition between non-bonding atomic orbitals and anti-bonding molecular orbitals They are of two types: * (anti-bonding) n * n * n * (non-bonding n to anti-bonding ) • n * occur in 200-700nm range. • Molar absorptivity: Low max = 10 - 100 • * (anti-bonding) n (non-bonding) (bonding) (bonding) Examples: • Compounds with double bonds involving unshared pair(s) of electrons • Aldehydes, Ketones • C=O, C=S, N=O etc., 40 Types of Electronic Transitions Transition between non-bonding atomic orbitals and anti-bonding molecular orbitals n * (non-bonding n to anti-bonding ) • * (anti-bonding) * (anti-bonding) Excitation of an electron in an unshared pair on Nitrogen, oxygen, sulphur or halogens to an antibonding orbital is called n * transitions. • n * occur in 150-250nm range. • Molar absorptivity: Low max = 100 - 3000 n (non-bonding) (bonding) (bonding) Example: Methanol max = 183nm ( = 500) max = 257nm ( = 486) Trimethylamine max = 227nm ( = 900) 1-Iodobutane 41 Types of Electronic Transitions * (bonding to anti-bonding ) * (bonding to anti-bonding ) * (bonding to anti-bonding ) * (anti-bonding) * (anti-bonding) n * (non-bonding n to anti-bonding ) n * (non-bonding n to anti-bonding ) n (non-bonding) (bonding) (bonding) Energy required for various transitions obey the order: * > n * > *> n * 42 Chromophores The coloured substances owe their colour to the presence of one or more unsaturated groups responsible for electronic absorption. These groups are called chromophores. Examples: C = C, C=C, C = N, C=N, C = O, N = N, etc.. Chromophores absorb intensely at the short wavelength But, some of them (e.g, carbonyl) have less intense bands at higher wavelength due to the participation of n electrons. Methyl orange 43 Auxochromes An auxochromes is an auxillary group which interact with chromophore and deepens colour; its presence causes a shift in the UV or visible absorption maximum to a longer wavelength Examples: NH2, NHR and NR2, hydroxy and alkoxy groups. Property of an auxochromic group: • Provides additional opportunity for charge delocalization and thus provides smaller energy increments for transition to excited states. • The auxochromic groups have atleast one pair of non-bonding electrons (lone pair) that can interact with the electrons and stabilizes the * state 44 Auxochromes: examples Auxochrome Unsubstitued chromophore max (nm) Substituted chromophore max (nm) -CH3 H2C=CH-CH = CH2 217 H2C=CH-CH=CHCH3 224 -OR H3C-CH=CH-COOH 204 H3C-C(OCH3) = CHCOOH 234 -C1 H2C=CH2 175 H2C = CHCl 185 45 Instrumentation I0 I1 detector monochromator/ beam splitter optics I0 reference UV-VIS sources sample log(I0/I) = A 200 700 , nm I I2 46 Instrumentation… Radiation source, monochromator and detector Two sources are required to scan the entire UV-VIS band: Deuterium lamp – covers the UV – 200-330 Tungsten lamp – covers 330-700 The lamps illuminate the entire band of UV or visible light; the monochromator (grating or prism) gradually changes the small bands of radiation sent to the beam splitter The beam splitter sends a separate band to a cell containing the sample solution and a reference solution The detector (Photomultiplier, photoelectric cells) measures the difference between the transmitted light through the sample (I) vs. the incident light (I0) and sends this information to the recorder 47 Sample Handling Virtually all UV spectra are recorded solution-phase Only quartz is transparent in the full 200-700 nm range; plastic and glass are only suitable for visible spectra 380 – 800nm Concentration: 0.1 to 100mg 10-5 to 10-2 molar concentration may safely be used Percentage of light transmitted: 20% to 65% At high concentrations, amount of light transmitted is low, increasing the possibility of error A typical sample cell (commonly called a cuvet): Cells can be made of plastic, glass or quartz (standard cells are typically 1 cm in path length) 48 UV - VISIBLE SPECTROSCOPY - Applications Structure identification Useful in the identification of a newly synthesized compound. The spectrum of unknown compound can be compared with the absorption spectrum of several known compounds in the literature. On comparison, If the spectrum of unknown compound correlates with a specifically known compound then the structures of both will also be similar. This method is called finger printing technique. Concentration of impurities During the purification process of a compound, if the absorption spectrum is recorded at a particular interval of time the decrease in the concentration of impurities can be monitored. The purification can be continued till it gives a less value of absorbance. To study the rate of a reaction To study the rate of formation of product in a reaction, the absorbance of product can be measured at definite intervals of time. We know that, the absorbance is directly proportional to concentration and hence the absorbance value will be increasing with respect to time. 49 UV Spectroscopy (Electronic Spectra) - Terminologies Beer-Lambert Law Absorbance Molar absorptivity Extinction coefficicent concentration Path length A = .c.l A, a measure of the amount of radiation that is absorbed , absorbance of a sample of molar concentration in 1 cm cell. An alternative term for the molar absorptivity. c, concentration in moles / litre l, the length of the sample cell in cm. max The wavelength at maximum absorbance max The molar absorbance at max Band Term to describe a uv-vis absorption which are typically broad. HOMO Highest Occupied Molecular Orbital LUMO Lowest Unoccupied Molecular Orbital Chromophore Auxochrome Structural unit responsible for the absorption. A group which extends the conjugation of a chromophore by sharing of nonbonding electrons Bathochromic shift The shift of absorption to a longer wavelength. Hypsochromic shift shift of absorption to a shorter wavelength Hyperchromic effect An increase in absorption intensity Hypochromic effect A decrease in absorption intensity Isosbestic point point common to all curves produced in the spectra of a compound taken at various pH 50 Infra RED Spectral region • Infrared refers to that part of the electromagnetic spectrum between the visible and microwave regions. • The IR region is divided into three regions as given below, Few absorptions of organic molecules ‘E’ corresponds to differences observed between vibrational states Very few absorptions occur here 51 Infra RED Spectroscopy – Wave number • • In IR, frequency is expressed as wave numbers Wave numbers have units of reciprocal cm (cm-1) 1 1 Wavenumber in cm = = x 104 in cm in m 1 • • Using this scale, the IR region is 4000-400 cm-1 The corresponding wavelength range is 2.5 m to 25 m 2.5 m = 4000 cm-1 25 m = 400 cm-1 52 Infra RED Spectroscopy – Wave number… Therefore, HIGHER WAVENUMBER LOWER WAVENUMBER Higher frequency Lower frequency Higher energy Lower energy Shorter wavelength Longer wavelength Energy, frequency, and wavenumber are directly proportional to each other. www.spectroscopyNOW.com 53 Infra RED Spectroscopy – An IR spectrum • In IR spectroscopy, an organic molecule is exposed to IR radiation. When the radiant energy matches the energy of a specific molecular vibration, absorption occurs. • Compares intensity of IR striking sample (Iin) with intensity of IR leaving sample (Iout) • 100%T no light absorbed by sample • 0% all light absorbed by sample 54 Infra RED Spectroscopy – An IR spectrum •An IR spectrum is a graph between Wavenumber (in cm-1) Vs Transmittance (%). IR spectrum of octane An absorption of IR energy is therefore represented by a “trough” in the curve. 55 Infra RED Spectroscopy – An IR spectrum… • Band intensities can also be expressed as absorbance; A = log10 (1/T) IR spectrum of octane 56 Infra RED Spectroscopy – Principle • Molecules are flexible. • Atoms in organic molecules are constantly vibrating around average positions. • Therefore, A vibrating bond can be thought of as two masses connected by a spring. • Molecules are vibrating in different modes i.e., different ways. • Bond lengths and bond angles are continuously changing due to this vibration. • These vibrational changes depend on the spatial arrangement and masses of the atoms in that particular molecule. 57 Infra RED Spectroscopy – Principle… • If infrared radiations are passed through the naturally vibrating molecules then IR waves matching with the vibrating frequencies of molecule will be absorbed and increases the amplitude of vibration in molecules. • Because, IR radiations does not have sufficient energy to cause the excitation of electrons, however, it causes atoms and groups of atoms of organic compounds to vibrate faster about the covalent bonds which connect them. • The IR spectrum (vibrational spectra) appears as band and not as line because; the vibrational changes are also accompanied by several numbers of rotational changes. 58 Infra RED Spectroscopy –Functional group & Finger print region • Every molecule absorbs IR waves at particular frequencies and produces absorption bands. • There are two sections in the IR spectra. Functional group region > 1500cm-1 Fingerprint region < 1500cm-1 Fingerprint region: The absorption band in the finger print region is due to the complex vibrations involving in entire molecules. It is impossible for any two different compounds (except enantiomers) to have precisely the same infrared spectrum. The pattern of absorptions in this regions are unique to any particular compound, just as a person’s fingerprints are unique. 59 Infra RED Spectroscopy – Finger print region can be subdivided in to three regins (i) 1500 – 1350cm-1 region: • The absorption bands near 1380cm-1 and 1365cm-1 show the presence of tertiary butyl group in the compound. • Gem-dimethyl shows a medium band near 1380cm-1 • Nitro compounds show one strong band at 1350cm-1 (ii) 1350 – 1000cm-1 region: • Compounds like alcohol, esters, lactones, acid anhydrides show characteristic absorptions in this region, due to the C-H stretching. • Primary alcohols form two strong bands at 1350-1260cm-1 and near 1050cm-1. • Phenols absorb near 1200cm-1 • Esters show two strong bands between 1380-1050cm-1. Absorption bands in the region 1150-1070cm-1 is due to C-O and C-O-C group in ethers. (iii) Below 1000cm-1 region: • The bands at 700cm-1 and at 970-960cm-1 are useful to distinguish between cis and trans alkenes. The higher value indicates that the hydrogen atoms in the alkene are trans w.r.t. each other. • The presence of mono substituted and also disubstituted (at ortho, para and meta positions) benzene shows characteristic absorptions in this region. • Example: A band in the region 750-700cm-1 is due to mono substituted benzene. 60 Infra RED Spectroscopy –Functional group region Functional group region: Like molecules, different functional groups absorb IR waves at particular frequencies and produces absorption bands in the IR spectrum. For example, the IR spectrum of all compounds with carbonyl group consists of absorption band in wave number range 1800cm-1 and 1650cm-1. • 4000-2500 cm-1 N-H, C-H, O-H (stretching) – 3300-3600 cm-1 N-H, O-H – 3000 cm-1 C-H • 2500-2000 cm-1 CC and CN (stretching) • 2000-1500 cm-1 double bonds (stretching) – 1680-1750 cm-1 C=O – 1640-1680 cm-1 C=C • Below 1500 cm-1 “fingerprint” region 61 Infra RED Spectroscopy –Functional group & Finger print region… Functional group region Fingerprint region 62 Infra RED Spectroscopy – IR Active and IR Inactive For a molecule to absorb infra red radiation, the molecule has to fulfill the following two requirements. 1. Correct wavelength of radiation The molecule absorbs IR radiations only when the natural frequency of vibration of atoms or groups of atoms or functional groups of molecule is same as the frequency of the incident radiation. i.e., If infrared radiations are passed through the naturally vibrating molecules then IR waves matching with the vibrating frequencies of molecule will be absorbed and increases the amplitude of vibration in molecules. For example: The natural frequency of HCl is 8.7 1013 sec1 When IR radiation is passed through the HCl and the spectrum is recorded. The spectrum consists of a absorption band at a frequency 8.7 1013 sec1 Hence, 8.7 1013 sec1 is the natural or characteristic frequency of the HCl molecule. 2.886 10 cm 3 1 63 Infra RED Spectroscopy – IR Active and IR Inactive….. 2. Electric dipole For absorbing IR radiations a molecule should possess electric dipole (dipole moment) and the absorption should causes change in electric dipole. Molecules with electric dipole will have slight positive and slight negative charge on its component atoms. For example: In HCl, due to the high electro negativity of Cl, the electron clouds will be more towards chlorine and less towards hydrogen. So, hydrogen will get slight positive charge and Cl will get slight negative charge, i.e., But IR radiation absorption is not possible in the symmetric diatomic molecules like H2, O2, and N2 etc., because, symmetric diatomic molecules do not possess electric dipole. , Therefore, IR radiation absorption and IR spectrum is not possible in these molecules. H Cl dipole moment OO IR Active llly H2O, NO IR Inactive No dipole moment llly H2, Cl2 64 Infra RED Spectroscopy – Theory - Various types of molecular vibrations bond length changes bond angle changes 65 Infra RED Spectroscopy – Theory - Various types of molecular vibrations Stretching: Rhythmical movement along the bond axis, which leads to the increase or decrease of interatomic distance. Symmetrical stretching Anti-symmetrical stretching 66 Infra RED Spectroscopy – Theory - Various types of molecular vibrations Bending: Bending vibrations takes place when a three atom system is a part of a molecule. Types of Bending vibrations: • Scissoring • Wagging • Rocking • Twisting 67 Infra RED Spectroscopy – Theory - Various types of molecular vibrations Scissoring: Two atoms attached to a central atom move away from and towards each other. 68 Infra RED Spectroscopy – Theory - Various types of molecular vibrations Wagging: Vibrating group swings back and forth out of plane of molecule. 69 Infra RED Spectroscopy – Theory - Various types of molecular vibrations Rocking: Vibrating group swings back and forth in the plane of molecule. 70 Infra RED Spectroscopy – Theory - Various types of molecular vibrations Twisting: Vibrating group rotates about the chemical bond which is attached to rest of molecule. 71 Infra RED Spectroscopy – Theory - Various types of molecular vibrations bond length changes bond angle changes 72 Infra RED Spectroscopy – Theory - Various types of molecular vibrations bond length changes bond angle changes 73 Infra RED Spectroscopy – Energy required for molecular vibrations Energy required Stretching bond length changes > Bending bond angle changes Energy needed to stretch a spring is more than that needed to bend it so the stretching absorption of a bond will appear at a higher frequency than the bending absorption of the same bond. Stretching > Bending > Wagging / Twisting 74 Infra RED Spectroscopy – Number of Fundamental vibrations • A molecule containing n atoms has 3n degrees of freedom (translational, rotational, vibrational) • In Linear molecule with n number of atoms has: 3 degrees called the translational degrees of freedom (specify the centre of mass of molecule representing the translational motion of molecule 2 degrees called the rotational degrees of freedom (specify the orientation of molecule about its centre of mass, representing the rotational motion of molecule 3n – 5 degrees called the vibrational degrees of freedom (specify the relative positions of the ‘n’ nuclei representing vibrational motion • For Example: In CO2 the number of theoretical absorption bands could be (3x3-5 = 4) four. Degrees of freedom Molecule Total Translational Rotational Vibrational 3n-5 HCl 6 3 2 1 CO2 9 3 2 4 75 Infra RED Spectroscopy – Instrumentation Nernst glower: mixture of rare earth metal oxides rod maintained at 1500C. Prism and grating made of metal halides like NaCl, KBr Thermocouple or bolometer gas, liquid or solution, solid substance can be used as samples and the solvents like CCl4, CS2 and CHCl3 can be used as solvents 76 Infra RED Spectroscopy - Applications (i) Structure elucidation of organic compound We know that, If IR radiations are passed through a molecule the different functional groups in the molecule absorb IR radiations at their natural frequencies. This phenomenon is helpful in elucidating the structure of organic compounds. For example, • If the IR spectrum of a organic molecule shows absorption band at 5.82 (wave number range 1800cm-1 and 1650cm-1) then it is possible to identify that a carbonyl group should be present in it. • On the other hand if the IR spectrum contains no absorption band in between 5.4 to 6.3, then it is certain that no carbonyl group is present in the compound. • Likewise, if the presence of different functional groups in a compound is identified using the IR spectrum then the structure of unknown compound can be deduced with the help of other available data. 77 Infra RED Spectroscopy – Applications… (ii) Structure identification • The IR spectrum is useful in the identification of a newly synthesized compound. • The IR spectrum of unknown compound can be compared with the absorption spectrum of several known compounds in the literature. • If the IR spectrum of unknown compound matches with a known compound then the structures of both will also be similar. This method is called finger printing technique. (iii) Identification of impurities • If any extra absorption bands are recorded in the IR spectrum of a known compound then it is certain that the compound is not pure. • Therefore, the compound should be purified further to remove all the impurities. • After purification, if the IR spectrum is recorded then there will not be any extra absorption bands. • Thus IR spectrum is useful in determining the purity of a compound. . 78 Infra RED Spectroscopy – Applications… (iv) Study of reaction kinetics • The rate of a reaction can be studied by recording IR spectrum of the reaction mixture at definite intervals of time. • From the IR spectrum disappearance of absorption bands recorded in the beginning (due to the presence of reactants) and appearance of new absorption bands (due to the formation of products) can be observed in the course of time. • Thus the progress of organic reaction can be studied. (v) To ascertain hydrogen bonding in a molecule • In a sample with inter and intramolecular hydrogen bonding, if the dilution is increased and the IR spectrum is recorded, the absorption band, due to intermolecular hydrogen bonding disappears and the absorption band due to intramolecular hydrogen bonding remains unchanged. 79 Flame Photometry Principle When an atomized metal is passed through a flame, the outer shell electrons of metals are excited and emit energy of characteristic wavelengths. The emitted radiations are measured directly. Theory When the test solution is introduced into a flame, then solvent evaporates out and leaves the solute particles as salt. The salt particle dissociates in to atoms and reaches the vapourised state due to the heat of flame. The atoms in ground state get excited to the higher energy state by absorbing thermal energy from the flame. If these excited atoms returns back to the ground state emits radiation at particular wavelength. On passing the emitted radiations through optical filter permits the exits of characteristic wavelength of a metal under study. 80 Flame Photometry - Instrumentation The test solution containing metal ions is passed in to the atomizer through the nebuliser. Test solution sucked through the nebuliser, air from compressor and fuel gas from the fuel cylinder mix together in the mixing chamber. Now, the mixture is introduced into the flame. Here, all the excited atoms emits radiations of different wavelength. If the emitted radiations are passed through optical filter then, the optical filter permits only the characteristic wavelength of element under study. The transmitted light falls on the photo cell, amplifier and then on the digital recorder to get plot. 81 Flame Photometry - Instrumentation Flame photometry is useful in analyzing the elements which are easily excited in the flame heat and to emit radiations. To analyze alkali-metals. Example: Li, Na, K etc. To analyze alkaline-earth metals. Example: Ca 82 83 Good Luck! Dr. K. SIVAKUMAR Department of Chemistry 84 SCSVMV University [email protected]