* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Paramyxoviruses
2015–16 Zika virus epidemic wikipedia , lookup
Surround optical-fiber immunoassay wikipedia , lookup
African trypanosomiasis wikipedia , lookup
Bioterrorism wikipedia , lookup
Oesophagostomum wikipedia , lookup
Leptospirosis wikipedia , lookup
Gastroenteritis wikipedia , lookup
Ebola virus disease wikipedia , lookup
Hepatitis C wikipedia , lookup
Hospital-acquired infection wikipedia , lookup
Influenza A virus wikipedia , lookup
Neonatal infection wikipedia , lookup
Schistosomiasis wikipedia , lookup
Marburg virus disease wikipedia , lookup
West Nile fever wikipedia , lookup
Orthohantavirus wikipedia , lookup
Human cytomegalovirus wikipedia , lookup
Middle East respiratory syndrome wikipedia , lookup
Eradication of infectious diseases wikipedia , lookup
Herpes simplex virus wikipedia , lookup
Lymphocytic choriomeningitis wikipedia , lookup
Henipavirus wikipedia , lookup
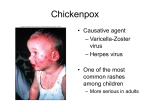
Dr. Nehal Draz Myxo = affinity to mucin Myxoviruses Orthomyxo viruses -Smaller -Segmented RNA genome -Liable to Agic variation Influenza viruses Paramyxo viruses -Larger -Single piece of RNA - Not liable to Agic variation - Parainfluenza - Mumps vairus - Measles virus - Respiratory syncytial virus Large Spherical envelopped Unsegmented –ve sense RNA The lipid envelope is associated with 2-virus specific glycoptns; HaemaglutininNeuraminidase (HN) ptn& fusion (F) ptn Commonest cause of bronchitis & pneumonia among infants< 1yr. Causes repeated infections throughout life, usually associated with moderate- to severe cold –like symptoms Severe lower respiratory tract disease may occur at any age, especially elderly & those with compromised cardiac, pulmonary or immune systems Specimens: nasal secretionsnasopharyngeal aspirate 1- Direct virus demonstration: - DIF: for detection of viral Ag - RT-PCR for detection of viral RNA 2- Viral isolation: - nasal secretions inoculated onto (HeLa) - Growth is recognized by development of CPE in the form of giant cells & syncytia Symptomatic treatment for mild disease Oxygen therapy & may be mechanical ventilation in children with severe disease Ribavirin aerosol No vaccine is yet available HPIVs are second to RSV as a common cause of lower respiratory tract disease in young children Similar to RSV, HPIVs can cause repeated infections throughout life, usually upper respiratory tract illness Can also cause severe lower respiratory tract infections ammong immunocompromised patients Each of the four HPIVs has different clinical & epidemiologic features The most distinctive clinical feature of HPIV-1& HPIV-2 is croup HPIV-3 is more associated with bronchiolitis & pneumonia HPIV-4 is infrequently detected, because it is less likely to cause severe disease Croup (laryngotracheobronchitis difficulty in breathing, hoarseness and a seal bark-like coughing Specimens: nasal secretionnasopharyngeal aspiratebronchoalveolar lavage 1- Direct virus demonstration: - DIF: for detection of viral Ag - RT-PCR for detection of viral RNA 2- Viral isolation: - Specimens are inoculated onto (MKTC) - Growth is recognized by hemadsorption using guinea pig RBCs or by direct IF 3- Serological tests: Based on Nt, HI, or ELISA for detection of IgM or IgG Paired acute & convalescent sera are necessary for IgG detection A four fold or more rise in the titre indicates infection Causes epidemic parotitis ( non suppurative inflammation of parotid) Mode of transmission: saliva Via aerosols & fomites The virus is secreted in urine so urine is a possible source of infection Infects children 5-15years Replicates in the nasopharynx ®ional LNs Incubation period: 2-25 d Lasts 3-5 d viremia -Salivary -Pancreas -Testes -ovaries glands meninges Long life immunity due to IgG neutralizing Abs Severe aseptic meningitis in adults Orchitis in adult males which might cause sterility Pancreatitis Oophritis & thyroiditis Specimens: - saliva - CSF - urine 1- Direct virus demonstration: - RT-PCR for detection of viral RNA 2- Viral isolation: - Specimens are inoculated onto (MKTC) or chick embryo - Growth is recognized by hemadsorption or by direct IF & by characteristic CPE giant cell formation 3- serology: ELISA is used for detection of IgM or IgG For IgG, paired acute & convalescent sera are necessary Four fold or more rise in IgG titer indicates infection Mumps vaccine Active immunization -Live attenuated -Given by subcutaneous injection -Long term immunity -Monovalent form or MMR vaccine Causes measles (robeola) One of the most contagious respiratory infections It can nearly affect every person (in a given population) by adolescence, in the absence of immunization programs Mode of transmission: - Large repiratory droplet -airborne Most infectious in the early stage Before the rash appears Replication initially in the upper & lower respiratory tract Followed by LNs replication Viremia & growth in a variety of epithelial tissue Incubation period: 1-2 wks In 2-3 days, no rash but fever, running nose, cough & conjunctivitis Koplick spots: slightly raised white dots, 2-3 mm in diameter are seen on the inside of the cheek shortly before rash onset persist for 1-3 days A characteristic maculopapular rash extending from face to extremities involving palms & soles : this seems to be associated with T-cells attacking virally infected endothelial cells in small blood vessels The rash lasts from 3-7 d & may be followed by skin exfoliation 1-Respiratory symptoms 2-Koplick spots Persist 1-3 days Disappear after the rash onset 2-3 days 3-Maculopapular rash Lasts for 3-7 days 4-Skin exfoliation Long life immunity due to IgG neutralizing Abs The virus invades the body via blood vessels reaches surface epithelium first in the respiratory tract where there are only 1-2 layers of epithelial cells Then in mucosae (Koplik's spots) and finally in the skin (rash). I- Respiratory Otitis media & bacterial pneumonia: common Giant cell pneumonia in patients with impaired CMI ( rare but fatal) II- Neurological Postinfectious encephalitis. Few days after the rash (1:1000) Subacute sclerosing panencephalitis (SSPE) (1:100.000) Specimens: nasal secretionsnasopharyngeal aspirate or swab- urine 1- Direct virus demonstration: - DIF: for detection of viral Ag - RT-PCR for detection of viral RNA 2- Viral isolation: - nasal secretions inoculated onto (MKTC) - Growth is recognized by development of CPE in the form of multinucleated giant cells containing both intranuclear & intracytoplasmic IBs 3- serology: ELISA is used for detection of IgM or IgG For IgM single serum specimen 1-2 wks after the rash onset For IgG, paired acute & convalescent sera are necessary Four fold or more rise in IgG titer indicates infection Passive immunization Measles IGs - For immunocompromised patients -Intramuscular within 6 days of exposure -Prevent measles symptoms in 80% of cases Active immunization Mumps vaccine -Live attenuated -Given by subcutaneous injection -Long term immunity -Monovalent form or MMR vaccine Causes German measles which is the mildest of common viral exanthems It is a member of rubiviruses but not an arbovirus Envelopped +ve sense ss RNA Posseses hemaglutinating ability 1- German measles: acute febrile illness with rash & lymphadenopathy affecting children & young adults 2- Congenital Rubella Syndrome: Serious abnormalities of the fetus as a consequence of maternal infection during early pregnancy Mode of transmission: droplet Initial viral replication occurs in the respiratory mucosa followed by multiplication in the cervical lymph nodes Viremia develops with spread to other tissues. As a result the disease symptoms develop in 50% of cases after an incubation period of 12-23 days Possibly 50% of infections are apparently subclinical Fever & malaise (prodromal symptoms) for 1-2 days Maculopapular rash appears on the face,then the trunk, then the extremities and disappears within 3 days Suboccipital and postauricular lymphadenopathy Extremely rare complications, self limiting encephalopathy Extremely rare (1/6000) Rubella encephalopathy 6 days after the rash appears Complete recovery with no sequalae Specimens: nasal secretionsnasopharyngeal aspirate or swab 1- Direct virus demonstration: - DIF: for detection of viral Ag - RT-PCR for detection of viral RNA 2- Viral isolation: - nasal secretions inoculated onto (MKTC) - Growth is recognized by interference with coxsakie virus 3- serology: ELISA is used for detection of IgM or IgG For IgM single serum specimen For IgG, paired acute & convalescent sera are necessary Four fold or more rise in IgG titer indicates infection Congenital rubella is a group of physical problems that occur in an infant when the mother is infected with the virus that causes German measles. Congenital rubella is caused by the destructive action of the rubella virus on the fetus at a critical time in development. The most critical time is the first trimester (the first 3 months of a pregnancy). After the fourth month, the mother's rubella infection is less likely to harm the developing fetus. The rate of congenital rubella has decreased dramatically since the introduction of the rubella vaccine. Risk factors for congenital rubella include: Not getting the recommended rubella immunization Contact with a person who has rubella (also called the 3-day measles or German measles) Pregnant women who are not vaccinated and who have not had rubella risk infection to themselves and damage to their unborn baby. Transient symptoms: growth retardation, anemia & thrombocytopenia Permanent defects: congenital heart diseases, total or partial blindness, deafness & mental retardation Progressive rubella panencephalitis: Extremely rare slow virus disease, develops in teens with death within 8 yrs During Pregnancy Detection of maternal IgM or rising IgG in serum Then, detection of rubella Ag in the amniotic fluid by DIF After Birth Live newborn: detection of IgM antirubella Abs in the serum of the baby by ELISA Stillbirth: virus isolation on MKTC vaccinate -Women in the childbearing age -School age children Pregnancy should be avoided 3 months after vaccination Maternal rubella infection confirmed during the first trimester???? Therapeutic abortion MMR Contains 3 live attenuated viruses: mumps, measles and rubella Given in 2 doses The first dose: to children 12-15 months of age by subcutaneous injection Why not before that? When is the second dose? Contraindications? Thank you