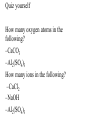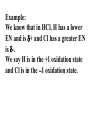* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Chemical Formulas
Membrane potential wikipedia , lookup
Acid–base reaction wikipedia , lookup
Aromaticity wikipedia , lookup
Isotopic labeling wikipedia , lookup
Ionic liquid wikipedia , lookup
X-ray photoelectron spectroscopy wikipedia , lookup
Stability constants of complexes wikipedia , lookup
Metastable inner-shell molecular state wikipedia , lookup
Bose–Einstein condensate wikipedia , lookup
Electron configuration wikipedia , lookup
Electrochemistry wikipedia , lookup
Debye–Hückel equation wikipedia , lookup
Chemical bond wikipedia , lookup
Homoaromaticity wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Nanofluidic circuitry wikipedia , lookup
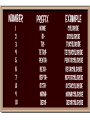
Chemical formulas indicate the relative number of atoms of each kind element in a chemical compound (ionic and molecular) Ionic compound the number of atoms in a formula units Capital letter each element NaCl Subscript # atoms CH3Cl Molecular compound the number of atoms in a molecule Ionic Compounds Monatomic ions - ions formed from a single atom Na+ or S2- usually tell by column on periodic table, some elements have more than one oxidation number or charge Binary compounds- only 2 elements in the compound Na2S Polyatomic ions - ions formed from more than one type of atom covalently bonded together OHPO43- NH4+ Quiz yourself How many oxygen atoms in the following? –CaCO3 –Al2(SO4)3 How many ions in the following? –CaCl2 –NaOH –Al2(SO4)3 Rules for writing formulas for ionic compounds 1. when making the formula the cation (positive ion) always goes 1st then the anion (negative ion) 2. the compound is neutral + = - charge 3. subscripts added to make charges cancel 4. When adding subscript to polyatomic ion it is put in ( )’s 5. Formula unit is always the simplest ratio of ions must ÷ subscripts The positive side of the formula must = the negative side of the element Examples on board 1. Sodium chloride Na+ ClNaCl (no charges written in the formula) 2. Calcium cholride Ca2+ ClCaCl2 3. Potassium sulfide K+ S2K2S 4. Aluminum chloride Al3+ ClAlCl3 5. Barium phosphate Ba2+ PO43Ba3(PO4)2 Cross over method - the charges become the subscripts of the other ion Beware subscripts must be simplest ratios Naming ionic compounds (stock system) 1. Binary compound - 2 elements •when naming the compound the name of the first element(+ion) stays the same •the last element ends in “ide” example NaCl sodium chloride Formula: ZnS Compound Name: Zinc Sulfide 2. Polyatomic ions- use their own name (on ion sheets). Example NH4Cl ammonium chloride Ba3(PO4) barium phosphate Na OH sodium hydroxide http://www.chem4kids.com/files/atom_compounds.html Stock System 3. Elements with more than one oxidation number (charge) -put the oxidation number in Roman numerals in ( )’s Example Pb(NO3)2 lead (II) nitrate Usually transition metals have more then one oxidation number also lead and tin Check ion sheet Molecular Compounds- made of neutral atoms sharing electrons & form neutral molecules 2 types of formulas molecular formula- represents a molecule C6H6 empirical formulas - simplest ratio of atoms (formula units are always empirical formulas) CH diatomic molecule - 2 atoms of the same element covalently bonded together There are 7 H2 N2 O2 F2 Cl2 Br2 I2 diatomic elements and they form the shape of a 7 on the periodic table except for H which is always an oddball Only diatomic when pure element not necessarily when forming a compound Use prefixes to name molecular compounds prefix subscript mono no subscript (1 atom) di 2 tri 3 tetra 4 penta 5 hexa 6 hepta 7 octa 8 nona 9 & deca 10 Rules for molecular compounds 1. The less electronegative element is given 1st and only given a prefix if it has a subscript >1 (never start a name with mono) 2. Second element- add a prefix and add “ide” (drop prefix ending if first letter in the name of the element is a vowel) P4O10 tetraphosphorous decoxide Oxidation Numbers •In polar covalent bonds, the electrons are located closer to one atom than the other. This is reflected in a partial charge, + and -, for each atom. •The oxidation state or oxidation number of an atom is an indication of the amount of charge each atom carries. The one with the greater EN gets both e- Example: We know that in HCl, H has a lower EN and is + and Cl has a greater EN is -. We say H is in the +1 oxidation state and Cl is in the -1 oxidation state. Acids endings change “-ate” to “-ic” H2SO4 “-ite” to “-ous” H2SO3 sulfuric sulfurous Important acids to know: HCl hydrochloric HNO3 nitric H3PO4 phosphoric HC2H3O2 acetic (also CH3COOH) H2CO3 carbonic