* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Redox reactions - SALEM-Immanuel Lutheran College
Hypervalent molecule wikipedia , lookup
Rate equation wikipedia , lookup
Chemical bond wikipedia , lookup
History of molecular theory wikipedia , lookup
Stoichiometry wikipedia , lookup
Debye–Hückel equation wikipedia , lookup
Hydrogen-bond catalysis wikipedia , lookup
Marcus theory wikipedia , lookup
Electron configuration wikipedia , lookup
Double layer forces wikipedia , lookup
Artificial photosynthesis wikipedia , lookup
Chemical reaction wikipedia , lookup
Electrolysis of water wikipedia , lookup
Hydrogen atom wikipedia , lookup
Microbial metabolism wikipedia , lookup
Relativistic quantum mechanics wikipedia , lookup
Atomic theory wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Water splitting wikipedia , lookup
Photoredox catalysis wikipedia , lookup
Oxidation state wikipedia , lookup
Electrochemistry wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
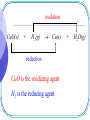
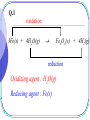
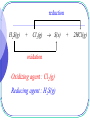
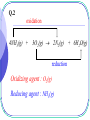
Early Definitions of Redox Reactions Redox Reactions in terms of Loss and Gain of Oxygen Oxidation : The addition of oxygen to a substance Reduction : The removal of oxygen from a substance 1 oxidation CuO(s) + H2(g) Cu(s) reduction CuO is the oxidizing agent H2 is the reducing agent 2 + H2O(g) Q.1 oxidation 3Fe(s) + 4H2O(g) Fe3O4(s) + 4H2(g) reduction Oxidizing agent : H2O(g) Reducing agent : Fe(s) 3 Redox Reactions in terms of Loss and Gain of Hydrogen Oxidation : The removal of hydrogen from a substance Reduction : The addition of hydrogen to a substance 4 reduction H2S(g) + Cl2(g) S(s) oxidation Oxidizing agent : Cl2(g) Reducing agent : H2S(g) 5 + 2HCl(g) Q.2 oxidation 4NH3(g) + 3O2(g) 2N2(g) + 6H2O(g) reduction Oxidizing agent : O2(g) Reducing agent : NH3(g) 6 The application is limited to reactions involving O and/or H Broader definitions were developed 7 Modern Definitions of Redox Reactions 1. Redox Reactions in terms of Electron Transfer 2. Redox Reactions in terms of Changes in Oxidation States or Oxidation Numbers 8 Redox Reactions in terms of Electron Transfer Oxidation : The loss of electrons Reduction : The gain of electrons 9 oxidation 2Na(s) + Cl2(g) 2NaCl(s) reduction 2Na(s) 2Na+(s) + 2e Cl2(g) + 2e 2Cl(s) 10 Q.3 oxidation Mg(s) + Cu2+(aq) Mg2+(aq) + Cu(s) reduction Mg(s) Cu2+(aq) + 11 Mg2+(aq) 2e + Cu(s) 2e Q.4 oxidation Mg(s) + H2O(g) MgO(s) + H2(g) reduction Mg(s) H2O(g) 12 Mg2+(s) + 2e ? + 2e H2(g) 13 • oxidation and reduction always accompany each other • Acid-base reactions are competitions for protons • Redox reactions are competitions for electrons Competitions for protons Gain of H+ stronger acid weaker acid Cl(aq) + HCl(aq) + H2O(l) stronger base weaker base Gain of H+ 14 H3O+(aq) Competitions for electrons Gain of e stronger R.A. weaker R.A. Mg(s) + Cu2+(aq) Mg2+(aq) + stronger stronger O.A O.A. weaker weaker O.A. O.A. Gain of e 15 Cu(s) Redox Reactions in terms of Changes in O.N. Oxidation : Increase in O.N. Reduction : Decrease in O.N. 16 Definition of Oxidation Number : the presumed electrical charge of the atom or ion in the substance 17 The bond pair in any non-polar bond or slightly polar bond between like atoms is divided equally between the two atoms. the oxidation number of an atom in an element is zero. E.g. H H , bond pair is equally shared presumed electrical charge on H is 0. O.N. of H is 0 18 The bond pair in any polar covalent bond (including ionic bond) between two unlike atoms is presumed to belong to the more electronegative atom. the oxidation number of an ion in an ionic compound is equal to its actual electrical charge. E.g. Na+Cl , O.N. of Na+ is +1 ; 19 O.N. of Cl is –1 the oxidation number of an atom in a compound with polar covalent bonds is equal to the charge it would have if it existed as an ion in that compound. E.g. H Cl , since Cl is more electronegative, the presumed electrical charges and thus O.N. of Cl and H are 1 and 1 respectively. 20 - + - O C O Since O is more electronegative, the presumed charge on O is –2 O.N. of O = –2 O.N. of C = +4 21 22 • To work out O.N., information about electronegativity and bond arrangement of the species are required. • To make it simple, a set of rules has been devised for assignment of O.N. Rules for Assigning Oxidation States : Oxidation state of an atom in an element is 0 Sum of oxidation states of a neutral compound is 0 Sum of oxidation states of a simple or polyatomic ion = charge on the ion 23 Elements with Fixed Oxidation States in Their Compounds : Group I metals +1 Group II metals +2 Al +3 H +1 in covalent compounds with non-metals; 1 when combined with Group I or II metals. E.g. metal hydrides such NaH, MgH2. 24 F 1 O 2, except in peroxides(1) , superoxides(0.5) and compounds with F where it is +ve. Cl 1 except in compounds with F and O. Q.5 (1) HClO4 (+1) + Cl + 4(-2) = 0 Cl = +7 Chloric(VII) acid HClO3 (+1) + Cl + 3(-2) = 0 Cl = +5 25 Chloric(V) acid Q.5 (1) HClO2 (+1) + Cl + 2(-2) = 0 Cl = +3 Chloric(III) acid HClO (+1) + Cl + (-2) = 0 Cl = +1 26 Chloric(I) acid Q.5 (2) Na2SO4 2(+1) + S + 4(-2) = 0 S = +6 Sodium sulphate(VI) Na2SO3 2(+1) + S + 3(-2) = 0 S = +4 Sodium sulphate(IV) 27 Q.5 (3) KNO3 (+1) + N + 3(-2) = 0 N = +5 Potassium nitrate(V) KNO2 (+1) + N + 2(-2) = 0 N = +3 Potassium nitrate(III) 28 Q.5 (4) KMnO4 (+1) + Mn + 4(-2) = 0 Mn = +7 Potassium manganate(VII) K2MnO4 2(+1) + Mn + 4(-2) = 0 Mn = +6 Potassium manganate(VI) 29 Q.5 (5) FeCl3 Fe + 3(-1) = 0 Fe = +3 Iron(III) chloride FeCl2 Fe + 2(-1) = 0 Fe = +2 Iron(II) chloride 30 Q.5 (6) Na2CO3 2(+1) + C + 3(-2) = 0 C = +4 Sodium carbonate* There is only one type of carbonate, O.N. of carbon needs not be indicated. 31 Q.5 (6) CO C + (-2) = 0 C = +2 Carbon monoxide CO2 C + 2(-2) = 0 C = +4 Carbon dioxide 32 Q.5 (7) CH4 C + 4(+1) = 0 C = -4 Methane H H C H 33 H C is more electronegative than H, O.N. of C is – 4 Q.5 (7) C2H6 2C + 6(+1) = 0 C = -3 Ethane 34 A bond pair is equally shared between two C atoms H -3 H 35 H -3 C C H H H Q.5 (7) C3H8 3C + 8(+1) = 0 8 C 3 propane 36 Electrons between C atoms are equally shared H 37 H H -3 C -2 C H H H -3 C H H Q.5 (8) S2O32 2S + 3(-2) = -2 S = +2 Thiosulphate 38 Electrons between S atoms are equally shared 0 S 0 +4 +4 S -2 O -2 O -2-2 O -2 -2 39 Q.5 (8) S4O62 4S + 6(-2) = -2 S = +2.5 Tetrathionate 40 Electrons between S atoms are equally shared O +5 S O 0 S 0 S O +5 S O O 41 O Q.5 (8) S2O82 2S + 8(-2) = -2 S = +7 The highest O.N. of S is +6 Peroxodisulphate(VI) 42 The highest O.N. = Group number The lowest O.N. = Group number – 8 and -4 E.g. N, Highest O.N. = +5; Lowest O.N. = -3 Cl, Highest O.N. = +7; Lowest O.N. = -1 43 -2 O +6 -2 O S -1 O -1 O O-2 -2 O +6 S O -2 44 -2 O Q.5 (9) 00 00 00 S C S Electrons are equally shared between C and S 45 Q.5 (9) C2H4O3 2C + 4(+1) + 3(-2) = 0 C = +1 Ozonide 46 +1 H -2 H O +1 H 0 C -1 O 47 +1 +1 C 0 O -1 H Q.6 (1) +3 +2 Fe3O4(s) + 8H+(aq) Fe2+(aq) + 2Fe3+(aq) + 4H2O(l) FeO Fe2O3 mixed oxide no change in O.N. non-redox reaction 48 Q.6 (2) +4 +2 Pb3O4(s) + 4H+(aq) PbO2(s) + 2Pb2+(aq) + 2H2O(l) 2PbO PbO2 mixed oxide no change in O.N. non-redox reaction 49 0 Q.6 (3) S2O32-(aq) + 2H+(aq) 0 +4 -2 O 50 0 +4 +4 S(s) + SO2(g) + H2O(l) S No change in O.N. S O -2 non-redox reaction O -2 Internal Redox Reactions When the oxidizing agent and the reducing agent of a redox reaction are the same substance, the redox reaction is known as an internal redox reaction 51 oxidation -2 2KClO3(s) +5 reduction 52 0 2KCl(s) -1 + 3O2(g) oxidation -2 2Pb(NO3)2(s) +5 0 2PbO(s) + 4NO2(g) + O2(g) +4 reduction 53 Q.7(a) R.A. : NH4+(s) oxidation -3 NH4NO3(s) +1 N2O(g) + 2H2O(g) +5 reduction 54 O.A. : NO3(s) Q.7(b) R.A. : Fe2+(s) oxidation +2 2FeSO4(s) +3 Fe2O3(s) + SO2(g) + SO3(g) +6 +4 reduction 55 O.A. : SO42(s) Q.7(c) R.A. : NH4+(s) oxidation -3 0 (NH4)2Cr2O7(s) Cr2O3(s) + 4H2O(g) + N2(g) +6 +3 reduction 56 O.A. : Cr2O72(s) Disproportionation A redox reaction in which an element (combined or uncombined) in an intermediate oxidation state is simultaneously reduced and oxidized 57 oxidation -1 2H2O2(aq) 2H2O(l) -2 reduction 58 + 0 O2(g) oxidation 0 Cl2(aq) + H2O(l) HCl(aq) + -1 reduction 59 +1 HOCl(aq) Q.8 (a) oxidation +4 +5 2NO2(g) + H2O(l) HNO3(aq) + HNO2(aq) +3 reduction 60 Q.8 (b) oxidation +3 +4 (COOH)2(s) CO(g) + CO2(g) + H2O(l) +2 reduction O +3 OH C C HO 61 +3 O Q.8 (c) oxidation +1 2CuI(s) Cu(s) 0 reduction 62 + +2 CuI2(aq) Balancing Redox Equations Two systematic methods : - 63 The Oxidation Number Method The Half Reaction Method Unlike the method of inspection, which is a general method of balancing chemical equations, • the oxidation number method only applies to redox reactions • the half reaction method only applies to electron-transfer reactions. 64 The Oxidation Number Method Principle : Total increase in O.N. = total decrease in O.N. 65 Example 1 All fixed RHS needs 6 H RHS needs 3H2O Step 3 : Balance the Rest of the Equation by Inspection -2 0 C2H5OH(l) + 3 O2(g) +4 -2 2 CO2(g) -2 + 3 H2O(g) Step 1 :O.N. Work: C, out -2 theChanges inIn O.N. In +4 O.N. : O, 0 -2 Step : Balance Changes in O.N. Total2increase in the O.N. = 2 (+6) = +12 Total decrease in O.N. = 12 = 6 (2) six oxygen atoms are needed. 66 Example 2 14 7 hydrogen oxygen atoms atoms on on thethe left right 7 water 14 hydrogen molecules ions are are needed needed The charges are automatically balanced fixed Step fixed 3 : Balance the Rest of the Equation by Inspection +3 +2 +3 Cr2O72(aq) + 6 Fe2+(aq) +14H+(aq) 2 Cr3+(aq) + 6 Fe3+(aq) + 7H2O(l) +6 Step 1 : Work out the Changes in O.N. In O.N. : Fe, +2 +3 In O.N. : Cr, +6 +3 Step : Balance Changes in O.N. Total2decrease inthe O.N. = 2 (-3) = -6 Total increase in O.N. = +6 = 6 (+1) six Fe2+ ions are needed 67 Q.9 (a) All fixed RHS needs 2H2O 0 +5 +6 +4 HNO3(aq) H2SO4(aq) + 6NO NO2(g) + 2 H2O(l) S(s) + 6HNO In O.N. : 0 +6 In O.N. : +5 +4 Total in O.N. = +6 Total in O.N. = -6 = 6(-1) 6 HNO3 are needed 68 Q.9 (b) All fixed RHS needs 1.5 H2O 4NH3(g) + 5O2(g) 4NO(g) + 6H2O(l) -3 NH3(g) + 5 0 O (g) 4 2 +2 -2 NO(g) + In O.N. : -3 +2 In O.N. : 0 -2 Total in O.N. = +5 Total in O.N. = -5 = 2.5(-2) 1.25 O2 are needed 69 -2 3 H O(l) 2 2 Q.9 (c) All fixed RHS needs 2H2O 0 C(s) +6 + 2 H2SO4(aq) +4 CO2(g) In O.N. : 0 +4 +4 + 2 SO2(g) In O.N. : +6 +4 Total in O.N. = +4 Total in O.N. = -4 = 2(-2) 2 H2SO4 are needed 70 + 2 H2O(l) Q.9 (d) Six HNO3 should be added to LHS to balance the six Cu(NO All fixed NO RHS needs 4H 3 in 3)22O 0 3Cu(s) 2 +5 2+2 +2 + 28 HNO3(aq) 3Cu(NO3)2(aq) + 2NO(g) + 4 H2O(l) 3 3 In O.N. : 0 +2 In O.N. : +5 +2 Total in O.N. = +2 Total in O.N. = -2 = (2/3)(-3) (2/3) HNO3 is needed 71 Q.9 (e) 4FeS2(s) + 11O2(g) +2 -1 2 FeS2(s) 11 0 + O2(g) 2 2Fe2O3(s) + 8SO2(g) +3 -2 Fe2O3(s) + 4 SO2(g) In O.N. : Fe, +2 +3 In O.N. : O, 0 -2 S, -1 +4 Total in O.N. = 2(+1) + 4(+5) = +22 Total in O.N. = -22 = 11(-2) 72 +4 -2 11 oxygen atoms are required Q.9 (e) Inspection method 11 2 2FeS2(s) + O2(g) Fe2O3(s) + 4SO2(g) Balancing Fe Balancing S Balancing O 4FeS2(s) + 11O2(g) 73 2Fe2O3(s) + 8SO2(g) Q.9 (f) 2Na(s) + 0 2NH3(g) +1 Na(s) + NH3(g) 2NaNH2(s) + H2(g) +1 NaNH2(s) In O.N. : Na, 0 +1 Total in O.N. = 1(+1) = +1 Total in O.N. = -1 1 hydrogen atom are needed 0.5 H2 is needed 74 10 + H2(g) 2 In O.N. : H, +1 0 Q.9 (g) All fixed 0.5 H2O on the right 4MnO4(aq) + 4H+(aq) 4MnO2(s) + 2H2O(l) + 3O2(g) +7 -2 MnO4(aq) + H+(aq) +4 MnO2(s) In O.N. : O, -2 0 In O.N. : Mn, +7 +4 Total in O.N. = 1(-3) = -3 Total in O.N. = +3 = 1.5(+2) 1.5 oxygen atoms are needed 0.75 O2 is needed 75 1 30 + H2O(l) + O2(g) 2 4 Q.9(h) Highly exothermic, glycerol burns with a purple flame +7 -2/3 +4 +3 +4 KMnO4(s) + C3H5(OH)3(l)K2CO3(s)+Mn2O3(s)+CO2(g)+ H2O(l) In O.N. : C, -2/3 +4 In O.N. : Mn, +7 +3 Total in O.N. = 3(4+2/3) = +14 Total in O.N. = 7/2(-4) = -14 7/2KMnO4(s) + C3H5(OH)3(l)K2CO3(s)+7/4Mn2O3(s)+CO2(g)+ H2O(l) 76 Q.9(h) 7/2KMnO4(s) + C3H5(OH)3(l)K2CO3(s)+7/4Mn2O3(s)+CO2(g)+ H2O(l) 14KMnO4(s) + 4C3H5(OH)3(l)K2CO3(s)+7Mn2O3(s)+CO2(g)+ H2O(l) 14KMnO4(s) + 4C3H5(OH)3(l)7K2CO3(s)+7Mn2O3(s)+CO2(g)+ H2O(l) 14KMnO4(s) + 4C3H5(OH)3(l)7K2CO3(s)+7Mn2O3(s)+5CO2(g)+ H2O(l) 14KMnO4(s) + 4C3H5(OH)3(l)7K2CO3(s)+7Mn2O3(s)+5CO2(g)+ 16H2O(l) 77 The Half Reaction Method In Acidic Solution : H+ and H2O are used to balance the half equations In Basic Solution : OH and H2O are used to balance the half equations 78 In Acidic Solution : - Example MnO4(aq) + Fe2+(aq) Mn2+(aq) + Fe3+(aq) +2 +7 (aq) Mn2+(aq) Reduction half equation : MnO Step 1 : Identify and Write Equations 4 for the Half Reactions +2 +3 Oxidation half equation : Fe2+(aq) Fe3+(aq) Step 2 : Balance Each Half Equation 79 For the reduction half equation : +7 (1) MnO4(aq) + 8H+(aq) + 5e +2 Mn2+(aq) + 4H2O(l) O.N. of Mn decreases by 5 units 5 electrons are gained per (3) Balance the Hydrogen Hydrogen Ion (1) Add electrons according toAdding the change in O.N. (2) Oxygen byby Adding Water Mn atom (Acidic Solution) For the oxidation half equation : (2) Fe2+(aq) Fe3+(aq) + e 2+ Step Balance the Electron in the Half Equations and (1)3 :O.N. of Fe increases byTransfer 1 unit 1 electron is lost per Fe (2)/(3) No need to balance oxygen and hydrogen (1) + 5Add (2) the Half Equations 5Fe2+(aq)+MnO4(aq)+8H+(aq) 5Fe3+(aq)+Mn2+(aq)+4H2O(l) 80 In Basic Solution : - Example 1 MnO4(aq) + Fe(OH)2(s) Fe(OH)3(s) +7 + Mn(OH)2(s) +2 Reduction half equation : MnO4(aq) Mn(OH)2(s) Step 1 : Identify and Write Equations for the Half Reactions +2 +3 Oxidation half equation : Fe(OH)2(s) Fe(OH)3(s) Step 2 : Balance Each Half Equation 81 For the reduction half equation : +7 (1) MnO4(aq) + 4H2O(l) +2 + 5e Mn(OH)2(s) + 6OH(aq) O.N. of Mn oxidation decreases by 5ofunits to5 the electrons arein gained per (3) (1) Balance Add electrons the Rest according the Equation change by Adding O.N. Water For the half equation : (2) Balance the charge by adding OH Mn atom (2) Fe(OH)2(s) + OH(aq) Fe(OH)3(s) + e 2+ (1)(2) O.N. of Fe increases by 1 unit 1 electron is lost per Fe Step 3 : Balance the Electron Transfer in the Half Equations and (1) +Balance 5 (2)the charge by adding OH Add the Half Equations MnO4 (aq)+4H2O(l)+5Fe(OH)2(s) Mn(OH)2(s)+OH (aq)+5Fe(OH)3(s) 82 In Basic Solution : - Example 2 Ag(s) + CN(aq) + O2(g) 0 Ag(CN)2(aq) Reduction half equation : O2(g) -2 ? Step 1 : Identify and Write Equations for the Half Reactions 0 +1 Oxidation half equation : Ag(s) Ag(CN)2(aq) Step 2 : Balance Each Half Equation 83 For the reduction half equation : (1) 0 O2(g) + 2H2O(l) + 4e -2 4OH(aq) O.N. decreases by 2 Rest units perthe oxygen 2 change electrons are (3) Balance the of Equation by Adding Water (1) Add electrons according to the in O.N. (2) Balance the charge by adding gained OH per O atom For the oxidation half equation : (2) Ag(s) + 2CN(aq) Ag(CN)2(aq) + e (1) O.N. of Ag increases by 1 unit 1 electron per Ag atom is lost Step 3 : Balance the Electron Transfer in the Half Equations and (2) Balance the charge by adding CN (1) + 4 Add (2)the Half Equations O2(g) + 2H2O(l) + 4Ag(s) + 8CN(aq) 4OH(aq) + 4Ag(CN)2(aq) 84 Q.10 (a) Cr2O72(aq) + C2H5OH(aq) Cr3+(aq) + CO2(g) +6 +3 Reduction half equation : Cr2O72(aq) Cr3+(aq) Step 1 : Identify and Write Equations for the Half Reactions +4 -2 Oxidation half equation : C2H5OH(aq) CO2(g) Step 2 : Balance Each Half Equation 85 For the reduction half equation : (1) Cr2O72(aq) + 6e + 14H+(aq) 2 Cr3+(aq) + 7H2O(l) O.N. of Cr byby 3Adding units 3 electrons are gained per Balance thedecreases Hydrogen Ion (Acidic Solution) For the oxidation halfHydrogen equation : Balance the Oxygen by Adding Water Cr atom (2) C2H5OH(aq) + 3H2O(l) 2CO2(g) + 12e + 12H+(aq) Balance the Electron Transfer in the Half Equations and Add the Balance the Oxygen by Adding Water O.N. of C increases by 6 unit 6 electrons are lost per C atom Balance the Hydrogen by Adding Hydrogen Ion (Acidic Solution) 2(1) (2) Half+Equations 2 Cr2O72(aq)+16H+(aq)+C2H5OH(aq) 4Cr3+(aq)+11H2O(l) + 2CO2(g) 86 Q.10 (b) Zn(s) + VO2+(aq) Zn2+(aq) + V2+(aq) +5 Reduction half equation : VO2+(aq) +2 V2 +(aq) Step 1 : Identify and Write Equations for the Half Reactions +2 0 Oxidation half equation : Zn(s) Zn2+(aq) Step 2 : Balance Each Half Equation 87 For the reduction half equation : (1) VO2+(aq) + 3e + 4H+(aq) V2 +(aq) + 2H2O(l) O.N.Balance ofthe V decreases by 3by units Water 3 electrons are gained per Balance Hydrogen the Oxygen by Adding Adding Hydrogen Ion (Acidic Solution) For the oxidation half equation :V atom (2) Zn(s) Zn2+(aq) + 2e Balance theincreases Electronby Transfer Half Equations the O.N. Zn 2 unit in the 2 electrons are lostand per Add Zn atom 2(1) +of3(2) Half Equations 2VO2+(aq)+ 8H+(aq)+3Zn(s) 2V2+(aq)+4H2O(l)+3Zn2+(aq) 88 Q.11 (a) Cr2O72(aq) + C2H5OH(aq) Cr(OH)3(s) + CO32(aq) +6 Reduction half equation : Cr2O72(aq) +3 Cr(OH)3(s) Step 1 : Identify and Write Equations for the Half Reactions +4 -2 Oxidation half equation : C2H5OH(aq) CO32(aq) Step 2 : Balance Each Half Equation 89 For the reduction half equation : (1) Cr2O72(aq) + 6e + 7H2O(l) 2Cr(OH)3(s) + 8OH(aq) O.N. of Crthe decreases by 3Equation units 3 electrons are gained per Balance Rest of the by Adding Water (aq) Balance the Charge by Adding OH Cr atom : For the oxidation half equation (2) C2H5OH(aq)+ 16OH(aq) 2CO32(aq) + 12e + 11H2O(l) (aq)Equations Balance the Electron Transfer in the Half andCAdd the Balance Balance the the Rest Charge ofby the Equation Adding OH Adding Water O.N. of C increases 6by unit 6by electrons are lost per atom Half+Equations 2(1) (2) 2Cr2O72(aq)+ 3H2O(l)+ C2H5OH(aq) 4Cr(OH)3(s)+ 2CO32(aq) 90 Q.11 (b) MnO4(aq) + C2O42(aq) MnO2(s) + CO32(g) +7 Reduction half equation : MnO4(aq) +4 MnO2(s) Step 1 : Identify and Write Equations for the Half Reactions +4 +3 Oxidation half equation : C2O42(aq) CO32(g) Step 2 : Balance Each Half Equation 91 For the reduction half equation : (1) MnO4(aq) + 3e + 2H2O(l) MnO2(s) + 4OH(aq) O.N. of Mn decreases byEquation 3 units by Adding 3 electrons are gained per Balance the Rest of the Water (aq) Balance the Charge by Adding OH Mn atom : For the oxidation half equation (2) C2O42(aq) + 4OH(aq) 2CO32(g) + 2e + 2H2O(l) (aq)Equations Balance theincreases Electron inbythe Half Add the Balance Rest the Charge of the Equation Adding Adding Water O.N.Balance of the C byTransfer 1by unit 1OH electron is lost perand C atom Half Equations 2(1) + 3(2) 2MnO4(aq)+3C2O42(aq)+ 4OH(aq) 2MnO2(s)+6CO32(g)+ 2H2O(l) 92 Q.12 (a) CxHyOz + Cr2O72 CO2 + Cr3+ +6 +3 Reduction half equation : Cr2O72(aq) Cr3+(aq) Step 1 : Identify and Write Equations for the Half Reactions +4 ? Oxidation half equation : CxHyOz(aq) CO2 x(?) y(1) z(2) 0 2z - y ? x 93 For the reduction half equation : (1) Cr2O72(aq) + 6e + 14H+(aq) 2 Cr3+(aq) + 7H2O(l) O.N. ofthe Crthe decreases byby 3 units Water 3 electrons gained per Balance Balance the Hydrogen Oxygen by Adding Adding Hydrogen Ion are (Acidic Solution) For oxidation half equation : Cr atom (2) CxHyOz(aq) + [2(z-2x)-y]H+(aq) xCO2 x 4 2z y e x + (z-2x)H2O(l) Balance Transfer in the Half Equations and Add the Balancethe theElectron Oxygen by Hydrogen by Adding Adding Water Hydrogen Ion (Acidic Solution) Half Equations O.N. of C increases by 4 94 2z y 2z y electrons are lost units 4 x x per C atom (1) Cr2O72(aq) + 6e + 14H+(aq) 2 Cr3+(aq) + 7H2O(l) (2) CxHyOz(aq) + [2(z-2x)-y]H+(aq) xCO2 x 4 2z y e + (z-2x)H2O(l) x 2z y x 4 (1) 6(2) x 2z y 2z y + + 6 C H O (aq) + 6[2(z-2x)-y]H+(aq) x 4 Cr2O72(aq) 14x 4 H x y z x x 2z y 3+ 2z y 2x 4 Cr (aq) 7x 4 H2O(l) + 6xCO2 x x 95 + 6(z-2x)H2O(l) Q.12 (b) 2z y 2z y + + 6 C H O (aq) + 6[2(z-2x)-y]H+(aq) x 4 Cr2O72(aq) 14x 4 H x y z x x 2z y 3+ 2z y 2x 4 Cr (aq) 7x 4 H2O(l) + 6xCO2 x x nCr O 2 2 7 nCxHyOz 96 + 6(z-2x)H2O(l) 2z y 4x 2z y x 4 x x 4x y 2z x 6 6 6 19.1 Redox Reactions (SB p.185) (a) By using oxidation numbers, determine whether the underlined species is an oxidizing agent or a reducing agent. 2Na(s) + 2HCl(aq) 2NaCl(aq) + H2(g) (a) Na(s) is the reducing agent as the oxidation number of Na is increased from 0 for Na(s) to +1 for NaCl(aq). 97 Answer 19.1 Redox Reactions (SB p.185) (b) By using oxidation numbers, determine whether the underlined species is an oxidizing agent or a reducing agent. Cu2+(aq) + Ni(s) Cu(s) + Ni2+(aq) (b) Cu2+(aq) is the oxidizing agent as the oxidation number of Cu is decreased from +2 for Cu2+(aq) to 0 for Cu(s). 98 Answer 19.1 Redox Reactions (SB p.185) (c) By using oxidation numbers, determine whether the underlined species is an oxidizing agent or a reducing agent. 2MnO4–(aq) + 5Cu(s) + 16H+(aq) 2Mn2+(aq) + 5Cu2+(aq) + 8H2O(l) (c) MnO4–(aq) is the oxidizing agent as the oxidation number of Mn is decreased from +7 for MnO4–(aq) to +2 for Mn2+(aq). 99 Answer 19.1 Redox Reactions (SB p.185) (d) By using oxidation numbers, determine whether the underlined species is an oxidizing agent or a reducing agent. 2SO2(g) + O2(g) 2SO3(g) (d) SO2(g) is the reducing agent as the oxidation number of S is increased from +4 for SO2(g) to +6 for SO3(g). Back 100 Answer 19.2 Balancing Redox Equations (SB p.187) Chlorine reacts with iron(II) sulphate solution to form chloride ions and iron(III) sulphate. Write the two half equations, and hence the overall equation for the reaction. State the reducing agent and oxidizing agent, and explain your answers. Answer 101 19.2 Balancing Redox Equations (SB p.187) Back Oxidation: Fe2+(aq) Fe3+(aq) + e– .......................…................ (1) Reduction: Cl2(g) + 2e– 2Cl–(aq) ............................................ (2) Multiplying equation (1) by 2, we obtain: 2Fe2+(aq) 2Fe3+(aq) + 2e– ...............…………...................... (3) Combining equations (2) and (3) together and eliminating the electrons on both sides, we have the overall equation: 2Fe2+(aq) + Cl2(g) 2Fe3+(aq) + 2Cl–(aq) Assigning oxidation numbers to Fe and Cl, we can identify the oxidizing agent and reducing agent of the reaction. 2Fe2+(aq) + Cl2(g) 2Fe3+(aq) + 2Cl–(aq) +2 0 +3 –1 Fe2+(aq) is the reducing agent as the oxidation number of Fe is increased from +2 for Fe2+(aq) to +3 for Fe3+(aq) in the reaction. Cl2(g) is the oxidizing agent as the oxidation number of Cl is decreased 102 from 0 for Cl2(aq) to –1 for Cl–(aq) in the reaction. 19.2 Balancing Redox Equations (SB p.190) Peroxodisulphate(VI) ions (S2O82–) react with manganese(II) ions in an alkaline medium to form sulphate ions (SO42–) and manganese(IV) oxide (MnO2). Write the overall ionic equation for the reaction by using the half equation method. State the oxidizing agent and reducing agent of the reaction. Answer 103 19.2 Balancing Redox Equations (SB p.190) Back The half equation for oxidation is: Mn2+(aq) + 4OH–(aq) MnO2(s) + 2H2O(l) + 2e– The half equation for reduction is: S2O82–(aq) + 2e– 2SO42–(aq) Combining the two half equations and eliminating the electrons on both sides, we obtain: Mn2+(aq) + 4OH–(aq) + S2O82–(aq) MnO2(s) + 2H2O(l) + 2SO42–(aq) +2 +7 +4 +6 S2O82–(aq) is the oxidizing agent as the oxidation number of S is decreased from +7 for S2O82–(aq) to +6 for SO42–(aq) in the reaction. Mn2+(aq) is the reducing agent as the oxidation number of Mn is increased from +2 for Mn2+(aq) to +4 for MnO2(s). 104 19.2 Balancing Redox Equations (SB p.190) Sodium sulphate(IV) solution turns acidified potassium dichromate(VI) solution from orange to green. Write the overall ionic equation for the reaction by using the half equation method. State the oxidizing agent and reducing agent of the reaction. Answer 105 19.2 Balancing Redox Equations (SB p.190) Back The half equation for oxidation is: SO32–(aq) + H2O(l) SO42–(aq) + 2H+(aq) + 2e–.........……...…..... (1) The half equation for reduction is: Cr2O72–(aq) + 14H+(aq) + 6e– 2Cr3+(aq) + 7H2O(l) ............……... (2) Multiplying equation (1) by 3, we obtain: 3SO32–(aq) + 3H2O(l) 3SO42–(aq) + 6H+(aq) + 6e– .......……......... (3) Combining equations (2) and (3) and eliminating the electrons on both sides, we have: Cr2O72–(aq) + 8H+(aq) + 3SO32–(aq) +6 +4 2Cr3+(aq) + 3SO42–(aq) + 4H2O(l) +3 +6 Sodium sulphate(IV) is the reducing agent as the oxidation number of S is increased from +4 for SO32–(aq) to +6 for SO42–(aq). Acidified potassium dichromate(VI) is the oxidizing agent as the oxidation number of Cr is 106 decreased from +6 for Cr2O72–(aq) to +3 for Cr3+(aq). 19.2 Balancing Redox Equations (SB p.191) Using the half equation method, write the overall ionic equations for the following reactions. State the oxidizing agent and reducing agent of the reaction, and explain your answers. (a) Reaction between lead and silver nitrate solution Answer 107 19.2 Balancing Redox Equations (SB p.191) (a) 108 The half equation for oxidation is: Pb(s) Pb2+(aq) + 2e– ................................... (1) The half equation for reduction is: Ag+(aq) + e– Ag(s) ...................................... (2) Multiplying equation (2) by 2, we obtain: 2Ag+(aq) + 2e– 2Ag(s) ............................... (3) Combining equations (1) and (3) and eliminating electrons on both sides, we obtain: Pb(s) + 2Ag+(aq) Pb2+(aq) + 2Ag(s) 0 +1 +2 0 Ag+(aq) is the oxidizing agent as the oxidation number of Ag is decreased from +1 for Ag+(aq) to 0 for Ag(s). Pb(s) is the reducing agent as the oxidation number of Pb is increased from 0 for Pb(s) to +2 for Pb2+(aq). 19.2 Balancing Redox Equations (SB p.191) Using the half equation method, write the overall ionic equations for the following reactions. State the oxidizing agent and reducing agent of the reaction, and explain your answers. (b) Reaction between acidified potassium manganate(VII) solution and iron(II) sulphate solution Answer 109 19.2 Balancing Redox Equations (SB p.191) Back (b) 110 The half equation for oxidation is: Fe2+(aq) Fe3+(aq) + e– ........................………………........ (1) The half equation for reduction is: MnO4–(aq) + 8H+(aq) + 5e– Mn2+(aq) + 4H2O(l) ............... (2) Multiplying equation (1) by 5, we obtain: 5Fe2+(aq) 5Fe3+(aq) + 5e– ....................…………………... (3) Combining equations (2) and (3) and eliminating electrons on both sides, we obtain: MnO4–(aq) + 8H+(aq) + 5Fe2+(aq) Mn2+(aq) + 4H2O(l) + 5Fe3+(aq) +7 +2 +2 +3 MnO4–(aq) is the oxidizing agent as the oxidation number of Mn is decreased from +7 for MnO4–(aq) to +2 for Mn2+(aq). Fe2+(aq) is the reducing agent as the oxidation number of Fe is increased from +2 for Fe2+(aq) to +3 for Fe3+(aq). 19.2 Balancing Redox Equations (SB p.191) Balance the following redox equation by using the oxidation number method. Cr2O72–(aq) + SO2(aq) 2Cr3+(aq) + SO42–(aq) Answer 111 19.2 Balancing Redox Equations (SB p.191) 1. Assigning oxidation numbers for the oxidizing agent, reducing agent and the products, we have: Cr2O72–(aq) + SO2(aq) 2Cr3+(aq) + SO42–(aq) +6 +4 +3 +6 2. The oxidation number of each Cr atom is decreased by 3, so the oxidation number of two Cr atoms is decreased by 6 totally. Besides, the oxidation number of S is increased by 2. O.N. increased by 2 Cr2O72–(aq) + SO2(aq) 2Cr3+(aq) + SO42–(aq) +6 +4 +3 +6 O.N. decreased by (3 2) = 6 112 19.2 Balancing Redox Equations (SB p.191) 3. Since the total increase in oxidation number of the reducing agent is equal to the total decrease in oxidation number of the oxidizing agent, Cr2O72–(aq) and SO2(aq) are in the ratio of 1 : 3. Cr2O72–(aq) + 3SO2(aq) 2Cr3+(aq) + 3SO42–(aq) 4. Balancing the number of O atoms by adding H2O(l) molecules, we have: Cr2O72–(aq) + 3SO2(aq) 2Cr3+(aq) + 3SO42–(aq) + H2O(l) 5. Balancing the number of H atoms by adding H+(aq) ions, then the balanced redox equation is obtained: Cr2O72–(aq) + 3SO2(aq) + 2H+(aq) 2Cr3+(aq) + 3SO42–(aq) + H2O(l) Back 113 19.2 Balancing Redox Equations (SB p.191) Balance the following redox equation by using the oxidation number method. (a) MnO4–(aq) + Fe2+(aq) Mn2+(aq) + Fe3+(aq) Answer 114 19.2 Balancing Redox Equations (SB p.191) (a) 1. Assigning the oxidation numbers for the oxidizing agents, reducing agents and products, we obtain: MnO4–(aq) + Fe2+(aq) Mn2+(aq) + Fe3+(aq) +7 +2 +2 +3 2. The oxidation number of Mn is decreased from +7 for MnO4–(aq) to +2 for Mn2+(aq) (i.e. decreased by 5). The oxidation number of Fe is increased from +2 for Fe2+(aq) to +3 for Fe3+(aq) (i.e. increased by 1). O.N. increased by 1 MnO4–(aq) + Fe2+(aq) Mn2+(aq) + Fe3+(aq) +7 +2 +2 +3 O.N. decreased by 5 115 19.2 Balancing Redox Equations (SB p.191) 3. Since the total increase in oxidation number of the reducing agent is equal to the total decrease in oxidation number of the oxidizing agent, MnO4–(aq) and Fe2+(aq) are in the ratio of 1 : 5. MnO4–(aq) + 5Fe2+(aq) Mn2+(aq) + 5Fe3+(aq) 4. Balancing the number of O atoms by adding H2O(l) molecules, we have: MnO4–(aq) + 5Fe2+(aq) Mn2+(aq) + 5Fe3+(aq) + 4H2O(l) 5. Balancing the number of H atoms by adding H+(aq) ions, then the following balanced equation is obtained. MnO4–(aq) + 5Fe2+(aq) + 8H+(aq) Mn2+(aq) + 5Fe3+(aq) + 4H2O(l) 116 19.2 Balancing Redox Equations (SB p.191) Balance the following redox equation by using the oxidation number method. (b) IO4–(aq) + I–(aq) I2(aq) 117 Answer 19.2 Balancing Redox Equations (SB p.191) (b) 1. Assigning the oxidation numbers for the oxidizing agents, reducing agents and products, we obtain: IO4–(aq) + I–(aq) I2(aq) +7 –1 0 2. The oxidation number of I is decreased from +7 for IO4–(aq) to 0 for I2(aq) (i.e. decreased by 7). On the other hand, the oxidation number of I is increased from –1 for I–(aq) to 0 for I2(aq) (i.e. increased by 1). O.N. increased by 1 IO4–(aq) + I–(aq) I2(aq) +7 –1 0 118 O.N. decreased by 7 19.2 Balancing Redox Equations (SB p.191) 3. Since the total increase in oxidation number of the reducing agent is equal to the total decrease in oxidation number of the oxidizing agent, IO4–(aq) and I–(aq) are in the ratio of 1 : 7. IO4–(aq) + 7I–(aq) 4I2(aq) 4. Balancing the number of O atoms by adding H2O(l) molecules, we have: IO4–(aq) + 7I–(aq) 4I2(aq) + 4H2O(l) 5. Balancing the number of H atoms by adding H+(aq) ions, then the following balanced equation is obtained. IO4–(aq) + 7I–(aq) + 8H+(aq) 4I2(aq) + 4H2O(l) Back 119