* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download 103-20b-VariationSalinitySeawater
Survey
Document related concepts
Global Energy and Water Cycle Experiment wikipedia , lookup
History of geology wikipedia , lookup
Biochemical oxygen demand wikipedia , lookup
History of climate change science wikipedia , lookup
Age of the Earth wikipedia , lookup
Geochemistry wikipedia , lookup
Soil salinity control wikipedia , lookup
Abyssal plain wikipedia , lookup
Freshwater environmental quality parameters wikipedia , lookup
Physical oceanography wikipedia , lookup
Tectonic–climatic interaction wikipedia , lookup
Anoxic event wikipedia , lookup
History of Earth wikipedia , lookup
Deep sea community wikipedia , lookup
Transcript
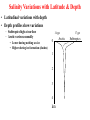
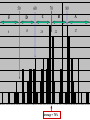
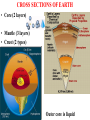
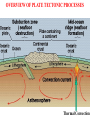
VARIATIONS IN SALINITY OF SEAWATER GEOL 1033 (Lessons 22-23, 25, and parts of Lesson 24) (ppt file 103-20b) Salinity Variations Latitude Salinity Variationswith with Latitude Salinity (ppt) Low at “meteorological equator” at about 5 º N lat. • Low• at “meteorological equator” at about 5 (=rainforests º N lat. on continents) (=rainforests on continents) • Highs at about 25 deg. N and 15 deg. S • Highs at (= about deg.belts N and 15 deg. S desert25 climatic on continents) (= desert climatic belts on continents) North-South Salinity Variations 37 35 33 60 N 40 20 0 Latitude 20 40 60 S Salinity Variations with Latitude & Depth • Latitudinal variations with depth • Depth profiles show variations – Subtropics high at surface – Arctic varies seasonally • Lower during melting sea ice • Higher during ice formation (dashes) 34 ppt 0 1 2 3 4 km Arctic 37 ppt Subtropics Major Gases in the Atmosphere and Seawater • Atmospheric gases (more soluble in colder, deeper water) are saturated at all depths except oxygen & carbon dioxide • Oxygen & carbon dioxide are involved in biological processes: – Photosynthesis – Respiration – Decay of organic matter (as HCO3-) Argon 0.94% 1.4% Oxygen & Carbon Dioxide in Seawater • Photic zone consumption of CO2 & production of O2 – Photosynthesis: CO2 + H2O organic matter + O2 • Aphotic zone respiration & decay – Produces CO2 & consumes O2 • High latitude density circulation “sinks” O2 to deep water Many Factors of Carbon Dioxide Cycle in Seawater • • • • • • • • • • • • Photosynthesis Respiration Decay of organic matter Atmospheric gas Carbonic acid Bicarbonate ion Carbonate ion Mg and Ca carbonates - inorganic precipitation Calcium carbonate skeletons (forams, clams, corals, etc.) Loss of carbon in organic matter buried in sediments C in OM in sediments often generates oil & gas deposits CCD The pH Scale • Average pH of seawater is about 7.8 • Buffered by CO2 – Prevents sudden and wide changes in pH – With a pH decrease, CaCO3 in skeletons & sediments dissolves – With a pH increase, CaCO3 precipitates Carbon Dioxide in Seawater Surface Seawater Calcium Carbonate Saturation • CO2 is more soluble in colder water • It forms carbonic acid (H2CO3) which dissociates to H+ ion & a bicarbonate ion (HCO3-) • H2CO3 dissolves CaCO3 • One H+ ion links to the CO32carbonate to form another bicarbonate (HCO3-) ion • This binding of the H+ stops seawater from becoming more acidic • Removal of CO2 gives up the H+ in HCO3- & reprecipitates CaCO3. • The freed H+ left behind lowers the pH back to normal. Earth's Ocean Surfaces north unsaturated -------------------- 30 -------equator-------- Saturated with -------------------- 30 CaCO3 unsaturated south Chlorinity • “Law of constant proportions” – Major and many minor constituents have a constant ratio between each other = “conservative” substances – Why? • Possibly due to the oceans being fairly well-mixed • Because of long residence times, especially sodium & chloride • Determine Cl- content in g/kg – The units g/kg are equivalent to ppt (o/oo) – Easier than determining all the salts – Cl- Related to the electrical conductivity & temperature • Multiply this value by the constant 1.80655 to get salinity – Example: 20.00 g/kg x 1.80655 = 36.13 o/oo total salinity – Accurate to + or – 0.005 – Determine on shipboard or onshore Residence Time • Residence times help to explain why some ions are more abundant than others Greatest Salinity Variations • Air-sea interface • Boundaries between different ocean currents • Coastal areas – Evaporative salt concentration or freshwater dilution – May 29, 1985: • Blomidon = 24.5 o/00 • Kingsport = 16.3 o/00 • Latitude • Depth Stopped here Nutrients • Less abundant than the dissolved atmospheric gases • Measure in ppm • Many are in limited supply, for example: – – – – – – Nitrite Nitrate, sometimes the “limiting factor” Ammonium Phosphate, can be the limiting factor Silica, SiO2, can be the limiting factor CaCO3, can be the limiting factor • Divergences recycle nutrients back to the photic zone: – Equatorial – Polar • Upwellings also return them to the photic zone Biological Production of Organic Matter in Present-Day World Oceans lowest lowest lowest lowest lowest Upwelling Areas = high Coastal & continental shelves = highest biological productivity Divergences END OF FILE 50 60 F D 6 15 70 80 C 28 B 22 A 27 38 39 40 41 42 43 44 45 46 47 48 49 50 51 52 53 54 55 56 57 58 59 60 61 62 63 64 65 66 67 68 69 70 71 72 73 74 75 76 77 78 79 80 81 82 83 84 85 86 87 88 89 90 91 92 93 94 Bins (percent) Average = 70% • Unit = mL/L • At 1 atm at sea level • STP = standard temperature & pressure ELEMENT ABUNDANCE IN THE CRUST and WHOLE EARTH • Abundance of elements in the crust by percentage weight: – – – – – – – – – Oxygen Silicon Aluminum Iron Calcium Sodium Potassium Magnesium all others O Si Al Fe Ca Na K Mg 47 28 8 5 3.6 3 2.6 2 1.5 • Whole Earth order of abundance: – – – – – – – – – Iron Oxygen Silicon Magnesium Nickel Sulfur Calcium Aluminum all others Fe O Si Mg Ni S Ca Al 35 30 15 13 2.4 2 1 1 <1 CHARACTERISTICS OF THE WORLD OCEAN • Oceans cover 71% of Earth's surface (This is equal to about 361 100 000 km2 or 3.611 x 108 km2) • Oceans represent about 98% of Earth's surface and near-surface water (1.37 x 109 km3) • Average depth of the oceans is about 3.8 km (~12,450'). • Average temperature of the oceans is about 4 deg. C. • Average salinity is about 34.482 o/oo (=34.482 g/kg) HEAT CAPACITY OF COMMON MATERIALS CROSS SECTIONS OF EARTH • Core (2 layers) • Mantle (3 layers) • Crust (2 types) Outer core is liquid CROSS SECTIONS OF EARTH Upper mantle/crustal layers: – – – lithosphere asthenosphere upper mesosphere OVERVIEW OF PLATE TECTONIC PROCESSES Thermal Convection GL1033x1 Test 2 Results – 9 Nov., 2004 (n=100) F D 4 15 C 33 B A 31 16 Average = 68%