* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Lesson 1 - Bonding in compounds overview
Chemistry: A Volatile History wikipedia , lookup
List of phenyltropanes wikipedia , lookup
Biochemistry wikipedia , lookup
Hydrogen-bond catalysis wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Electrochemistry wikipedia , lookup
History of chemistry wikipedia , lookup
Gas chromatography–mass spectrometry wikipedia , lookup
Debye–Hückel equation wikipedia , lookup
Electron configuration wikipedia , lookup
Drug discovery wikipedia , lookup
Halogen bond wikipedia , lookup
Organic chemistry wikipedia , lookup
Metastable inner-shell molecular state wikipedia , lookup
Bent's rule wikipedia , lookup
Molecular orbital diagram wikipedia , lookup
Hydrogen bond wikipedia , lookup
Coordination complex wikipedia , lookup
Electronegativity wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Molecular dynamics wikipedia , lookup
Resonance (chemistry) wikipedia , lookup
Atomic theory wikipedia , lookup
Inorganic chemistry wikipedia , lookup
Bond valence method wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Metalloprotein wikipedia , lookup
IUPAC nomenclature of inorganic chemistry 2005 wikipedia , lookup
Organosulfur compounds wikipedia , lookup
Homoaromaticity wikipedia , lookup
Nanofluidic circuitry wikipedia , lookup
History of molecular theory wikipedia , lookup
Ionic compound wikipedia , lookup
Metallic bonding wikipedia , lookup
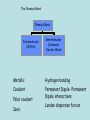
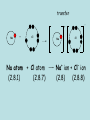
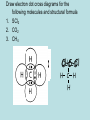
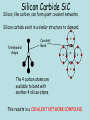
(B) Bonding and Structure Bonding in Compounds overview (C) Bonding and Structure Bonding in Compounds After completing this topic you should be able to : Overview • • Learn how the elements can form bonds in compounds and examine properties of bonding types. An introduction to the variety of intermolecular forces which exist between molecules. The Chemical Bond Chemical Bond Intramolecular (Within) Intermolecular (between) Van der Waals Metallic Hydrogen bonding Covalent Permanent Dipole- Permanent Dipole interactions Polar covalent Ionic London dispersion forces Ionic Compounds Ionic Compounds Ions - metals lose electrons and form positive ions - non-metals gain electrons to form negative ions - electrons are transferred from metals to non-metals transfer - + Na + Cl Na atom + Cl atom (2.8.1) (2.8.7) Na Cl Na+ ion + Cl- ion (2.8) (2.8.8) Ionic Compounds The positive and negative ions are attracted (electrostatic bond ) to each other. Ionic bond (electrostatic attraction) Na+ Cl- A giant lattice structure is formed. Each Na+ ion is surrounded by 6 Cl- ions. While each Cl- is surrounded by 6 Na+ ions. Ionic bonding is the electrostatic force of attractionbetween positively and negatively charged ions. This ionic network compound has many ionic bonds so ionic compounds have high m.p.s Ionic Compounds A giant lattice structure is formed when each Na+ ion is surrounded by 6 Cl- ions and each Cl- ion is surrounded by 6 Na+ ions. Sodium Chloride The formula of sodium chloride is NaCl, showing that the ratio of Na+ to Cl- ions is 1 to 1. The m.p. of NaCl is 801 0C The size of the ions will effect the strength of the ionic bond and how the ions pack together. e.g. NaF m.p. 1000oC, NaI 660oC Molecular Ions, e.g. SO4 Oxygen A single covalent bond. Sulphur O 2 additional electrons e.g. Copper can donate the extra 2 electrons needed. Cu Cu 2+ + 2e O S O O Copper sulphate contains the Cu2+ and the SO42- ions. There is, therefore, covalent bonding and ionic bonding in copper sulphate A solution of copper sulphate can conduct electricity. Molten ionic compounds can also conduct electricity. Bond Strengths Bond Type Strength (kJ mol –1) Metallic 80 to 600 Ionic 100 to 500 Covalent 100 to 500 Hydrogen 40 Dipole-Dipole 30 London’s forces 1 to 20 Covalent Molecular Compounds Covalent Bonding Sharing electrons • takes place between non-metal and non-metal • shared electrons count as part of the outer shell of both Atoms • shared electrons attract the nuclei of both atoms • this attraction is called the covalent bond Hydrogen chloride H H Cl (linear) Cl HCl Ammonia H H H N H H (pyrimidal) NH3 H N Water O H H (bent) H2O H O H Draw electron dot cross diagrams for the following molecules and structural formula 1. SCl2 2. CO2 3. CH4 X X S X X H Cl-S-Cl O=C=O H C H H Bond Strengths Bond Type Strength (kJ mol –1) Metallic 80 to 600 Ionic 100 to 500 Covalent 100 to 500 Hydrogen 40 Dipole-Dipole 30 London’s Forces 1 to 20 Covalent Molecular Compounds Discrete molecules are formed when two or more atoms share electrons. The atoms are non-metal elements. An example is methane. Methane: CH4 H H H C H H H C H H Methane has strong intra-molecular and weak inter-molecular. It’s b.p. is -183oC Covalent Molecular Compounds Non- metals elements can form double and triple covalent bonds. ethane C2H6 H H H C C H H H ethene C2H4 H H H C C H H H H H C C H H H H C C H H H C Double covalent bond Covalent molecular compounds have low m.p.’s because the weak forces holding the molecules together require only small amounts of thermal energy to break them. Covalent Molecular Compounds Properties Low m.p.’s and b.p.’s., this increases with size of the molecule and the increasing number of atoms in the molecule. m.p.’s of the carbon halides 171 Temp / oC 90 -23 -183 CF4 CCl4 CBr4 CI4 m.p.’s increase because the strength of the London dispersion forces increase with the increasing size of the molecule. So more Energy is needed to separate molecules. Covalent Network Compounds Silicon Carbide SiC Silicon, like carbon, can form giant covalent networks. Silicon carbide exist in a similar structure to diamond. Tetrahedral shape C Covalent Bond C Si C C The 4 carbon atoms are available to bond with another 4 silicon atoms. This results in a COVALENT NETWORK COMPOUND Silicon Carbide SiC Silicon carbide (carborundum) has a chemical formula is SiC. As this compound is linked by strong covalent bonding, it has a high m.p. (2700oC). It is a hard substance as it is very difficult to break the covalent lattice. SiC is used as an abrasive for smoothing very hard materials. Each Si is bonded to 4 C’s and each C is bonded to 4 Si’s. Hence the chemical formula, SiC Silicon Dioxide SiO2 Silicon and oxygen make up nearly 75% of the Earth’s crust. They are therefore the most common elements in the Earth’s crust. They combine together to make a covalent network compound called silicon dioxide. This is usually found in the form of sand or quartz. Each Si atom is bonded to 4 O atoms, and each O atom is bonded to 2 Si atoms. Hence the chemical formula, SiO2 . Silicon dioxide (silica) also has a high m.p. (1610 oC) and like SiC, it is very hard and used as an abrasive. It is relatively un-reactive. New Higher Chemistry E Allan J Harris