* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Phase I
Polysubstance dependence wikipedia , lookup
Discovery and development of beta-blockers wikipedia , lookup
Drug interaction wikipedia , lookup
Pharmacognosy wikipedia , lookup
Drug discovery wikipedia , lookup
Clinical trial wikipedia , lookup
Adherence (medicine) wikipedia , lookup
Psychopharmacology wikipedia , lookup
Drug design wikipedia , lookup
Pharmacokinetics wikipedia , lookup
Pharmaceutical industry wikipedia , lookup
Prescription costs wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Neuropharmacology wikipedia , lookup
NK1 receptor antagonist wikipedia , lookup
Pharmacogenomics wikipedia , lookup
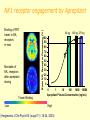
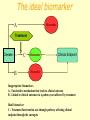
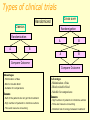
Phase I Healthy volunteers Required steps • Clinical protocol preparation • Informed consent • Approval of the ethical committee Objectives: 1 - To identify a safe & tolerated dose (MTD) 2 - To compare PK profile in man versus animal/s 3 - To see if compound show proper absorption, bioavailability 4 -To detect effects unrelated to the expected action 5 -To detect any predictable toxicity Phase I Methods: 1. Inclusion criteria • Healthy volunteers : Uniformity of subjects: age, sex, nutritional status Exception: Patients only for toxic drugs Eg AntiHIV, Anticancer 2. Exclusion criteria – Pregnant women 3. First in a small group of 20 to 25 • • Start with a dose of about 1/100 NOAEL animal dose Slowly increase the dose to find safe tolerated dose (stop at 1/5 animal NOAEL) • 4. 5. 6. 7. 8. If safe in a larger group of up to about 50 –75 No blinding Performed by clinical pharmacologists Centre has emergency care & facility for kinetics study Performed in a single centre Takes 3 – 6 months Phase IIA and B Objectives • Detailed PK & PD in patients • Establish doses for future Ph III trials • Efficacy against a defined therapeutic endpoint • Therapeutic benefits Methods First in patient Performed by Clinicians in the hospital Few centres IIA [20 – 200 patients with relevant disease] PK in patients: Establish a dose range to be used in late phase Maximum tolerated dose and Safety e AEs Method: same as phase I IIB patients [ 50 – 500] Double blind Compared with a placebo or standard drug Takes 6 months to 2 years (35% of success) Proof of concept (POC) Phas eI Phas e IIA Phas e IIB PhII I Proo f of Con cept (Po C) • May be considered as Phase IIA or IIB or can occur sometime between Phase IIA and Phase IIB • Decision based on studies specifically chosen to demonstrate or “prove” the drug will be efficacious and safe in Phase III • These studies are mainly of two types: – Target engagement – Effect on disease biomarkers Target engagement biomarker In vivo receptor occupancy in human: - PET technology Is the most selective and sensitive (pico- to nanomolar range) method for measuring receptor density and interactions in vivo. Very important tool for translation from preclinical to clinical research - In vivo radioligand binding - Ex vivo radioligand binding PET POSITRON EMISSION TOMOGRAPHY Binding of PET tracer to NK1 receptors in man Blockade of NK1 receptors after aprepitant dosing Tracer Binding Low Brain NK1 Receptor Occupancy (%) NK1 receptor engagement by Aprepitant 40 mg 125 mg 375 mg 100 90 80 70 60 50 40 30 20 10 0 0 1 10 100 1000 10000 Aprepitant Plasma Concentration (ng/mL) High (Hargreaves J Clin Psych 63: (suppl 11): 18-24, 2003) BIOMARKER Characteristics of a “good” biomarker: - Biological feasibility - Dose-related response to intervention - Easy to measure - Reproducible, specific and sensitive with high predictive value - Acceptable by experts - Acceptable by Regulatory Authorities The ideal biomarker Biomarker A Treatment Disease C B Biomarker Clinical Endpoint Biomarker Inappropriate biomarkers A - Unrelated to mechanism that leads to clinical outcome B - Linked to clinical outcome via a pathway not affected by treatment Ideal biomarker C - Treatment Intervention acts through pathway affecting clinical endpoint through the surrogate Phase III: establishing the therapeutic value Objectives • Confirm doses and efficacy established in Phase II • Verify possible interaction with other drugs in a large population of patients • Identify possible patient subpopulations • Compare effect with that of drugs in the market Methods - Performed by Clinicians in the hospital - Large scale (500 – 2000 patients) - Multicentric Ensures geographic & ethnic variations - Diff patient subgroups Eg pediatric, geriatric, renal impaired - Randomised allocation of test drug /placebo / standard drug - Double blinded - Cross over design - Vigilant recording of all adverse drug reactions - Rigorous statistical evaluation of all clinical data • Takes a long time: up to 5 years [25% success] Types of clinical trials Cross over Randomized Classics Randomization Randomization A A B B A B Compare Outcome Compare Outcome Advantages - Minimization of bias - Blind or double blind - Suitable for comparisons Issues - Half of the patients do not get the treatment - High number of patients to minimize outliers - Time and resource consuming Advantages - Minimization of bias - Blind or double blind - Suitable for comparisons Issues - High number of patients to minimize outliers - Time and resource consuming - Potential risk of sinergy between treatment Registration • Each state has his own regulatory agency and rules may different from state to state • Efficacy and safety are the major concerns • Data and protocols are carefully analyzed • Support documentation could be cumbersome: 50-400 volumes (30,000-150,000 pages) • The process require a continuous dialogue between regulatory agency and pharmaceutical company • If the drug is approved it will be labelled for a single indication Phase IV:Marketing e postmarketing surveillance • Adequate commercialization of the product • Explore the possibility to use the drug for additional therapeutic indications • Enlarge the knwoledge about the potential side effects • Evaluate the ratio between benefit and ling term adverse effects. P2Y12 antagonists on the market and in clinical trials Two currently FDA-approved P2Y12 antagonists: ticlopidine and clopidogrel are thienopyridines. The active compound is a metabolite of this thienopyridine and is an irreversible inhibitor. Novel P2Y12 antagonists under development, have a faster onset of action, as well as more potent, and less variable, inhibition of platelet function ex vivo. Compassioned used • The term "compassionate use" refers to the treatment of a seriously ill patient using a new, unapproved drug • Requirements: – – – – Drugs that have shown activity in phase III No other treatments are available Simplified protocols It is necessary the informed consent Abbreviated new drug application This procedure is applicable only for very seriuos pathology It is applied to molecules that could represent a significant improvement other existing therapy. To have access to this procedure there are two major requirements: - Safety of the patient is guarantee by the use of the drug in controlled condition - There are compelling laboratory data indicating a clear efficacy