* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download smart_materials_1 - Aldercar High School
Pseudo Jahn–Teller effect wikipedia , lookup
Materials science wikipedia , lookup
Molecular orbital diagram wikipedia , lookup
Electrochemistry wikipedia , lookup
Multiferroics wikipedia , lookup
Inductively coupled plasma mass spectrometry wikipedia , lookup
Biochemistry wikipedia , lookup
Size-exclusion chromatography wikipedia , lookup
Strengthening mechanisms of materials wikipedia , lookup
Self-assembled monolayer wikipedia , lookup
Aromaticity wikipedia , lookup
Metastable inner-shell molecular state wikipedia , lookup
Coordination complex wikipedia , lookup
Evolution of metal ions in biological systems wikipedia , lookup
Molecular graphics wikipedia , lookup
Physical organic chemistry wikipedia , lookup
Nanofluidic circuitry wikipedia , lookup
Computational chemistry wikipedia , lookup
Electron configuration wikipedia , lookup
Atomic theory wikipedia , lookup
Shape-memory alloy wikipedia , lookup
Condensed matter physics wikipedia , lookup
Rutherford backscattering spectrometry wikipedia , lookup
Nanochemistry wikipedia , lookup
Resonance (chemistry) wikipedia , lookup
Hypervalent molecule wikipedia , lookup
State of matter wikipedia , lookup
Molecular scale electronics wikipedia , lookup
Metallic bonding wikipedia , lookup
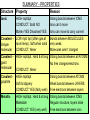
SUMMARY - Descriptions IONS ONLY IONIC Crystals Dissolve in water eg sodium chloride (salt) SIMPLE MOLECULAR Usually Gases eg CO2, H2O MOLECULES ONLY METALLIC Strong malleable solids IONS + Don’t dissolve FREE eg copper ELECTRONS ATOMS joined into GIANT MOLECULE S GIANT MOLECULAR Hard strong solids Don’t dissolve eg diamond (graphite – special case) SUMMARY - PROPERTIES Structure Property Reason Ionic HIGH mpt/bpt CONDUCT: Solid NO Molten YES Dissolved YES Strong bonds between IONS Ions can’t move Ions can move to carry current Covalent – LOW mpt/ bpt (often gas at room temp). Soft when solid Simple molecular CONDUCT: Never Bonds between MOLECULES very weak. Molecules aren’t charged Covalent – HIGH mpt/bpt. Hard & strong Strong bonds between all ATOMS giant No free charges/electrons molecular CONDUCT: Never Covalent graphite HIGH mpt/bpt Soft & slippery CONDUCT: YES (fairly well) Strong bonds between ATOMS Weak bonds between LAYERS Free electrons between layers Metallic HIGH mpt/bpt. Hard & strong Strong bonds between IONS Malleable Regular structure, layers slide CONDUCT: YES (very well) Free electrons between ions Structure of an Alloy 1. Draw this picture. 2. What 2 things are obvious from it? Different size atoms Irregular structure This makes alloys HARDER than pure metals. Smart Materials Smart materials' 'are materials that have one or more properties that can be significantly changed in a controlled fashion by external stimuli, such as stress, temperature, moisture, pH, electric or magnetic fields. Smart Liquids When a magnetic field is applied to the fluid it becomes semi-solid and when the field is removed, the fluid returns to a free-flowing liquid. • http://www.youtube.com/watch?v=kL8R8SfuXp8 Shape Memory Alloys A shape memory alloy is an alloy that "remembers" its original, cold, forged shape, and which returns to that shape after being deformed by applying heat. http://www.youtube.com/ watch?v=fsBHF_j2FJ4 Exam Question Nanomaterials Are very small but how small? Human hair 9 micrometers is equivalent to 0.000009 m 40 microns 9 microns 0.000000009 m is 9 nm or 0.009 micrometers 0.25 nanometers is the average length of a bond between atoms!!! Very Small Can Make Properites Very Different Some Potential Uses • • • • • • • • Filtration Catalysis Tissue engineering Drug delivery Diagnostics Memory storage Quantum computers Optoelectronics Quantum dots Nanotubes • Cylindrical carbon molecules have novel properties that make them potentially useful in many applications in nanotechnology, electronics, optics and other fields of materials science, as well as potential uses in architectural fields. They exhibit extraordinary strength and unique electrical properties, and are efficient thermal conductors. Their final usage, however, may be limited by their potential toxicity and controlling their property changes in response to chemical treatment.