* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Tetracyclines
Drug design wikipedia , lookup
Discovery and development of neuraminidase inhibitors wikipedia , lookup
Psychopharmacology wikipedia , lookup
Discovery and development of tubulin inhibitors wikipedia , lookup
Toxicodynamics wikipedia , lookup
Drug discovery wikipedia , lookup
Pharmacognosy wikipedia , lookup
Metalloprotease inhibitor wikipedia , lookup
Drug interaction wikipedia , lookup
Discovery and development of non-nucleoside reverse-transcriptase inhibitors wikipedia , lookup
Neuropharmacology wikipedia , lookup
Discovery and development of integrase inhibitors wikipedia , lookup
Neuropsychopharmacology wikipedia , lookup
Discovery and development of cephalosporins wikipedia , lookup
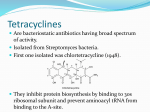
Chapter 39 Tetracyclines (四环素)& Chloramphenicol(氯霉素) Tetracyclines Tetracyclines -Chemical structure •Tetracycline (四环素) Tetracyclines • Clinical used tetracyclines: • Tetracycline(四环素); • Demeclocycline(地美环素, 去甲金霉 素); • Metacycline(美他环素, 甲烯土霉素); • Doxycycline(多西环素, 强力霉素); • Minocycline(米诺环素, 美满霉素). (Antimicrobial activity enhanced from up to down) Tetracyclines -Overview Tetracyclines Crude product Tetracycline(四环素),Chlortetracycline (金霉素),Oxytetracycline (土霉素) Semisynthetic derivative Doxycycline(多西环素), Minocycline(米诺环素) Tetracyclines Antimicrobial activity (1) Bacteriostatic (2) Bactericidal (at high concentration ) (3) Minocycline > Doxycycline > Tetracycline Tetracyclines Antimicrobial spectrum Broad-spectrum antibiotic (1) Active against a wide range of aerobic and anaerobic gram-positive and gram-negative bacteria. (2) Effective against Rickettsia(立克次体),Coxiella burnetii(螺旋体),Mycoplasma pneumoniae(支原 体),Chlamydia (衣原体), and Plasmodium (疟原虫). (3) They are not active against fungi,virus. Tetracyclines Mechanism of action (1) Enter bacteria by passive diffusion through the protein channel formed by porin proteins of outer cell membrane(G- organisms) and active transport by an energy-dependent system that pumps all tetracyclins across cytoplasmic membrane (G+ organisms) . (2) Inhibit protein synthesis in susceptible microorganisms. (3) Increase the permeability of the cell membrane Mechanism of resistance Tetracyclines Mechanism of action Tetracyclines Mechanism of action: ①Chloramphenicol ②Macrolides, Clindamycin ③Tetracyclines •Inhibits binding of 30S subunit with A site •Interfering with the binding of aminoacyl-tRNA with aminoacyl site(A site) Mechanism of resistance Tetracyclines (1) Decreased intracellular accumulation due to either impaired influx or increased efflux by a active transport protein pump. (2) Ribosome protection that interfere with the tetracycline binding to the ribosome. (3) Enzyme inactivation of tetracycline. Tetracyclines ADME (1) Absorption are impaired by food (except doxycycline and minocycline ) • Tetracyclines manly differ in their absorption after oral administration. (2) Distributed widely to tissue and body fluid except for CSF. (3) Excreted mainly in bile and urine. ADME Tetracyclines (4) Tetracyclines across the placenta and are also excreted in the milk. (5) Bound to- and damage- growing bones and teeth • As a result of chelation with calcium Tetracyclines Clinical Uses (1)Rickettsial(立克次体) infections. (2)Mycoplasma(支原体) infections. (3)Chlamydia(衣原体) infection. (4)Leptospira(螺旋体) infection. (5)Bacterial infection. Tetracyclines Adverse reaction (1)Gastrointestinal effects. (2)Superinfections. (3) Bony Structures and Teeth: Deposition of the drugs in growing teeth and bones. (4)Hepatic toxicity and renal toxicity. (5)Photosensitivity. (6)Pseudotumer cerebri(脑假瘤) (7)Vestibular reaction(前庭反应 ) Tetracyclines Recent research •Tetracycline and its derivatives doxycycline and minocycline were found to have antiinflammatory and anti-apoptotic properties. •Protect mice from brain ischemia, traumatic brain injury, Huntington’s disease, etc. •The mechanism partially though caspase-1, 3, iNOS, COX-2 etc. Tetracyclines • A newly approved tetracycline analog, tigecycline, is a glycylcycline and a semisynthetic derivative of minocycline. Tetracyclines • Tigecycline, the first glycylcycline to reach the clinic, has several unique features that warrant its consideration apart from the older tetracyclines. Many tetracycline-resistant strains are susceptible to tigecycline because the common resistance determinants have no activity against it. Its spectrum is very broad. Coagulase-negative staphylococci and Staphylococcus aureus, including methicillin-resistant, vancomycin-intermediate, and vancomycin-resistant strains; streptococci, penicillin susceptible and resistant; enterococci, including vancomycin-resistant strains; gram-positive rods; Enterobacteriaceae; multidrug-resistant strains of Acinetobacter sp; anaerobes, both gram-positive and gramnegative; atypical agents, rickettsiae, chlamydia, and legionella; and rapidly growing mycobacteria all are susceptible. Proteus and P aeruginosa, however, are intrinsically resistant. Tetracyclines • • Tigecycline, formulated for intravenous administration only, is given as a 100-mg loading dose; then 50 mg every 12 hours. As with all tetracyclines, tissue and intracellular penetration is excellent; consequently, the volume of distribution is quite large and peak serum concentrations are somewhat blunted. Elimination is primarily biliary, and no dosage adjustment is needed for patients with renal insufficiency. In addition to the tetracycline class effects, the chief adverse effect of tigecycline is nausea, which occurs in up to one third of patients, and occasionally vomiting. Neither nausea nor vomiting usually requires discontinuation of the drug. Tigecycline is FDA-approved for treatment of skin and skin-structure infection and intra-abdominal infections. Because active drug concentrations in the urine are relatively low, tigecycline may not be effective for urinary tract infections and has no indication for this use. Because it is active against a wide variety of multidrug-resistant nosocomial pathogens (eg, methicillinresistant S aureus, extendedspectrum -lactamase-producing gram-negatives, and Acinetobacter species), tigecycline is a welcome addition to the antimicrobial drug group. Chloramphenicol Chloramphenicol (氯霉素) Chemical structure p 1246 p776pharm Chloramphenicol Antimicrobial activity (1)Chloramphenicol possesses a wide antimicrobial spectrum. (2) Primarily bacteriostatic , although it may be bactericidal to certain species. Chloramphenicol Mechanism of action 1. Inhibit protein synthesis in susceptible bacteria, and to a lesser extent, in mammalian cell 2. Acts primarily by binding reversibly to the 50 S ribosomal subunit • Near the site of action of macrolides and clindamycin, which it inhibits competitively). Mechanism of action Chloramphenicol Mechanism of action: ①Chloramphenicol ②Macrolides, Clindamycin ③Tetracyclines Mechanism of Resistance Chloramphenicol (1) Resistance usually caused by a plasmid-encoded acetyltransferaes(乙 酰转移酶) that inactive the drugs. (2) The permeability of bacterial cell membrane is changed. Clinical uses Chloramphenicol (1) Bacterial meningitis. (2) Typhoid fever(伤寒) and other types of systemic Salmonella infections. (3) Eye bacterial infection. (4) Anaerobic infection. (5) Rickettsial disease (立克次体病) and brucellosis (布鲁杆菌病), etc. Adverse reactions Chloramphenicol (1) Gastrointestinal Disturbances (2) Bone Marrow Disturbances: • By dose-related toxic effect that presents anemia, leukopenia and or thrombocytopenia (血小板减少) • By special response manifested by aplastic anemia, leading in many cases to fatal pancytopenia (全血细胞减少). Adverse reactions Chloramphenicol (2) Toxicity for newborn infants: • Gray baby syndrome(灰婴综合征). (3)other reactions: • hypersensitivity reaction, etc. Drugs interactions Chloramphenicol • Inhibits hepatic microsomal cytochrome P450 enzyme, and thus may prolong the t 1/2 of drugs that metabolized by this system, e.g. warfarin, phenytoin, etc.