* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download MS PowerPoint
Cracking (chemistry) wikipedia , lookup
Enantioselective synthesis wikipedia , lookup
Strychnine total synthesis wikipedia , lookup
Fischer–Tropsch process wikipedia , lookup
Tiffeneau–Demjanov rearrangement wikipedia , lookup
Ring-closing metathesis wikipedia , lookup
Kinetic resolution wikipedia , lookup
Petasis reaction wikipedia , lookup
Nucleophilic acyl substitution wikipedia , lookup
Asymmetric hydrogenation wikipedia , lookup
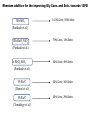
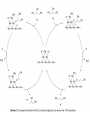
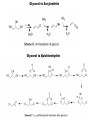
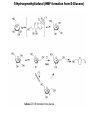
Renewable Chemicals: Dehydroxylation of Glycerol and Polyols DOI 10.1002/cssc.201100162 Arundhathi RACHA Department of Materials Engineering Science, Graduate School of Engineering Science, Toyonaka, Osaka University- Osaka 560-8531, Japan Renewable Chemicals: Dehydroxylation of Glycerol and Polyols (Jeroen ten Dam and Ulf Hanefeld) (ChemSusChem 2011,4,1017-1034; DOI 10.1002/cssc.201100162) Renewable chemicals from natural resources e.g, sugars, cellulose and hemicellulose in a sustainable way. Dehydroxylation, a fundamentally different approach of defunctioanlize of hydroxyl groups to yield renewable chemicals. Defunctionalizing of biomass vs. functionalizing of oil Design of catalysts that can selectively remove some of the functionalities . Selective conversion of polyols, i.e dehydration and hydrogenation. Selective dehydroxylation of Biomass Six approaches for deoxgenation 1. Dehydration of vicinal diols and hydrogenation of carbonyl groups 2. Dehydration of alcohols and hydrogenation of carbon–carbon double bonds 3. Condensation of alcohols and hydrogenolysis of the resulting cyclic ethers 4. Hydrogenolysis of ethers 5. Ketonization of carboxylic acids 6. Hydrogenation of carboxylic acids Selective dehydroxylation of polyols Acid-catalyzed E1 mechanism Base-catalyzed E2 mechanism Hydrogenolysis Sequential elimination (dehydration) and hydrogenation process. Endothermic (dehydration) versus exothermic (hydrogenation) –product selectivity. Need for design the catalyst which directs the overall process to occur via one specific dehydration pathway. Dehydration -Hydrogenation Dehydration selectivity determining the value-added chemicals from glycerol Bronsted acid assist in eliminating secondary hydroxyl group Lewis acid assist in eliminating primary hydroxyl group Dehydration of alcohols and hydrogenation of carbon–carbon double bonds Condensation of alcohols and hydrogenolysis of the resulting cyclic ethers The reaction sequence is not applicable to glycerol, since the condensation of alcohols in order to form cyclic ethers is limited to five-membered (or larger) ring systems. Glycerol to 1,2-propanediol Under alkaline conditions, 12PD is being formed via glyceraldehyde Under acidic conditions, acetol is generally is the key intermediate !,2-propanediol from Glycerol Glycerol to 1,3-propanediol Selective production of 13PD is more challenging than 12PD Active catalyst for 12PD production, selective catalyst for 13PD production The factors that are important to influence 13PD formation are slowly being unraveled Effect of impregnated metals and solvent on WO3/ZrO2 support for 13-PD selectivity Protic solvent improves the reaction towards 13PD by facilitating proton transfer from solid acid to secondary alcohol by stabilizing a charged intermediate. Additives improving the conversion and selctivity towards 13PD Pt/WO3/ZrO2 facilitates the homolytic cleavage of hydrogen on platinum and subsequent spillover Tungsten additive for the improving Gly Conv and Selc towards 13PD Fixed bed reactor Pt/WO3/ZrO2 403 K 70% Conv.; 46% Selec. (Quin et al.) Pt/WO3/TiO2 (Gong et al.) 7.5% Conv.; 44% Selec. Pt/WO3/TiO2/SiO2 (Gong et al.) 15% Conv.; 51% Selec. Cu-STA/SiO2 70% Conv.; 46% Selec. (Huang et al.) Rhenium additive for the improving Gly Conv. and Selc. towards 13PD Rh/SiO2 14.3% Conv.; 9.8% Selec. (Furikado et al.) Rh-ReOx/SiO2 79% Conv.; 14% Selec. (Furikado et al.) Ir-ReOx/SiO2 50% Conv.; 49% Selec. (Furikado et al.) Pt-Re/C 20% Conv.; 34% Selec. (Daniel et al.) Pt-Re/C (Tomishige et al.) 45% Conv.; 29% Selec. Glycerol to acrolein Glycerol to Lactic acid Glycerol to Acrylonitrile Glycerol to Epichlorohydrin Glycerol to Ethylene glycol Longer Chain polyols 5-Hydroxymethylfurfural (HMF formation form D-Glucose) Conclusions 1. The smallest polyol Glycerol can be deoxygenated into a range of useful chemicals . 2. Major deoxygenation products from Glycerol are 12PD, 13PD and acrolein. 3. The catalyst for acrolein formation might be a good lead for new catalyst development for 13Pd, as both share the same intermediate. 4. The process can also be applied for long chain polyols for the production of important derivative like 5-HMF. References: Please refer to Renewable chemicals: Dehydroxylation of Glycerol and Polyols Jeroen ten Dam and Ulf Hanefeld ChemSusChem 2011, 4, 1017-1034; DOI: 10.1002/cssc.201100162