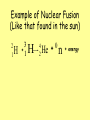* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download Nuclear Fission & Fusion
Nuclear and radiation accidents and incidents wikipedia , lookup
Muon-catalyzed fusion wikipedia , lookup
Valley of stability wikipedia , lookup
Nuclear fission product wikipedia , lookup
Nuclear binding energy wikipedia , lookup
Nuclear fission wikipedia , lookup
Nuclear fusion wikipedia , lookup
Nuclear transmutation wikipedia , lookup
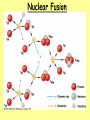
Nuclear Fission & Fusion History: Hahn & Strassman (1939) •Bombarded Uranium-235 samples with neutrons expecting the Uranium-235 to capture neutrons •Instead, the products showed different chemical properties that they could not explain Meitner & Frisch •Explained Hahn & Strassman results. •Instead of heavier Uranium, it had split into smaller elements = Nuclear Fission Nuclear Forces same 1. Electric repulsion – ________ charge particles repel each other 2. Strong Nuclear Force – causes protons and attract each other neutrons to ________ greater •Stable Nuclei = strong nuclear force is ________ than repulsion force •Unstable Nuclei = strong nuclear force is less ________ than repulsion force oHave too many or too few neutrons in nucleus oHave more than 83 protons in nucleus oWill decay into a more stable nucleus Uranium Enrichment • Uranium is found as uranium oxide which when purified has a rich yellow color and is called "yellowcake". After reduction, the uranium must go through an isotope enrichment process. Even with the necessity of enrichment, it still takes only about 3 kg of natural uranium to supply the energy needs of one American for a year. Nuclear Fission – splitting of heavier nuclei into lighter nuclei. 235 92 U 1 + 0 n 137 56 Ba 84 + 36 Xe 1 +15 0 + energy How much energy? E=mc2 _______= Energy _______ mass x (_____ speed of _ _____) light 2 n c=3.0x108 mass defect E=mc2 explains _____ _____ (total mass of nucleus is less than sum of individual particles) Nuclear Chain Reactions: •Nuclear fission releases more neutrons which trigger more fission reactions •The number of ________ neutrons released determines the success of a chain reaction Applications of Controlling Chain Reactions 1. Atomic Bomb (fission bomb) – Triggering a chain reaction in U-235 or Pu-239 Must have a minimum amount of radioactive isotope CRITICAL MASS to sustain a chain reaction =_________ _______ 2. Nuclear Power Plants – Convert heat energy from fission electricity chain reaction into __________. control _____ rods that Control chain reaction with ________ absorb ________ neutrons emitted after fission reaction. Nuclear Reactor – Images Fukushima – March 2011 Video on Fukushima The Atomic Bomb Nuclear Fission Nuclear Fusion - Energy released when two light nuclei combine or fuse •However, a large amount of energy is required to start a fusion reaction: repulsion forces o Need this energy to overcome ________ of protons. o Extremely high temperatures can provide start-up energy. More energy in fusing hydrogen that fission of uranium Example of Nuclear Fusion (Like that found in the sun) 2 1 H 3 + 1 4 2 H He 0 + n + energy Nuclear Fusion Stars energy is produced through fusion reactions Fusion occurs until Fe is produced because less energy is released than required to fuse Fe nuclei = Star____ burns____ out _____ Cold Fusion: Efforts are being made to start and sustain a fusion reaction at lower temperatures, in other words with a lower amount of input energy Hydrogen Bomb (H-Bomb) Ivy Mike -- 1952