Neutron Cross Section Standards for the Energy Region Above 20
... polarized neutron beam in addition a polarized hydrogen target. For completeness, some representative neutron-proton capture data are included. ...
... polarized neutron beam in addition a polarized hydrogen target. For completeness, some representative neutron-proton capture data are included. ...
B. - Physicsland
... 2 minutes 5 minutes 9 minutes Explanation: This question would be appropriate with Appendix D, Exponential Growth and Doubling Time. Can you see that working backward, each minute has half the number of neutrons? ...
... 2 minutes 5 minutes 9 minutes Explanation: This question would be appropriate with Appendix D, Exponential Growth and Doubling Time. Can you see that working backward, each minute has half the number of neutrons? ...
INTRODUCTORY NUCLEAR PHYSICS
... the current era (the recently discovered “heavy” decay modes, such as 14C, double P decay, P-delayed nucleon emission, Mossbauer effect, and so on). The third unit surveys nuclear reactions, including fission and fusion and their applications. The final unit deals with topics that fall only loosely ...
... the current era (the recently discovered “heavy” decay modes, such as 14C, double P decay, P-delayed nucleon emission, Mossbauer effect, and so on). The third unit surveys nuclear reactions, including fission and fusion and their applications. The final unit deals with topics that fall only loosely ...
Chm 1
... b. masses. d. numbers of electrons. ____ 14. All isotopes of hydrogen contain a. one neutron. c. one proton. b. two electrons. d. two nuclei. ____ 15. The atomic number of oxygen, 8, indicates that there are eight a. protons in the nucleus of an oxygen atom. b. oxygen nuclides. c. neutrons outside t ...
... b. masses. d. numbers of electrons. ____ 14. All isotopes of hydrogen contain a. one neutron. c. one proton. b. two electrons. d. two nuclei. ____ 15. The atomic number of oxygen, 8, indicates that there are eight a. protons in the nucleus of an oxygen atom. b. oxygen nuclides. c. neutrons outside t ...
Chapter 18
... produced by different types of radioactive decay, and write equations for nuclear decays. • Identify examples of nuclear fission, and describe potential benefits and hazards of its use. ...
... produced by different types of radioactive decay, and write equations for nuclear decays. • Identify examples of nuclear fission, and describe potential benefits and hazards of its use. ...
E = mc 2 - Gordon State College
... • is the rate of decay for a radioactive isotope. • is the time required for half of an original quantity of an element to decay. • is constant and independent of any physical or chemical change the atom may undergo. • can be calculated at any given moment by measuring the rate of decay of a known q ...
... • is the rate of decay for a radioactive isotope. • is the time required for half of an original quantity of an element to decay. • is constant and independent of any physical or chemical change the atom may undergo. • can be calculated at any given moment by measuring the rate of decay of a known q ...
A. Alpha particle. - MDC Faculty Web Pages
... • is the rate of decay for a radioactive isotope. • is the time required for half of an original quantity of an element to decay. • is constant and independent of any physical or chemical change the atom may undergo. • can be calculated at any given moment by measuring the rate of decay of a known q ...
... • is the rate of decay for a radioactive isotope. • is the time required for half of an original quantity of an element to decay. • is constant and independent of any physical or chemical change the atom may undergo. • can be calculated at any given moment by measuring the rate of decay of a known q ...
transmutation of nuclides
... Apparent radioactivity of a radioactive nuclide mixture differs from that of consecutive decay and growth. As we shall discuss later, some nuclides have more than one mode of decay. For example, a sample of 237Ac is a source of alpha and beta particles, and this type is often called branch decay. Th ...
... Apparent radioactivity of a radioactive nuclide mixture differs from that of consecutive decay and growth. As we shall discuss later, some nuclides have more than one mode of decay. For example, a sample of 237Ac is a source of alpha and beta particles, and this type is often called branch decay. Th ...
Radioactive Decays – transmutations of nuclides
... Apparent radioactivity of a radioactive nuclide mixture differs from that of consecutive decay and growth. As we shall discuss later, some nuclides have more than one mode of decay. For example, a sample of 237Ac is a source of alpha and beta particles, and this type is often called branch decay. Th ...
... Apparent radioactivity of a radioactive nuclide mixture differs from that of consecutive decay and growth. As we shall discuss later, some nuclides have more than one mode of decay. For example, a sample of 237Ac is a source of alpha and beta particles, and this type is often called branch decay. Th ...
1ST SEM MT CHAP 22 REVIEW
... OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: ...
... OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: OBJ: ...
radioactivity and radioactive decay - rct study guide
... Following a transformation the nucleus is usually more stable than it was, but it may not be completely stable. So, another transformation will take place in which the nucleus will again emit radiation. The amount of energy given off and the type of emission that occurs will depend on the configurat ...
... Following a transformation the nucleus is usually more stable than it was, but it may not be completely stable. So, another transformation will take place in which the nucleus will again emit radiation. The amount of energy given off and the type of emission that occurs will depend on the configurat ...
chem pre ap atom and nuclear practice test
... c. of each element are identical in size, mass, and other properties. d. of different elements cannot combine. 2. Which of the following statements is true? a. Atoms of the same element may have different masses. b. Atoms may be divided in ordinary chemical reactions. c. Atoms can never combine with ...
... c. of each element are identical in size, mass, and other properties. d. of different elements cannot combine. 2. Which of the following statements is true? a. Atoms of the same element may have different masses. b. Atoms may be divided in ordinary chemical reactions. c. Atoms can never combine with ...
DOE FUNDAMENTALS HANDBOOK NUCLEAR PHYSICS AND REACTOR THEORY Volume 1 of 2
... for use by DOE category A reactors. The subject areas, subject matter content, and level of detail of the Reactor Operator Fundamentals Manuals were determined from several sources. DOE Category A reactor training managers determined which materials should be included, and served as a primary refere ...
... for use by DOE category A reactors. The subject areas, subject matter content, and level of detail of the Reactor Operator Fundamentals Manuals were determined from several sources. DOE Category A reactor training managers determined which materials should be included, and served as a primary refere ...
Nuclear Physics Fundamental and Application Prof. H. C. Verma
... nuclear, a particle is coming here and then it is getting deviated, this is that hyperbolic path. You know all that geometry can be done, here I am interested in finding this minimum separation here, minimum separation. So, if you write the whole equation, I do not need that, but still the equation ...
... nuclear, a particle is coming here and then it is getting deviated, this is that hyperbolic path. You know all that geometry can be done, here I am interested in finding this minimum separation here, minimum separation. So, if you write the whole equation, I do not need that, but still the equation ...
Nuclear Physics 1 NWNC
... for use by DOE category A reactors. The subject areas, subject matter content, and level of detail of the Reactor Operator Fundamentals Manuals were determined from several sources. DOE Category A reactor training managers determined which materials should be included, and served as a primary refere ...
... for use by DOE category A reactors. The subject areas, subject matter content, and level of detail of the Reactor Operator Fundamentals Manuals were determined from several sources. DOE Category A reactor training managers determined which materials should be included, and served as a primary refere ...
Uranium
... is the reason uranium still exists on the Earth. Three additional isotopes of uranium are not naturally present but can be produced by nuclear transformations. These are uranium-232, uranium-233, and uranium-236. Like the natural uranium isotopes, these three also decay by emitting an alpha particle ...
... is the reason uranium still exists on the Earth. Three additional isotopes of uranium are not naturally present but can be produced by nuclear transformations. These are uranium-232, uranium-233, and uranium-236. Like the natural uranium isotopes, these three also decay by emitting an alpha particle ...
Slide 1
... mass of a proton plus electron (and the antineutrino). • When a neutron decays, there is less mass. • Decay will not spontaneously occur for reactions where more mass results. A proton decaying into a neutron can occur only with external energy input. ...
... mass of a proton plus electron (and the antineutrino). • When a neutron decays, there is less mass. • Decay will not spontaneously occur for reactions where more mass results. A proton decaying into a neutron can occur only with external energy input. ...
EPDG ILT Template - Nuclear Community
... incoming neutron. Therefore, there is no conservation of kinetic energy; however, there is conservation of momentum. Radiative capture is the absorption of a neutron by the target nucleus, resulting in an excited nucleus that subsequently (typically within a small fraction of a second) releases i ...
... incoming neutron. Therefore, there is no conservation of kinetic energy; however, there is conservation of momentum. Radiative capture is the absorption of a neutron by the target nucleus, resulting in an excited nucleus that subsequently (typically within a small fraction of a second) releases i ...
Pearson Physics Level 30 Unit VIII Atomic Physics: Chapter 16
... 100 but, unlike the electrostatic force, it acts only over very short distances—a few femtometres at most—and acts on both protons and neutrons. The range of the electromagnetic force is infinite, but it acts only on charged particles, such as protons and electrons. (b) The protons repel each other ...
... 100 but, unlike the electrostatic force, it acts only over very short distances—a few femtometres at most—and acts on both protons and neutrons. The range of the electromagnetic force is infinite, but it acts only on charged particles, such as protons and electrons. (b) The protons repel each other ...
THE ATOMIC NUCLEUS AND RADIOACTIVITY
... decay into a proton plus an electron (and also an antineutrino, a tiny particle we will not discuss here). About half of a bunch of lone neutrons will decay in 11 minutes. Particles that decay by spontaneously emitting charged particles and energy are said to be radioactive. Radioactivity inside ato ...
... decay into a proton plus an electron (and also an antineutrino, a tiny particle we will not discuss here). About half of a bunch of lone neutrons will decay in 11 minutes. Particles that decay by spontaneously emitting charged particles and energy are said to be radioactive. Radioactivity inside ato ...
NUCLEI of ATOMS Vladislav Konovalov Abstract
... example, one of α-planes of a nucleus of an isotope 20Ca48 will look, as shown in a figure 7. "At little change of number of nucleons some non-regular changes of radius take place. For example, at transition from 20Са40 to 20Са48 radius of distribution of charges practically does not change (that is ...
... example, one of α-planes of a nucleus of an isotope 20Ca48 will look, as shown in a figure 7. "At little change of number of nucleons some non-regular changes of radius take place. For example, at transition from 20Са40 to 20Са48 radius of distribution of charges practically does not change (that is ...
Tamene Hailu - Addis Ababa University Institutional Repository
... hydrogen. The recoil proton has high kinetic energy and penetrating power .If the recoil nuclei are considered to appear under the influence of gamma ray, then, the gamma ray energy must be so high that it is impossible to fit it to energy balance of the nuclear reaction activated by alpha particle ...
... hydrogen. The recoil proton has high kinetic energy and penetrating power .If the recoil nuclei are considered to appear under the influence of gamma ray, then, the gamma ray energy must be so high that it is impossible to fit it to energy balance of the nuclear reaction activated by alpha particle ...
Interim Exam - Review H-Chem 2015
... 69. Which change in the temperature of a 1-gram sample of water would cause the greatest increase in the average kinetic energy of its molecules? 1. 1°C to 10°C 3. 50°C to 60°C 5. 500°C to 501°C ...
... 69. Which change in the temperature of a 1-gram sample of water would cause the greatest increase in the average kinetic energy of its molecules? 1. 1°C to 10°C 3. 50°C to 60°C 5. 500°C to 501°C ...
Neutrons and Protons
... • Various quantum states for nucleons in the nucleus • Similar to the hydrogen atom: one electron in each quantum state. • Two states at each energy (spin up & spin down) ...
... • Various quantum states for nucleons in the nucleus • Similar to the hydrogen atom: one electron in each quantum state. • Two states at each energy (spin up & spin down) ...
Introduction to Subatomic Physics
... According to collision participants (photonuclear reactions, heavy ion reactions, proton induced reactions, neutron production reactions …) According to reaction energy (exothermic, endothermic reactions) According to energy of impinging particles (low energy, high energy, relativistic collision, ul ...
... According to collision participants (photonuclear reactions, heavy ion reactions, proton induced reactions, neutron production reactions …) According to reaction energy (exothermic, endothermic reactions) According to energy of impinging particles (low energy, high energy, relativistic collision, ul ...
Nuclear fission
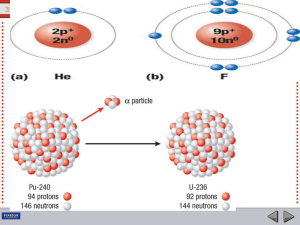
In nuclear physics and nuclear chemistry, nuclear fission is either a nuclear reaction or a radioactive decay process in which the nucleus of an atom splits into smaller parts (lighter nuclei). The fission process often produces free neutrons and photons (in the form of gamma rays), and releases a very large amount of energy even by the energetic standards of radioactive decay.Nuclear fission of heavy elements was discovered on December 17, 1938 by German Otto Hahn and his assistant Fritz Strassmann, and explained theoretically in January 1939 by Lise Meitner and her nephew Otto Robert Frisch. Frisch named the process by analogy with biological fission of living cells. It is an exothermic reaction which can release large amounts of energy both as electromagnetic radiation and as kinetic energy of the fragments (heating the bulk material where fission takes place). In order for fission to produce energy, the total binding energy of the resulting elements must be less negative (higher energy) than that of the starting element.Fission is a form of nuclear transmutation because the resulting fragments are not the same element as the original atom. The two nuclei produced are most often of comparable but slightly different sizes, typically with a mass ratio of products of about 3 to 2, for common fissile isotopes. Most fissions are binary fissions (producing two charged fragments), but occasionally (2 to 4 times per 1000 events), three positively charged fragments are produced, in a ternary fission. The smallest of these fragments in ternary processes ranges in size from a proton to an argon nucleus.Apart from fission induced by a neutron, harnessed and exploited by humans, a natural form of spontaneous radioactive decay (not requiring a neutron) is also referred to as fission, and occurs especially in very high-mass-number isotopes. Spontaneous fission was discovered in 1940 by Flyorov, Petrzhak and Kurchatov in Moscow, when they decided to confirm that, without bombardment by neutrons, the fission rate of uranium was indeed negligible, as predicted by Niels Bohr; it wasn't.The unpredictable composition of the products (which vary in a broad probabilistic and somewhat chaotic manner) distinguishes fission from purely quantum-tunnelling processes such as proton emission, alpha decay and cluster decay, which give the same products each time. Nuclear fission produces energy for nuclear power and drives the explosion of nuclear weapons. Both uses are possible because certain substances called nuclear fuels undergo fission when struck by fission neutrons, and in turn emit neutrons when they break apart. This makes possible a self-sustaining nuclear chain reaction that releases energy at a controlled rate in a nuclear reactor or at a very rapid uncontrolled rate in a nuclear weapon.The amount of free energy contained in nuclear fuel is millions of times the amount of free energy contained in a similar mass of chemical fuel such as gasoline, making nuclear fission a very dense source of energy. The products of nuclear fission, however, are on average far more radioactive than the heavy elements which are normally fissioned as fuel, and remain so for significant amounts of time, giving rise to a nuclear waste problem. Concerns over nuclear waste accumulation and over the destructive potential of nuclear weapons may counterbalance the desirable qualities of fission as an energy source, and give rise to ongoing political debate over nuclear power.