* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Essential Fish Habitat Assessment Revised Final Report – May 2014
The Marine Mammal Center wikipedia , lookup
Challenger expedition wikipedia , lookup
Fish reproduction wikipedia , lookup
Marine pollution wikipedia , lookup
Diving in the Maldives wikipedia , lookup
Ecosystem of the North Pacific Subtropical Gyre wikipedia , lookup
Demersal fish wikipedia , lookup
Deep sea fish wikipedia , lookup
MARIANA ISLANDS TRAINING AND
T E S T IN G
ESSENTIAL FISH HABITAT ASSESSMENT
FINAL REPORT
MAY 2014
Submitted By:
Commander, United States Pacific Fleet
Department of the Navy
250 Makalapa Drive
Pearl Harbor, Hawaii 96860-3131
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
LIST OF ACRONYMS AND ABBREVIATIONS
µPa
micropascal(s)
2
μPa -s
EIS
micropascal squared second
A-A
Air-to-Air
AAV
Amphibious Assault Vehicle
A-S
EMATT
EOD
EW
Air-to-Surface
AAW
Anti-Air Warfare
ac.
acre(s)
Environmental Impact Statement
Expendable Mobile ASW Training Target
Explosive Ordnance Disposal
Electronic Warfare
EW OPS
Electronic Warfare Operations
EXTORP
Exercise Torpedo
F
Fahrenheit
ACM
Air Combat Maneuver
ADEX
Air Defense Exercise
FEP
AG
airgun
FFG
AIC
Air Intercept Control
FDM
FIREX
Farallon de Medinilla
Fishery Ecosystem Plan
Frigate
Fire Support Exercise
ALMDS
Airborne Laser Mine Detection System
AMNS
Airborne Mine Neutralization System
AMW
Amphibious Warfare
FMC
Fishery Management Council
submarine tender
FMP
Fishery Management Plan
AS
ASUW
FLAREX
fm
Anti-Surface Warfare
ASW
F.R.
Anti-Submarine Warfare
ft.
barium chromate
ft.
BAMS
Broad Area Maritime Surveillance
G
BMUS
Bottomfish Management Unit Species
BaCrO4
BOMBEX
C
CG
CHCRT
Hg(CNO)2
HRC
Currently Harvested Coral Reef Taxa
cm
Hz
centimeter(s)
CMUS
Crustacean Management Unit Species
CNMI
Commonwealth of the Northern Mariana Islands
COMNAVMAR
CRE
HF
Chaff Exercise
IEER
IMPASS
Commander, Naval Forces Marianas
in.
High-Frequency
Fulminate of Mercury
Hawaii Range Complex
Hertz
Improved Extended Echo Ranging
Integrated Maritime Portable Acoustic Scoring
inch(es)
3
cubic inch(es)
Intelligence, Surveillance, Reconnaissance
Combat Rubber Raiding Craft
in.
CSAR
Combat Search and Rescue
ISR
dB
Habitat Area of Particular Concern
and Simulation
Coral Reef Ecosystems
CRRC
CVN
Gunnery Exercise
hectare(s)
HAPC
cruiser
CHAFFEX
square foot/feet
depth
ha
Code of Federal Regulations
Federal Register
gauss
h
Celsius
C.F.R.
fathom(s)
foot/feet
2
GUNEX
Bombing Exercise
Flare Exercise
ISTT
aircraft carrier
kg
decibel(s)
Improved Surface Tow Target
kilogram(s)
dBA
decibel(s), A-weighted
kHz
kilohertz
DDG
destroyer
km
kilometer(s)
Directional Command Activated Sonobuoy
lb.
DICASS
DS
DVLA
DWADS
E
pound(s)
Doppler Sonar
LCAC
Landing Craft Air Cushion
Distributed Vertical Line Array
LCM
Landing Craft, Mechanized
Deep Water Active Distributed System
LCS
Littoral Combat Ship
East
LCU
Landing Craft, Utility
EEZ
Exclusive Economic Zone
EFH
Essential Fish Habitat
LF
LHA
i
Low-Frequency
amphibious assault ship
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
LHD
amphibious assault ship
LPD
amphibious transport dock
LSD
dock landing ship
m
R
r0
RDX
meter(s)
2
square meter(s)
3
cubic meter(s)
m
m
PUTR
MAC
Multistatic Active Coherent
MCM
Mine Countermeasure Exercise
MF
mg/L
mi.
re
REXTORP
RHIB
RMMV
MISSILEX
radius
charge radius
Royal Demolition Explosive
referenced to
Recoverable Exercise Torpedo
Rigid Hull Inflatable Boat
Remote Multi-Mission Vehicle
Mid-Frequency
RMS
Remote Minehunting System
milligrams per liter
ROV
Remotely Operated Vehicle
mile(s)
MIRC
Portable Underwater Tracking Range
S
Mariana Islands Range Complex
S-A
Missile Exercise
MITT
Mariana Islands Training and Testing
MIW
Mine Warfare
mm
millimeter(s)
SCUBA
South
Surface-to-Air
Self-Contained Underwater Breathing
Apparatus
SD
SDST
SINKEX
Swimmer Detection sonar
Ship Deployable Seaborne Target
Sinking Exercise
MPA
Maritime Patrol Aircraft
MSA
Magnuson-Stevens Fishery Conservation
and Management Act
SOP
standard operating procedure
MSO
Maritime Security Operations
SPL
Sound Pressure Level
MUS
Management Unit Species
S-S
N
n/a
Navy
NEPM
NEW
nm
2
nm
SMCMEX
SSBN
fleet ballistic missile submarine
not applicable
SSGN
guided missile submarine
SSN
United States Department of the Navy
Study Area
Non-Explosive Practice Munitions
Net Explosive Weight
STW
nautical mile(s)
SUA
square nautical mile(s)
SWATH
National Marine Fisheries Service
TACP
NOAA
National Oceanic and Atmospheric
TNT
Administration
nV
OASIS
OEIS
OPAREA
oz.
Pb(N3)2
PbO
PC
PCB
TORP
Naval Special Warfare
TORPEX
nanovolt(s)
TRACKEX
Organic Airborne and Surface Influence Sweep
Overseas Environmental Impact Statement
Operating Area
ounce(s)
Potentially Harvested Coral Reef Taxa
PMUS
Pelagic Management Unit Species
Special Use Airspace
Small Waterplane Area Twin Hull
Tactical Air Control Party
trinitrotoluene
Torpedoes
Torpedo Exercise
Tracking Exercise
Unmanned Aerial Vehicle
U.S.
U.S.C.
polychlorinated biphenyl
Strike Warfare
UAV
UNDET
lead (II) oxide
MITT Study Area
temporary threshold shift
UISS
Patrol Coastal Ship
attack submarine
TTS
UAV OPS
lead azide
PHCRT
PRIA
Surface-to-Surface
North
NMFS
NSW
Mine Countermeasure Exercise – Surface
VHF
W
WPRFMC
U.S. Pacific Remote Island Areas
UAV Operations
Unmanned Influence Sweep System
Underwater Detonation
United States
United States Code
Very High Frequency
West
Western Pacific Regional Fishery
Management Council
psu
Practical Salinity Unit
yd.
yard(s)
PTS
permanent threshold shift
YP
Yard Patrol Craft
ii
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
TABLE OF CONTENTS
1 INTRODUCTION .........................................................................................................................1-1
2 DESCRIPTION OF THE ACTION AND THE ACTION AREA ................................................................2-1
2.1 SUMMARY OF THE MARIANA ISLANDS TRAINING AND TESTING ENVIRONMENTAL IMPACT STATEMENT/OVERSEAS
ENVIRONMENTAL IMPACT STATEMENT PROPOSED ACTION ANALYZED IN THE ESSENTIAL FISH HABITAT ASSESSMENT ..2-1
2.2 DESCRIPTION OF SONAR, ORDNANCE, TARGETS, AND OTHER SYSTEMS ....................................................2-7
2.2.1 SONAR AND OTHER ACTIVE ACOUSTIC SOURCES ...................................................................................... 2-7
2.2.2 ORDNANCE/MUNITIONS ..................................................................................................................... 2-8
2.2.3 MILITARY EXPENDED MATERIALS .......................................................................................................... 2-9
2.3 CLASSIFICATION OF NON-IMPULSE AND IMPULSE SOURCES ANALYZED .....................................................2-9
2.3.1 SOURCE CLASSES ANALYZED FOR TRAINING AND TESTING ACTIVITIES ........................................................ 2-10
2.3.2 SUMMARY OF NON-IMPULSE AND IMPULSE SOURCES ............................................................................ 2-12
2.4 DESCRIPTION OF THE ACTION AREA ...............................................................................................2-13
2.5 OVERVIEW OF THE STRESSORS ANALYZED FOR EFFECTS DETERMINATIONS .............................................. 2-18
3 ESSENTIAL FISH HABITAT ...........................................................................................................3-1
3.1 WESTERN PACIFIC REGIONAL FISHERY MANAGEMENT COUNCIL .............................................................3-2
3.1.1 BOTTOMFISH MANAGEMENT UNIT ....................................................................................................... 3-4
3.1.1.1 Description and Identification of Essential Fish Habitat ............................................................. 3-4
3.1.1.2 Habitat Areas of Particular Concern ........................................................................................... 3-8
3.1.1.3 Figures and Maps ........................................................................................................................ 3-8
3.1.2 CRUSTACEANS MANAGEMENT UNIT .................................................................................................... 3-13
3.1.2.1 Description and Identification of Essential Fish Habitat ........................................................... 3-13
3.1.2.2 Habitat Areas of Particular Concern ......................................................................................... 3-13
3.1.2.3 Figures and Maps ...................................................................................................................... 3-13
3.1.3 CORAL REEF ECOSYSTEMS MANAGEMENT UNIT .................................................................................... 3-18
3.1.3.1 Currently Harvested Coral Reef Taxa Complex ......................................................................... 3-18
3.1.3.2 Figures and Maps ...................................................................................................................... 3-18
3.1.3.3 Potentially Harvested Coral Reef Taxa Complex....................................................................... 3-29
3.1.4 PELAGIC MANAGEMENT UNIT ............................................................................................................ 3-31
3.1.4.1 Description and Identification of Essential Fish Habitat ........................................................... 3-31
3.1.4.2 Habitat Areas of Particular Concern ......................................................................................... 3-34
3.1.4.3 Figures and Maps ...................................................................................................................... 3-34
3.2 DESCRIPTION OF HABITATS .........................................................................................................3-36
3.2.1 WATER COLUMN.............................................................................................................................. 3-39
3.2.1.1 Currents, Circulation Patterns, and Water Masses .................................................................. 3-40
3.2.1.2 Water Column Characteristics and Processes .......................................................................... 3-41
3.2.1.3 Bathymetry ............................................................................................................................... 3-44
3.2.1.4 Water Column Essential Fish Habitat ....................................................................................... 3-47
3.2.2 SUBSTRATES .................................................................................................................................... 3-47
3.2.2.1 Soft Shores ................................................................................................................................ 3-54
3.2.2.2 Hard Shores............................................................................................................................... 3-54
iii
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
3.2.2.3 Soft Bottoms ............................................................................................................................. 3-55
3.2.2.4 Hard Bottoms ............................................................................................................................ 3-56
3.2.2.5 Artificial Structures ................................................................................................................... 3-57
3.2.3 BIOGENIC HABITATS.......................................................................................................................... 3-62
3.2.3.1 Vegetated Shores ...................................................................................................................... 3-62
3.2.3.2 Submerged Rooted Vegetation Beds ........................................................................................ 3-63
3.2.3.3 Attached Macroalgae Beds ....................................................................................................... 3-70
3.2.3.4 Coral Reefs and Communities ................................................................................................... 3-70
4 ASSESSMENT OF IMPACTS .........................................................................................................4-1
4.1 POTENTIAL IMPACTS TO ESSENTIAL FISH HABITAT ...............................................................................4-1
4.1.1 ACOUSTIC STRESSORS ......................................................................................................................... 4-3
4.1.1.1 Non-Impulsive Stressors ............................................................................................................. 4-7
4.1.1.2 Impulsive Stressors ................................................................................................................... 4-15
4.1.2 ENERGY STRESSORS .......................................................................................................................... 4-29
4.1.2.1 Electromagnetic Devices ........................................................................................................... 4-29
4.1.3 PHYSICAL DISTURBANCE AND STRIKE STRESSORS.................................................................................... 4-31
4.1.3.1 Vessels....................................................................................................................................... 4-32
4.1.3.2 In-Water Devices....................................................................................................................... 4-37
4.1.3.3 Military Expended Materials..................................................................................................... 4-38
4.1.3.4 Seafloor Devices........................................................................................................................ 4-46
4.1.4 CONTAMINANT STRESSORS ................................................................................................................ 4-47
4.1.4.1 Explosives and Explosive Byproducts........................................................................................ 4-47
4.1.4.2 Metals ....................................................................................................................................... 4-49
4.1.4.3 Chemicals .................................................................................................................................. 4-50
4.1.4.4 Other Materials ......................................................................................................................... 4-51
4.1.5 STUDY AREA COMBINED IMPACT OF STRESSORS .................................................................................... 4-52
5 MITIGATION MEASURES ............................................................................................................5-1
5.1 STANDARD OPERATING PROCEDURES ..............................................................................................5-1
5.2 MITIGATION MEASURES ...............................................................................................................5-1
6 CONCLUSIONS ...........................................................................................................................6-1
7 REFERENCES ..............................................................................................................................7-1
APPENDIX A LIST OF FEDERALLY MANAGED SPECIES ....................................................................... A-1
APPENDIX B PRIMARY HABITAT TYPES DESIGNATED AS ESSENTIAL FISH HABITAT............................ A-1
B PRIMARY HABITAT TYPES DESIGNATED AS ESSENTIAL FISH HABITAT ............................................................ B-1
B.1 ESSENTIAL FISH HABITAT DESIGNATIONS BY PRIMARY HABITAT TYPE FOR EACH SPECIES/MANAGEMENT UNIT AND
LIFE STAGE ....................................................................................................................................... B-1
iv
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
LIST OF TABLES
TABLE 2-1: TYPICAL TRAINING AND TESTING ACTIVITIES IN THE ACTION AREA ............................................................................ 2-2
TABLE 2-2: IMPULSE TRAINING AND TESTING SOURCE CLASSES ANALYZED .............................................................................. 2-10
TABLE 2-3: NON-IMPULSE TRAINING AND TESTING SOURCE CLASSES ANALYZED ...................................................................... 2-11
TABLE 2-4: ANNUAL USE OF NON-IMPULSE SOURCES DURING TRAINING AND TESTING ACTIVITIES WITHIN THE ACTION AREA .......... 2-12
TABLE 2-5: ANNUAL NUMBER OF IMPULSE SOURCE DETONATIONS DURING TRAINING AND TESTING ACTIVITIES WITHIN THE ACTION AREA
........................................................................................................................................................................ 2-13
TABLE 2-6: NEARSHORE TRAINING AND TESTING AREAS ...................................................................................................... 2-17
TABLE 2-7: DESCRIPTION OF STRESSORS ........................................................................................................................... 2-19
TABLE 2-8: TRAINING ACTIVITIES OCCURRING IN THE ACTION AREA ....................................................................................... 2-23
TABLE 2-9: PROPOSED NAVAL AIR SYSTEMS COMMAND TESTING ACTIVITIES IN THE ACTION AREA .............................................. 2-31
TABLE 2-10: PROPOSED NAVAL SEA SYSTEMS COMMAND TESTING ACTIVITIES IN THE ACTION AREA ........................................... 2-32
TABLE 2-11: PROPOSED OFFICE OF NAVAL RESEARCH TESTING ACTIVITIES IN THE ACTION AREA ................................................. 2-34
TABLE 3-1: ESSENTIAL FISH HABITAT AND HABITAT AREAS OF PARTICULAR CONCERN DESIGNATIONS FOR THE MARIANA ARCHIPELAGO
FISHERY ECOSYSTEM PLAN MANAGEMENT UNIT .......................................................................................................... 3-5
TABLE 3-2: CORAL REEF ECOSYSTEM HABITAT AREAS OF PARTICULAR CONCERN CRITERIA DESIGNATIONS IN THE MARIANA ARCHIPELAGO
........................................................................................................................................................................ 3-31
TABLE 3-3: ESSENTIAL FISH HABITAT AND HABITAT AREA OF PARTICULAR CONCERN DESIGNATED BY WESTERN PACIFIC REGIONAL FISHERY
MANAGEMENT COUNCIL ....................................................................................................................................... 3-33
TABLE 3-4: COASTAL AND MARINE ECOLOGICAL CLASSIFICATION STANDARD CROSSWALK .......................................................... 3-36
TABLE 3-5: WATER COLUMN ESSENTIAL FISH HABITAT AND HABIT AREAS OF PARTICULAR CONCERN REFERENCES WITHIN THE MARIANA
ISLANDS TRAINING AND TESTING STUDY AREA ........................................................................................................... 3-47
TABLE 3-6: SUBSTRATE ESSENTIAL FISH HABITAT AND HABIT AREAS OF PARTICULAR CONCERN REFERENCES WITHIN THE MARIANA
ISLANDS TRAINING AND TESTING STUDY AREA ........................................................................................................... 3-48
TABLE 3-7: BIOGENIC HABITATS IN FISHERY MANAGEMENT COUNCIL AREA AND THEIR ESSENTIAL FISH HABITAT SYNONYMS ........... 3-62
TABLE 4-1: LIST OF STRESSORS ANALYZED ........................................................................................................................... 4-2
TABLE 4-2: STRESSORS BY WARFARE AND TESTING AREA ....................................................................................................... 4-3
TABLE 4-3: SONAR AND OTHER ACTIVE ACOUSTIC SOURCE CLASSES FOR THE PROPOSED ACTION (ANNUAL HOURS OR NUMBER OF
ITEMS) ................................................................................................................................................................. 4-8
TABLE 4-4: REPRESENTATIVE ORDNANCE, NET EXPLOSIVE WEIGHTS, AND DETONATION DEPTHS ................................................ 4-16
TABLE 4-5: ESTIMATED EXPLOSIVE EFFECTS RANGES FOR FISH WITH SWIM BLADDERS............................................................... 4-17
TABLE 4-6: TRAINING AND TESTING ACTIVITIES THAT INCLUDE SEAFLOOR EXPLOSIONS .............................................................. 4-19
TABLE 4-7: BOTTOM DETONATIONS FOR TRAINING AND TESTING ACTIVITIES UNDER PROPOSED ACTION ...................................... 4-23
TABLE 4-8: EXPLOSIONS IN THE WATER COLUMN FROM TRAINING ACTIVITIES (EXCLUDING EXPLOSION ON OR NEAR THE BOTTOM), AND
THEIR IMPACT ON WATER COLUMN ESSENTIAL FISH HABITAT ...................................................................................... 4-24
TABLE 4-9: EXPLOSIONS IN THE WATER COLUMN FROM TESTING ACTIVITIES (EXCLUDING EXPLOSION ON OR NEAR THE BOTTOM), AND
THEIR IMPACT ON WATER COLUMN ESSENTIAL FISH HABITAT ...................................................................................... 4-26
TABLE 4-10: REPRESENTATIVE WEAPONS NOISE CHARACTERISTICS........................................................................................ 4-27
TABLE 4-11: REPRESENTATIVE VESSEL TYPES, LENGTHS, AND SPEEDS..................................................................................... 4-32
TABLE 4-12: REPRESENTATIVE TYPES, SIZES, AND SPEEDS OF IN-WATER DEVICES..................................................................... 4-37
TABLE 4-13: ANNUAL NUMBERS AND IMPACTS OF MILITARY EXPENDED MATERIALS PROPOSED FOR USE UNDER THE PROPOSED ACTION...
........................................................................................................................................................................ 4-44
TABLE 4-14: BYPRODUCTS FROM THE UNDERWATER DETONATION OF A HIGH BLAST EXPLOSIVE ................................................. 4-48
TABLE 4-15: FAILURE RATES AND LOW-ORDER DETONATION RATES OF MILITARY ORDNANCE.................................................... 4-48
TABLE 4-16: CONSTITUENTS REMAINING AFTER LOW-ORDER DETONATIONS AND FROM UNCONSUMED EXPLOSIVES ...................... 4-49
TABLE 4-17: COMBINED IMPACT ON MARINE SUBSTRATES FROM THE PROPOSED ACTION ......................................................... 4-52
TABLE 5-1: SUMMARY OF RECOMMENDED MITIGATION MEASURES ........................................................................................ 5-2
TABLE 6-1: POTENTIAL IMPACTS ON ESSENTIAL FISH HABITAT FROM EACH STRESSOR.................................................................. 6-1
v
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
LIST OF FIGURES
FIGURE 2-1: THE AT-SEA PORTION OF THE MARIANA ISLANDS TRAINING AND TESTING STUDY AREA COMPRISES THE ACTION AREA .. 2-14
FIGURE 2-2: MARIANA ISLANDS RANGE COMPLEX AIRSPACE ................................................................................................ 2-15
FIGURE 2-3: NEARSHORE TRAINING AND TESTING AREAS..................................................................................................... 2-16
FIGURE 3-1: WESTERN PACIFIC REGIONAL FISHERY MANAGEMENT COUNCIL JURISDICTION WITHIN THE MARIANA ISLANDS TRAINING AND
TESTING STUDY AREA ............................................................................................................................................. 3-2
FIGURE 3-2: WESTERN PACIFIC REGIONAL FISHERY MANAGEMENT COUNCIL GEOGRAPHIC AREA .................................................. 3-3
FIGURE 3-3: ESSENTIAL FISH HABITAT FOR ALL EGGS AND LARVAL LIFESTAGES OF BOTTOMFISH DESIGNATED ON GUAM, TINIAN, AND
FARALLON DE MEDINILLA ........................................................................................................................................ 3-9
FIGURE 3-4: ESSENTIAL FISH HABITAT FOR ALL JUVENILE AND ADULT LIFESTAGES OF BOTTOMFISHES DESIGNATED ON FARALLON DE
MEDINILLA ......................................................................................................................................................... 3-10
FIGURE 3-5: ESSENTIAL FISH HABITAT FOR ALL JUVENILE AND ADULT LIFESTAGES OF BOTTOMFISH AND HABITAT AREAS OF PARTICULAR
CONCERN DESIGNATED ON GUAM .......................................................................................................................... 3-11
FIGURE 3-6: ESSENTIAL FISH HABITAT FOR ALL JUVENILE AND ADULT LIFESTAGES OF BOTTOMFISH AND HABITAT AREAS OF PARTICULAR
CONCERN DESIGNATED ON TINIAN .......................................................................................................................... 3-12
FIGURE 3-7: ESSENTIAL FISH HABITAT FOR ALL EGGS AND LARVAL LIFESTAGES OF CRUSTACEANS DESIGNATED ON GUAM, TINIAN, AND
FARALLON DE MEDINILLA ...................................................................................................................................... 3-14
FIGURE 3-8: ESSENTIAL FISH HABITAT FOR ALL JUVENILE AND ADULT LIFESTAGES OF CRUSTACEANS DESIGNATED ON GUAM ............ 3-15
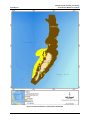
FIGURE 3-9: ESSENTIAL FISH HABITAT FOR ALL JUVENILE AND ADULT LIFESTAGES OF CRUSTACEANS DESIGNATED ON TINIAN ........... 3-16
FIGURE 3-10: ESSENTIAL FISH HABITAT FOR ALL JUVENILE AND ADULT LIFESTAGES OF CRUSTACEANS DESIGNATED ON FARALLON DE
MEDINILLA ......................................................................................................................................................... 3-17
FIGURE 3-11: ESSENTIAL FISH HABITAT FOR VARIOUS LIFESTAGES OF THE CURRENTLY HARVESTED CORAL REEF TAXA-CORAL REEF
ECOSYSTEM) ON GUAM, TINIAN, AND FARALLON DE MEDINILLA ................................................................................... 3-19
FIGURE 3-12: ESSENTIAL FISH HABITAT FOR ALL JUVENILE AND ADULT LIFESTAGES OF THE CURRENTLY HARVESTED CORAL REEF TAXACORAL REEF ECOSYSTEM ON GUAM ........................................................................................................................ 3-20
FIGURE 3-13: ESSENTIAL FISH HABITAT FOR ALL JUVENILE AND ADULT LIFESTAGES OF FLAGTAILS AND MULLETS (CURRENTLY HARVESTED
CORAL REEF TAXA-CORAL REEF ECOSYSTEM) ON GUAM ............................................................................................. 3-21
FIGURE 3-14: ESSENTIAL FISH HABITAT FOR ALL ADULT LIFESTAGES OF RUDDERFISHES (CURRENTLY HARVESTED CORAL REEF TAXA-CORAL
REEF ECOSYSTEM) ON GUAM ................................................................................................................................. 3-22
FIGURE 3-15: ESSENTIAL FISH HABITAT FOR ALL JUVENILE AND ADULT LIFESTAGES OF THE CURRENTLY HARVESTED CORAL REEF TAXACORAL REEF ECOSYSTEM ON TINIAN ........................................................................................................................ 3-23
FIGURE 3-16: ESSENTIAL FISH HABITAT FOR ALL JUVENILE AND ADULT LIFESTAGES OF FLAGTAILS AND MULLETS (CURRENTLY HARVESTED
CORAL REEF TAXA-CORAL REEF ECOSYSTEM) ON TINIAN ............................................................................................. 3-24
FIGURE 3-17: ESSENTIAL FISH HABITAT FOR ALL ADULT LIFESTAGES OF RUDDERFISHES (CURRENTLY HARVESTED CORAL REEF TAXA-CORAL
REEF ECOSYSTEM) ON TINIAN................................................................................................................................. 3-25
FIGURE 3-18: ESSENTIAL FISH HABITAT FOR ALL JUVENILE AND ADULT LIFESTAGES OF THE CURRENTLY HARVESTED CORAL REEF TAXACORAL REEF ECOSYSTEM ON FARALLON DE MEDINILLA ............................................................................................... 3-26
FIGURE 3-19: ESSENTIAL FISH HABITAT FOR ALL JUVENILE AND ADULT LIFESTAGES OF THE FLAGTAILS AND MULLETS (CURRENTLY
HARVESTED CORAL REEF TAXA-CORAL REEF ECOSYSTEM) ON FARALLON DE MEDINILLA .................................................... 3-27
FIGURE 3-20: ESSENTIAL FISH HABITAT FOR ALL ADULT LIFESTAGES OF RUDDERFISHES (CURRENTLY HARVESTED CORAL REEF TAXA-CORAL
REEF ECOSYSTEM) ON FARALLON DE MEDINILLA ........................................................................................................ 3-28
FIGURE 3-21: ESSENTIAL FISH HABITAT FOR ALL LIFESTAGES OF THE POTENTIALLY HARVESTED CORAL REEF TAXA-CORAL REEF ECOSYSTEM
AND HABIT AREAS OF PARTICULAR CONCERN DESIGNATED ON GUAM, TINIAN, AND FARALLON DE MEDINILLA ...................... 3-30
FIGURE 3-22: ESSENTIAL FISH HABITAT FOR ALL LIFESTAGES OF PELAGIC FISHES DESIGNATED ON GUAM, TINIAN, AND FARALLON DE
MEDINILLA ......................................................................................................................................................... 3-35
FIGURE 3-23: THREE-DIMENSIONAL REPRESENTATION OF A CONTINENTAL MARGIN AND ABYSSAL ZONE...................................... 3-40
FIGURE 3-24: SURFACE CIRCULATION OF THE PACIFIC OCEAN AND OUTLINE OF THE NORTH PACIFIC SUBTROPICAL GYRE ................. 3-41
FIGURE 3-25: SEA SURFACE TEMPERATURE SHOWING THE SEASONAL VARIATION IN THE MARIANA ISLANDS TRAINING AND TESTING
STUDY AREA ....................................................................................................................................................... 3-43
FIGURE 3-26: SEAFLOOR SURROUNDING THE MARIANA ISLANDS........................................................................................... 3-46
FIGURE 3-27: BOTTOM SUBSTRATE AROUND GUAM ........................................................................................................... 3-49
vi
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
FIGURE 3-28: BOTTOM SUBSTRATE IN APRA HARBOR ......................................................................................................... 3-50
FIGURE 3-29: BOTTOM SUBSTRATE AROUND SAIPAN .......................................................................................................... 3-51
FIGURE 3-30: BOTTOM SUBSTRATE AROUND TINIAN........................................................................................................... 3-52
FIGURE 3-31: BOTTOM SUBSTRATE AROUND FARALLON DE MEDINILLA .................................................................................. 3-53
FIGURE 3-32: KNOWN SHIPWRECKS AND OTHER OBSTRUCTIONS WITHIN 12 NAUTICAL MILES OF GUAM, ROTA, TINIAN, AND SAIPAN ......
........................................................................................................................................................................ 3-59
FIGURE 3-33: FISH AGGREGATING DEVICES SURROUNDING GUAM ........................................................................................ 3-60
FIGURE 3-34: FISH AGGREGATING DEVICES AROUND TINIAN AND SAIPAN ............................................................................... 3-61
FIGURE 3-35: DISTRIBUTION OF SEAGRASS AND MANGROVE COMMUNITIES IN THE MARIANA ISLANDS TRAINING AND TESTING STUDY
AREA: (A) GUAM, (B) APRA HARBOR, AND (C) TINIAN AND SAIPAN ............................................................................... 3-63
FIGURE 3-36: MARINE VEGETATION SURROUNDING GUAM ................................................................................................. 3-65
FIGURE 3-37: MARINE VEGETATION IN APRA HARBOR ........................................................................................................ 3-66
FIGURE 3-38: MARINE VEGETATION SURROUNDING TINIAN ................................................................................................. 3-67
FIGURE 3-39: MARINE VEGETATION SURROUNDING SAIPAN ................................................................................................ 3-68
FIGURE 3-40: MARINE VEGETATION SURROUNDING FARALLON DE MEDINILLA ........................................................................ 3-69
FIGURE 3-41: BENTHIC HABITATS OF THE SASA BAY............................................................................................................ 3-73
FIGURE 3-42: BENTHIC HABITATS OF SAN LUIS BEACH ........................................................................................................ 3-74
FIGURE 3-43: BENTHIC HABITATS OF KILO WHARF ............................................................................................................. 3-75
FIGURE 3-44: BENTHIC HABITATS OF GLASS BREAKWATER ................................................................................................... 3-76
FIGURE 3-45: CORAL COVERAGE SURROUNDING TINIAN...................................................................................................... 3-77
FIGURE 3-46: CORAL COMMUNITIES SURROUNDING FARALLON DE MEDINILLA ........................................................................ 3-80
FIGURE 4-1: ESTIMATE OF SPREADING LOSS FOR A 235 DECIBELS REFERENCED TO 1 MICROPASCAL SOUND SOURCE ASSUMING SIMPLE
SPHERICAL SPREADING LOSS .................................................................................................................................. 4-10
FIGURE 4-2: PREDICTION OF DISTANCE TO 10 PERCENT MORTALITY OF MARINE INVERTEBRATES EXPOSED TO AN UNDERWATER
EXPLOSION ......................................................................................................................................................... 4-18
FIGURE 4-3: MINE NEUTRALIZATION AND BEACH LANDING SITES IN RELATION TO MARINE VEGETATION ...................................... 4-21
FIGURE 4-4: MINE NEUTRALIZATION SITES AND BEACH LANDING SITES IN RELATION TO CORAL .................................................. 4-22
FIGURE 4-5: TINIAN AMPHIBIOUS LANDING BEACHES IN RELATION TO MARINE VEGETATION ..................................................... 4-34
FIGURE 4-6: TINIAN AMPHIBIOUS LANDING BEACHES IN RELATION TO CORAL .......................................................................... 4-35
FIGURE 4-7: A MK-58 SMOKE FLOAT OBSERVED IN AN AREA DOMINATED BY CORAL RUBBLE ON THE CONTINENTAL SLOPE ............ 4-40
FIGURE 4-8: AN UNIDENTIFIED, NON-MILITARY STRUCTURE OBSERVED ON THE RIDGE SYSTEM RUNNING PARALLEL TO THE CONTINENTAL
SHELF BREAK ...................................................................................................................................................... 4-40
FIGURE 4-9: (LEFT) A 76-MILLIMETER CARTRIDGE CASING ON SOFT BOTTOM. (RIGHT) A BLACKBELLY ROSEFISH (HELICOLENUS
DACTYLOPTERUS) USING THE CASING FOR SHELTER WHEN DISTURBED ........................................................................... 4-41
vii
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
This Page Intentionally Left Blank
viii
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
1 INTRODUCTION
As required by the Magnuson-Stevens Fishery Conservation and Management Act (MSA), the purpose of
this document is to present the findings of the Essential Fish Habitat (EFH) Assessment (EFHA)
conducted by the United States (U.S.) Department of the Navy (Navy). The objective of this EFHA is to
evaluate how military training and testing activities proposed to occur within the Mariana Islands
Training and Testing (MITT) Study Area (Study Area) may affect EFH designated by the Western Pacific
Regional Fishery Management Council (WPRFMC). This EFHA includes a description of the Navy’s
Proposed Action, an overview of the EFH designated within the activity area, an analysis of the direct
and cumulative effects on EFH for the managed fish and their food resources, and proposed mitigation
measures selected to minimize any potential adverse effects that could result from the Proposed Action.
Additional details regarding the MITT activities, the affected environment, and the potential
environmental effects associated with ongoing and proposed military activities are contained in the
Draft MITT Environmental Impact Statement (EIS)/Overseas EIS (OEIS) (U.S. Department of the Navy
2013). The Final Marine Resources Assessment for the Japan and Mariana Archipelagos (U.S.
Department of the Navy 2013) also contains comprehensive descriptions of the marine environment,
including climate; marine geology; physical, chemical, and biological oceanography; marine habitats; and
protected species in the Study Area. This document is available to the public and can be obtained from
the Navy’s Marine Resources Assessments website. 1
https://www.navfac.navy.mil/products_and_services/ev/products_and_services/marine_resources/marine_resource_assess
ments.html
1
1-1
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
This Page Intentionally Left Blank
1-2
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
2 DESCRIPTION OF THE ACTION AND THE ACTION AREA
2.1
SUMMARY OF THE MARIANA ISLANDS TRAINING AND TESTING ENVIRONMENTAL IMPACT
STATEMENT/OVERSEAS ENVIRONMENTAL IMPACT STATEMENT PROPOSED ACTION
ANALYZED IN THE ESSENTIAL FISH HABITAT ASSESSMENT
The Navy prepared an EIS/OEIS to assess the potential environmental impacts associated with two
categories of military readiness activities: training and testing. The EIS/OEIS also assessed sonar
maintenance and gunnery exercises (GUNEXs) conducted concurrently with ship transits and pierside
sonar activity as part of overhaul, modernization, maintenance, and repair activities. The Action covered
in this EFHA is the training and testing activities described in Alternative 1 (Preferred Alternative) in the
MITT EIS/OEIS. The Action Area is described in detail below in Section 2.4 (Description of the Action
Area).
The Navy, U.S. Air Force, U.S. Marine Corps, and U.S. Coast Guard routinely train in the Action Area in
preparation for national defense missions. Typical training and testing activities and exercises covered in
this EFHA are briefly described in Table 2-1, and in more detail within the MITT Draft EIS/OEIS
(Alternative 1) (U.S. Department of the Navy 2013). Each military training activity described meets a
requirement that can be traced ultimately to requirements set forth by the National Command
Authority. 2
The Navy and other services have been conducting military readiness activities in the Action Area for
decades. The tempo and types of training and testing activities have fluctuated because of the
introduction of new technologies, the evolving nature of international events, advances in warfighting
doctrine and procedures, and changes in force structure (e.g., organization of ships, weapons, and
military personnel). Such developments influence changes in the frequency, duration, intensity, and
location of required training and testing activities. The Navy categorizes training and testing activities
into functional warfare areas called primary mission areas. Most training and testing activities analyzed
in the MITT EIS/OEIS and EFHA fall into the following eight primary mission areas:
•
•
•
•
•
•
•
•
Anti-Air Warfare
Strike Warfare
Amphibious Warfare (AMW)
Anti-Surface Warfare
Anti-Submarine Warfare (ASW)
Electronic Warfare
Mine Warfare (MIW)
Naval Special Warfare
Not all activities can be categorized in one of these areas. The research and acquisition community (i.e.,
testing community) also categorizes some, but not all, of its testing activities under these primary
mission areas. Testing activities analyzed in the MITT EIS/OEIS and in the EFHA are categorized into the
following areas:
•
Life Cycle Activities Shipboard Protection Systems and Swimmer Defense Testing
2
“National Command Authority” is a term used by the U.S. military and government to refer to the ultimate lawful source of
military orders. The term refers collectively to the President of the United States (as Commander-in-Chief) and the U.S.
Secretary of Defense.
2-1
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
•
•
New Ship Construction
Naval Research
Additionally, some activities are described in the EIS/OEIS as Major Training Activities and Other
Activities. A summary of the training and testing activities included as part of the Action is presented in
Tables 2-8 through 2-11 at the end of this chapter. Data in the tables includes the name of the activity,
the number of times per year the activity occurs, annual number of ordnance used during the activity
(explosive and non-explosive), and the location(s) where the activity occurs.
Table 2-1: Typical Training and Testing Activities in the Action Area
Activity Name
Anti-Air Warfare (AAW)
Gunnery Exercise (Surface-to-Air)
(GUNEX [S-A]) – Large-caliber
Gunnery Exercise (Surface-to-Air)
(GUNEX [S-A]) – Medium-caliber
Missile Exercise (Surface-to-Air)
(MISSILEX [S-A])
Strike Warfare (STW)
Combat Search and Rescue (CSAR)
Amphibious Warfare (AMW)
Naval Surface Fire Support Exercise –
Land-Based Target
(FIREX [Land])
Activity Description
Surface ship crews defend against threat aircraft or missiles with guns.
Surface ship crews defend against threat aircraft or missiles with guns.
Surface ship crews defend against threat missiles and aircraft with
missiles.
CSAR units use helicopters, night vision and identification systems, and
insertion and extraction techniques under hostile conditions to locate,
rescue, and extract personnel.
Surface ship crews use large-caliber guns to fire on land-based targets in
support of forces ashore.
Amphibious Rehearsal, No Landing
Amphibious shipping, landing craft, and elements of the Marine Air Ground
Task Force rehearse amphibious landing operations without conducting an
actual landing on shore.
Amphibious Assault
Forces move ashore from ships at sea for the immediate execution of
inland objectives.
Amphibious Raid
Small unit forces move swiftly from ships at sea for a specific short-term
mission. Raids are quick operations with as few Marines as possible.
Unmanned Aerial Vehicles Ops
(UAV OPS)
Military units employ unmanned aerial vehicles to launch, operate, and
gather intelligence for specified amphibious missions.
Anti-Surface Warfare (ASUW)
Gunnery Exercise (Air-to-Surface) –
Small-caliber
Fixed-wing and helicopter aircrews, including embarked personnel, use
small-caliber guns to engage surface targets.
Gunnery Exercise (Air-to-Surface) –
Medium-caliber
Fixed-wing and helicopter aircrews, including embarked personnel, use
medium-caliber guns to engage surface targets.
Missile Exercise (Air-to-Surface) –
Rocket
(MISSILEX [A-S] – Rocket)
Missile Exercise (Air-to-Surface) –
Missile
(MISSILEX [A-S] – Missile)
Laser Targeting (at sea)
Fixed-wing and helicopter aircrews fire precision-guided and unguided
rockets against surface targets.
Fixed-wing and helicopter aircrews fire precision-guided missiles against
surface targets.
Fixed-winged, helicopter, and ship crews illuminate enemy targets with
lasers.
2-2
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-1: Typical Training and Testing Activities in the Action Area (continued)
Activity Name
Activity Description
Anti-Surface Warfare (ASUW) (continued)
Bombing Exercise (Air-to-Surface)
Fixed-wing aircrews deliver bombs against surface targets.
(BOMBEX [A-S])
Torpedo Exercise (Submarine-toSurface)
Submarine attacks a surface target using exercise or live-fire torpedoes.
Missile Exercise (Surface-to-Surface)
(MISSILEX [S-S])
Surface ship crews defend against threat missiles and other surface ships
with missiles.
Gunnery Exercise Surface-to-Surface
(Ship) – Large-caliber
Ship crews engage surface targets with ship’s large-caliber guns.
(GUNEX S-S [Ship])
Gunnery Exercise Surface-to-Surface
(Ship) – Small- and Medium-caliber
Ship crews engage surface targets with ship’s small- and medium-caliber
guns.
(GUNEX S-S [Ship])
Sinking Exercise (SINKEX)
Aircraft, ship, and submarine crews deliver ordnance on a seaborne target,
usually a deactivated ship, which is deliberately sunk using multiple
weapon systems.
Gunnery Exercise Surface-to-Surface
(Boat)
Small boat crews engage surface targets with small- and medium-caliber
weapons.
(GUNEX S-S [Boat])
Maritime Security Operations (MSO)
Helicopter and surface ship crews conduct a suite of Maritime Security
Operations (e.g., Vessel Search, Board, and Seizure; Maritime Interdiction
Operations; Force Protection; and Anti-Piracy Operation).
Air-to-Surface Missile Test
This event is similar to the training event missile exercise (air-to-surface).
Test may involve fixed-wing aircraft launching missiles at surface maritime
targets to evaluate the weapon system or as part of another system’s
integration test.
Kinetic Energy Weapon Testing
A kinetic energy weapon uses stored electromagnetic energy released in a
burst to accelerate a non-explosive projectile.
Anti-Submarine Warfare (ASW)
Countermeasure Testing
Various systems (e.g., towed arrays and defense systems) are employed
to detect, localize, and track incoming weapons.
Tracking Exercise/Torpedo Exercise –
Helicopter
Helicopter crews search, track, and detect submarines. Exercise torpedoes
may be used during this event.
(TRACKEX/TORPEX – Helo)
Tracking Exercise – Maritime Patrol
Aircraft Extended Echo Ranging
Sonobuoys
Maritime patrol aircraft crews search, detect and track submarines using
explosive source sonobuoys or multistatic active coherent system.
Tracking Exercise/Torpedo Exercise –
Maritime Patrol Aircraft
Maritime patrol aircraft crews search, detect, and track submarines.
Recoverable air launched torpedoes may be employed against submarine
targets.
(TRACKEX/TORPEX – MPA)
Tracking Exercise/Torpedo Exercise –
Surface
Surface ship crews search, track, and detect submarines. Exercise
torpedoes may be used during this event.
(TRACKEX/TORPEX – Surface)
Tracking Exercise/Torpedo Exercise –
Submarine
Submarine crews search, detect, and track submarines and surface ships.
Exercise torpedoes may be used during this event.
(TRACKEX/TORPEX – Sub)
2-3
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-1: Typical Training and Testing Activities in the Action Area (continued)
Activity Name
Activity Description
Anti-Submarine Warfare (ASW) (continued)
This event is similar to the training event ASW TRACKEX – Maritime Patrol
Aircraft. The test evaluates the sensors and systems used by maritime
Anti-Submarine Warfare Tracking Test –
patrol aircraft to detect and track submarines and to ensure that aircraft
Maritime Patrol Aircraft (Sonobuoy)
systems used to deploy the tracking systems perform to specifications and
meet operational requirements.
Anti-Submarine Warfare Torpedo Test
This event is similar to the training event torpedo exercise. The Test
evaluates ASW systems onboard rotary-wing and fixed-wing aircraft and
the system’s ability to search for, detect, classify, localize, track, and attack
a submarine or similar target. Some tests from fixed-wing aircraft will
involve releasing torpedoes and sonobuoys from high altitudes.
Broad Area Maritime Surveillance
(BAMS) – MQ-4C Triton Testing
The BAMS system will fill a complementary role to the P-8A aircraft,
providing maritime reconnaissance support to the Navy. The current BAMS
system in testing and development is called “Triton.” It will be equipped
with electro-optical/infrared sensors, can remain on station for 30 hours,
and fly at approximately 60,000 feet (18,288 meters).
Torpedo (Explosive and Non-explosive)
Testing
Air, surface, or submarine crews employ live/exercise torpedoes against
submarines or surface vessels.
At-sea Sonar Testing
At-sea testing to ensure systems are fully functional in an open ocean
environment.
Major Training Activities
Joint Expeditionary Exercise
A 10-day at-sea and ashore exercise which brings different branches of the
United States (U.S.) military together in a joint environment that includes
planning and execution efforts as well as military training activities at sea,
in the air, and ashore. More than 8,000 personnel may participate and
could include the combined assets of a Carrier Strike Group and
Expeditionary Strike Group, Marine Expeditionary Units, Army Infantry
Units, and Air Force aircraft.
Joint Multi-Strike Group Exercise
A 10-day at-sea and ashore exercise in which up to three Carrier Strike
Groups integrated with U.S. Air Force and U.S. Marine Corps forces would
conduct at-sea training and STW exercises simultaneously.
Marine Air Ground Task Force Exercise
(Amphibious) – Battalion
A 10-day at-sea and shore exercise which conducts over-the-horizon, shipto-objective maneuver for the elements of the Expeditionary Strike Group
and the Amphibious Marine Air Ground Task Force. The exercise utilizes
all elements of the Marine Air Ground Task Force (Amphibious),
conducting training activities ashore with logistic support of the
Expeditionary Strike Group and conducting amphibious landings.
2-4
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-1: Typical Training and Testing Activities in the Action Area (continued)
Activity Name
Activity Description
Major Training Activities (continued)
Special Purpose Marine Air Ground Task
Force Exercise
A 10-day at-sea and ashore exercise similar to Marine Air Ground Task
Force (Amphibious) – Battalion, but task organized to conduct a specific
mission (e.g., Humanitarian Assistance, Disaster Relief, Non-combatant
Evacuation Operations).
Electronic Warfare (EW)
Electronic Warfare Operations
(EW OPS)
Aircraft, surface ship, and submarine crews attempt to control portions of
the electromagnetic spectrum used by enemy systems to degrade or deny
the enemy’s ability to take defensive actions.
Counter Targeting Chaff Exercise
(CHAFFEX) – Ship
Surface ships defend against an attack by deploying chaff, a radarreflective material, which disrupt threat targeting and missile guidance
radars.
Counter Targeting Chaff Exercise
(CHAFFEX) – Aircraft
Fixed-winged aircraft and helicopter crews defend against an attack by
deploying chaff, a radar-reflective material, which disrupt threat targeting
and missile guidance radars.
Flare Test
Flare tests evaluate newly developed or enhanced flares, flare dispensing
equipment, or modified aircraft systems against flare deployment. Tests
may also train pilots and aircrew in the use of newly developed or modified
flare deployment systems. Flare tests are often conducted with other test
events, and are not typically conducted as standalone tests. Chaff and
flares are expended for this test event.
Mine Warfare (MIW)
Civilian Port Defense
Naval MIW activities conducted at various ports and harbors, in support of
maritime homeland defense/security.
Mine Laying
Fixed-winged aircraft and vessel crews drop/launch non-explosive mine
shapes.
Mine Neutralization – Explosive
Ordnance Disposal (EOD)
Personnel disable threat mines. Explosive charges may be used.
Limpet Mine Neutralization
System/Shock Wave Generator
Navy divers place a small charge on a simulated underwater mine.
Submarine Mine Exercise
Submarine crews practice detecting mines in a designated area.
Airborne Mine Countermeasure (MCM) –
Mine Detection
Helicopter aircrews detect mines using towed and laser mine detection
systems (e.g., AN/AQS-20, Airborne Laser Mine Detection System).
Mine Countermeasure Exercise – Towed
Sonar
Surface ship crews detect and avoid mines while navigating restricted
areas or channels using towed active sonar.
Mine Countermeasure Exercise –
Surface (SMCMEX)
Mine countermeasure ship crews detect, locate, identify, and avoid mines
while navigating restricted areas or channels using active sonar.
Mine Neutralization – Remotely
Operated Vehicle Sonar
Helicopter aircrews disable mines using remotely operated underwater
vehicles.
Mine Countermeasure (MCM) – Towed
Mine Neutralization
Ship crews and helicopter aircrews tow systems (e.g., Organic and Surface
Influence Sweep, MK 104/105) through the water that are designed to
disable or trigger mines.
Naval Special Warfare (NSW)
Personnel Insertion/Extraction
Military personnel train for covert insertion and extraction into target areas
using helicopters, fixed-wing aircraft (insertion only), small boats, and
submersibles.
2-5
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-1: Typical Training and Testing Activities in the Action Area (continued)
Activity Name
Activity Description
Naval Special Warfare (NSW) (continued)
Direct Action (Tactical Air Control Party
[TACP]/Joint Tactical Air Control)
Military personnel train for controlling of combat support aircraft and
providing target designation, airspace de-confliction, and terminal control
for Close Air Support. Teams also train in use of small arms and mortars.
Underwater Demolition
Qualification/Certification
Navy divers conduct training and certification in placing underwater
demolition charges.
Intelligence, Surveillance,
Reconnaissance (ISR)
Special Warfare units train to collect and report battlefield intelligence.
Underwater Survey
Navy divers train in survey of underwater conditions and features in
preparation for insertion, extraction, or intelligence, surveillance, and
reconnaissance activities.
Other Training Activities
Surface Ship Sonar Maintenance
In-port and at-sea maintenance of sonar systems.
Submarine Sonar Maintenance
In-port and at-sea maintenance of sonar systems.
Small Boat Attack
Small boats or personal watercraft conduct attack activities on units afloat.
Submarine Navigation
Submarine crews locate underwater objects and ships while transiting out
of port.
Search and Rescue at Sea
U.S. Coast Guard and military personnel train with ships, fixed-wing and
rotary aircraft to locate and rescue missing personnel and vessels at sea.
Precision Anchoring
Releasing of anchors in designated locations.
New Ship Construction
Anti-Submarine Warfare (ASW) Mission
Package Testing
Ships and their supporting platforms (e.g., helicopters, unmanned aerial
vehicles) detect, localize, and prosecute submarines.
Mine Countermeasures (MCM) Mission
Package Testing
Ships conduct MCM operations.
Anti-Surface Warfare (ASUW) Mission
Package Testing
Ships and their supporting platforms (e.g., helicopters, unmanned aerial
vehicles) detect, localize, and prosecute surface vessels.
2-6
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-1: Typical Training and Testing Activities in the Action Area (continued)
Activity Name
Activity Description
Life Cycle Activities
Ship Signature Testing
Tests ship and submarine radars, electromagnetic, or acoustic signatures.
Shipboard Protection Systems and Swimmer Defense Testing
Pierside Integrated Swimmer Defense
Swimmer defense testing ensures that systems can effectively detect,
characterize, verify, and engage swimmer/diver threats in harbor
environments.
Naval Research
North Pacific Acoustic Lab Philippine
Sea 2018–19 Experiment (Deep Water)
2.2
The experiment area encompasses international waters. The initial
experiment was completed in May 2011; an acoustic tomography array, a
distributed vertical line array (DVLA), and moorings were deployed in the
deep-water environment of the northwestern Philippine Sea. The acoustic
tomography array and DVLA have remained in situ at the experiment site
since that time, collecting oceanographic and acoustic data used to study
deep-water propagation and to characterize the temperature and velocity
structure in this oceanographically complex and highly dynamic region. In
addition, data will be collected during two periods of intensive experimental
at-sea operations in May and July of 2018. During fall 2018, data will be
collected passively by remotely sensing seagliders. Research vessels,
acoustic test sources, side scan sonar, ocean gliders, the existing moored
acoustic tomographic array and distributed vertical line array, and other
oceanographic data collection equipment will be used to collect information
on the ocean environment. The final phases of the experiment will be
completed during March–May 2019. The resulting analyses will aid in
developing a more complete understanding of deep water sound
propagation and the temperature-velocity profile of the water column in this
part of the world.
DESCRIPTION OF SONAR, ORDNANCE, TARGETS, AND OTHER SYSTEMS
The Navy uses a variety of sensors, platforms, weapons, and other devices, including ones used to
ensure the safety of Sailors and Marines, to meet its mission. Training and testing with these systems
may introduce acoustic (sound) energy into the environment. This section presents and organizes sonar
systems, ordnance, munitions, targets, and other systems in a manner intended to facilitate
understanding of the activities in which these systems are used.
2.2.1
SONAR AND OTHER ACTIVE ACOUSTIC SOURCES
Modern sonar technology includes a variety of sonar sensor and processing systems. In concept, the
simplest active sonar emits sound waves, or “pings,” sent out in multiple directions. The sound waves
then reflect off of the target object in multiple directions. The sonar source calculates the time it takes
for the reflected sound waves to return; this calculation determines the distance to the target object.
More sophisticated active sonar systems emit a ping and then rapidly scan or listen to the sound waves
in a specific area. This provides both distance to the target and directional information. Even more
advanced sonar systems use multiple receivers to listen to echoes from several directions
simultaneously and provide efficient detection of both direction and distance. It should be noted that
active sonar is rarely used continuously throughout the listed activities. In general, when sonar is in use,
the sonar “pings” occur at intervals, referred to as a duty cycle, and the signals themselves are very
short in duration. For example, sonar that emits a 1-second ping every 10 seconds has a 10 percent duty
cycle. The Navy utilizes sonar systems and other acoustic sensors in support of a variety of mission
2-7
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
requirements. Primary uses include the detection of and defense against submarines (ASW) and mines
(MIW), safe navigation and effective communications, use of unmanned undersea vehicles, and
oceanographic surveys.
2.2.2
ORDNANCE/MUNITIONS
Most ordnance and munitions used during training and testing activities fall into three basic categories:
projectiles, missiles, and bombs. Explosive ordnance can be further defined by net explosive weight
(NEW), which is the actual weight in pounds of the explosive substance without the packaging, casings,
bullets, etc. Net explosive weight is a measure of the strength of bombs and other explosives. For
example, a 2,000-pound (lb.) (907.2-kilogram [kg]) bomb may have anywhere from 600 to 1,000 lb.
(272.2 to 453.8 kg) of NEW.
Projectiles are fired during GUNEXs from a variety of weapons, including pistols and rifles to largecaliber turret-mounted guns on the decks of military ships. Projectiles can be either explosive munitions
(e.g., certain cannon shells) or non-explosive practice munitions (NEPM) (e.g., rifle/pistol bullets).
Explosive rounds can be fused to either explode on impact or in the air (i.e., just prior to impact).
Projectiles are broken down into three basic categories: small caliber (up to approximately 0.5 inch [in.]),
medium caliber (greater than 0.5 in., up to approximately 2.24 in. in diameter), and larger caliber (up to
5 in.).
Missiles are rocket- or jet-propelled munitions used to attack ships, aircraft, and land-based targets, as
well as defend ships against other missiles. Guidance systems and advanced fusing technology ensure
that missiles reliably impact on or detonate near their intended target. Missiles are categorized
according to their intended target and can be further classified according to NEW. Rockets are included
within the category of missiles.
Bombs are unpowered munitions dropped from aircraft on land and water targets. Bombs are in two
categories: general-purpose bombs and subscale practice bombs. Similar to missiles, bombs are further
classified according to the NEW of the bomb.
There are other munitions and ordnance used in naval at-sea training and testing activities that do not
fit into one of the above categories and are discussed below:
•
•
•
•
Demolition Charges: Divers place explosive charges in the marine environment during some
training and testing activities. These activities may include the use of timed charges, in which
the charge is placed, a timer is started, and the charge detonates at the set time. Munitions,
which are typically composed of C-4 explosive with the necessary detonators and cords, are
used to support mine neutralization, demolition, and other warfare activities. All demolition
charges are further classified according to the NEW of the charge.
Anti-Swimmer Grenades: Maritime security forces use hand grenades to defend against enemy
Self-Contained Underwater Breathing Apparatus (SCUBA) divers.
Torpedoes: Explosive torpedoes are required in some training and testing activities. Torpedoes
are described as either lightweight or heavyweight and are further categorized according to the
NEW.
Extended Echo Ranging Sonobuoys: Extended Echo Ranging sonobuoys include Improved
Extended Echo Ranging sonobuoys and mini sound-source seeker sonobuoys that use explosive
charges as the active sound source instead of electrically produced sounds.
2-8
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
2.2.3
MILITARY EXPENDED MATERIALS
Navy training and testing activities may introduce or expend various items, such as munitions and
targets, into the marine environment as a direct result of using these items for their intended purpose.
In addition to these items, some accessory materials—related to the carriage or release of these items—
may also be released. These materials, referred to as military expended materials, are not recovered,
and are analyzed as potential stressors. For detailed information on military expended materials used in
the Action Area, refer to Chapter 2 (Description of Proposed Action and Alternatives) of the MITT
EIS/OEIS.
Military expended materials analyzed in this EFHA include, but are not limited to, the following:
•
•
•
•
•
•
•
•
2.3
Sonobuoys: Some sonobuoys consist of decelerators/parachutes, and the sonobuoys
themselves.
Torpedo Launch Accessories: Torpedoes are usually recovered; however, materials such as
decelerators/parachutes used with air-dropped torpedoes, guidance wires used with
submarine-launched torpedoes, and ballast weights are expended. Explosive-filled torpedoes
expend torpedo fragments.
Decelerators/Parachutes: Aircraft-launched sonobuoys, lightweight torpedoes (such as the MK
46 and MK 54), illumination flares, and targets use nylon decelerators/parachutes ranging in size
from 18 to 48 in. (46 to 122 centimeters [cm]) in diameter.
Projectiles and Bombs: Projectiles, bombs, or fragments from explosive projectiles and bombs
are expended during training and testing exercises. These items are primarily constructed of
lead (most small-caliber projectiles) or steel (medium- and large-caliber projectiles and all
bombs).
Missiles and Rockets: Non-explosive missiles and missile fragments from explosive missiles are
expended during training and testing activities. Propellant, and any explosive material involved,
is consumed during firing and detonation. Rockets are similar to missiles, and both
non-explosive and fragments may be expended.
Countermeasures: Countermeasures (acoustic, chaff, flares) are expended as a result of training
exercises, with the exception of towed acoustic countermeasures.
Targets: Some targets are designed to be expended; other targets, such as aerial drones and
remote-controlled boats, are recovered for re-use. Targets struck with ordnance will result in
target fragments.
Ballast/Anchors: Bottom mine shapes and other sea floor devices (e.g., portable underwater
tracking range transponders) use ballast to sink to a predetermined depth or to anchor to the
bottom. The device then releases the ballast or anchor (generally lead/sand/concrete). While
the ballast/anchor is not recovered, the sea floor device floats to the surface for recovery.
CLASSIFICATION OF NON-IMPULSE AND IMPULSE SOURCES ANALYZED
In this application, underwater sound is described as one of two types: impulse and non-impulse.
Explosions and other percussive events are sources of impulse sounds. Sonar and other active acoustic
systems are categorized as non-impulse sound sources. A description of each type of source class is
provided in Tables 2-4 and 2-5. Non-impulse sources are grouped on the frequency, source level when
warranted, and the application in which the source would be used. Impulse sources are grouped based
on the NEW of the munitions or explosive devices.
2-9
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
2.3.1
SOURCE CLASSES ANALYZED FOR TRAINING AND TESTING ACTIVITIES
Tables 2-2 and 2-3 show the impulse sources (e.g., explosives) and non-impulse sources (e.g., sonar)
associated with military training and testing activities in the Action Area.
Table 2-2: Impulse Training and Testing Source Classes Analyzed
Source Class
Representative Munitions
Net Explosive Weight (lb.)
E1
Medium-caliber projectiles
0.1–0.25
E2
Medium-caliber projectiles
0.26–0.5
E3
Large-caliber projectiles
> 0.5–2.5
E4
Improved Extended Echo Ranging
Sonobuoy
> 2.5–5.0
E5
5-inch projectiles
> 5–10
E6
15 lb. shaped charge
> 10–20
E8
250 lb. bomb
> 60–100
E9
500 lb. bomb
> 100–250
E10
1,000 lb. bomb
> 250–500
E11
650 lb. mine
> 500–650
E12
2,000 lb. bomb
> 650–1,000
Note: lb. = pound(s)
2-10
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-3: Non-Impulse Training and Testing Source Classes Analyzed
Source Class Category
Low-Frequency (LF): Sources that
produce low-frequency (less than 1
kHz) signals
Mid-Frequency (MF): Tactical and
non-tactical sources that produce
mid-frequency (1–10 kHz) signals
High-Frequency (HF) and Very
High-Frequency (VHF):
Tactical and non-tactical sources
that produce high-frequency (greater
than 10 kHz but less than 200 kHz)
signals
Anti-Submarine Warfare (ASW):
Tactical sources such as active
sonobuoys and acoustic
countermeasures systems used
during the conduct of anti-submarine
warfare testing activities
Torpedoes (TORP): Source classes
associated with the active acoustic
signals produced by torpedoes
Source Class
Description
LF4
Low-frequency sources equal to 180 dB and up to 200 dB
LF5
Low-frequency sources less than 180 dB
LF6
Low-frequency sonar currently in development (e.g., ASW
sonar associated with the LCS)
MF1
Hull-mounted surface ship sonar (e.g., AN/SQS-53C and
AN/SQS-60)
MF2
Hull-mounted surface ship sonar (e.g., AN/SQS-56)
MF3
Hull-mounted submarine sonar (e.g., AN/BQQ-10)
MF4
Helicopter-deployed dipping sonar (e.g., AN/AQS-22 and
AN/AQS-13)
MF5
Active acoustic sonobuoys (e.g., DICASS)
MF6
Active underwater sound signal devices (e.g., MK-84)
MF8
Active sources (greater than 200 dB) not otherwise binned
MF9
Active sources (equal to 180 dB and up to 200 dB)
MF10
Active sources (greater than 160 dB, but less than 180 dB)
not otherwise binned
MF11
Hull-mounted surface ship sonars with an active duty cycle
greater than 80%
MF12
High duty cycle – variable depth sonar
HF1
Hull-mounted submarine sonar (e.g., AN/BQQ-10)
HF4
Mine detection, classification, and neutralization sonar
(e.g., AN/SQS-20)
HF5
Active sources (greater than 200 dB)
HF6
Active sources (equal to 180 dB and up to 200 dB)
ASW1
Mid-frequency DWADS
ASW2
Mid-frequency MAC sonobuoy (e.g., AN/SSQ-125)
ASW3
Mid-frequency towed active acoustic countermeasure
systems (e.g., AN/SLQ-25)
TORP1
Lightweight torpedo (e.g., MK-46, MK-54, or Anti-Torpedo
Torpedo)
TORP2
Heavyweight torpedo (e.g., MK-48)
Acoustic Modems (M): Systems
used to transmit data acoustically
through water
M3
Mid-frequency acoustic modems (greater than 190 dB)
Swimmer Detection Sonar (SD):
Systems used to detect divers and
submerged swimmers
SD1
High-frequency sources with short pulse lengths, used for
the detection of swimmers and other objects for the
purpose of port security.
Notes: dB = decibels, DICASS = Directional Command Activated Sonobuoy System, DWADS = Deep Water Active Distributed
System, kHz = kilohertz, LCS = Littoral Combat Ship, MAC = Multi-static Active Coherent
2-11
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
2.3.2
SUMMARY OF NON-IMPULSE AND IMPULSE SOURCES
Table 2-4 provides a quantitative annual summary of training and testing activities by non-impulse
source class analyzed in this EFHA.
Table 2-4: Annual Use of Non-Impulse Sources During Training and Testing Activities within the Action Area
Source Class Category
Source Class
Low-Frequency (LF) Sources that produce signals
less than 1 kilohertz (kHz)
Mid-Frequency (MF) Tactical and non-tactical
sources that produce signals from 1 to 10 kHz
High-Frequency (HF) and Very High-Frequency
(VHF) Tactical and non-tactical sources that produce
signals greater than 10 kHz but less than 200 kHz
Anti-Submarine Warfare (ASW) Tactical sources
used during anti-submarine warfare training and
testing activities
Annual Use
Metric
LF4
123
# of hours
LF5
11
# of hours
LF6
40
# of hours
MF1
1,872
# of hours
MF2
625
# of hours
MF3
192
# of hours
MF4
214
# of hours
MF5
2,588
# of items
MF6
33
# of items
MF8
123
# of hours
MF9
47
# of hours
MF10
231
# of hours
MF11
324
# of hours
MF12
656
# of hours
HF1
113
# of hours
HF4
1,060
# of hours
HF5
336
# of hours
HF6
1,173
# of hours
ASW1
144
# of hours
ASW2
660
# of items
ASW3
3,935
# of hours
ASW4
32
# of items
TORP1
115
# of items
TORP2
62
# of items
Acoustic Modems (M) Transmit data acoustically
through the water
M3
112
# of hours
Swimmer Detection Sonar (SD) Used to detect
divers and submerged swimmers
SD1
2,341
# of hours
Torpedoes (TORP) Source classes associated with
active acoustic signals produced by torpedoes
Table 2-5 provides a quantitative annual summary of training and testing impulse source classes
analyzed in this EFHA.
2-12
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-5: Annual Number of Impulse Source Detonations During Training and Testing Activities within the
Action Area
Explosive
Class
Net Explosive Weight
Annual In-Water
Detonations
E1
(0.1–0.25 lb.)
10,140
E2
(0.26–0.5 lb.)
106
E3
(> 0.5–2.5 lb.)
933
E4
(> 2.5–5 lb.)
420
E5
(> 5–10 lb.)
684
E6
(> 10–20 lb.)
76
E8
(> 60–100 lb.)
16
E9
(> 100–250 lb.)
4
E10
(> 250–500 lb.)
12
E11
(> 500–650 lb.)
6
E12
(> 650–2,000 lb.)
184
Note: lb. = pound(s)
2.4
DESCRIPTION OF THE ACTION AREA
The Action Area for the EFHA is the MITT Study Area excluding the land-based training areas. The Action
Area is composed of established at-sea ranges that encompass waters surrounding Guam and the
Commonwealth of the Northern Mariana Islands (CNMI), operating areas (OPAREAs), and special use
airspace in the region of the Mariana Islands that includes the existing Mariana Islands Range Complex
(MIRC) (497,469 square nautical miles [nm2]), additional areas on the high seas (487,132 nm2), and a
transit corridor between the MIRC and the Hawaii Range Complex (HRC). 3 The transit corridor is outside
the geographic boundaries of the MIRC and is a direct route across the high seas for Navy assets in
transit between the MIRC and the HRC (Figure 2-1).
The at-sea components of the MIRC include nearshore and offshore training and testing areas, ocean
surface and subsurface areas, and special use airspace (Figure 2-2). These areas extend from the waters
south of Guam to north of Pagan (CNMI), and from the Pacific Ocean east of the Mariana Islands to the
Philippine Sea to the west.
The Action Area also includes pierside locations in the Apra Harbor Naval Complex where surface ship
and submarine sonar maintenance testing occurs. For purposes of this EFHA, pierside locations include
channels and routes to and from the Navy port in the Apra Harbor Naval Complex, and associated
wharves and facilities within the Navy port and shipyard. The Action Area also includes nearshore
training and testing areas as depicted in Figures 2-3 and 2-4 and described in Table 2-6.
3
Vessel transit corridors are the routes typically used by Navy assets to traverse from one area to another. The route depicted
in Figure 2-1 is a direct route between the MIRC and the HRC, making it a quick and fuel-efficient transit. The depicted transit
corridor is notional and may not represent actual routes used. Actual routes navigated are based on a number of factors
including, but not limited to, weather and training requirements; however, the corridor represents the environment potentially
impacted by the Proposed Action.
2-13
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 2-1: The At-Sea Portion of the Mariana Islands Training and Testing Study Area Comprises the Action Area
2-14
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 2-2: Mariana Islands Range Complex Airspace
2-15
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 2-3: Nearshore Training and Testing Areas
2-16
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-6: Nearshore Training and Testing Areas
Nearshore Training
and Testing Areas
Description
Pacific Ocean off Orote
Point, Apra Harbor,
Island of Guam, Mariana
Islands; Small Arms
Firing Range
Used for Small Arms Training. Down range
Surface Danger Zone extends out over the
nearshore waters of Guam off Orote Point.
Finegayan Small Arms
Range
Used for small arms training. Down range Surface
Danger Zone extends out over the nearshore
waters of Guam off Haputo Point and overlays
part of the “Small Arms Safety Drop Zone” shown
on NOAA Chart 81048, Guam.
Pati Point Combat Arms
Training Maintenance
Small Arms Range
Used for small arms training. Down range Surface
Danger Zone extends out over the nearshore
waters of Guam off Pati Point.
Small Arms Firing Area
An area used by surface vessel crews to conduct
small arms training. This firing area is over water
west of Guam, beyond 3 nm of Guam and within
territorial waters, and within a Navy “Firing
Danger Area” charted on NOAA Chart 81048,
Guam.
Agat Bay Mine
Neutralization Site
Used by divers training to conduct underwater
detonations (UNDETs). The Exclusion Zone has a
minimum 640-meter (m) radius and is located
beyond 3 nm of Guam and within territorial
waters.
Piti Point Mine
Neutralization Site
Used by divers training to conduct UNDETs. The
Exclusion Zone has a minimum 640 m radius and
is located within 3 nm of Guam.
Apra Harbor UNDET
Site
Used by divers training to conduct UNDETs. The
Exclusion Zone has a minimum 640 m radius over
water, and is located within Apra Harbor. The
Glass Breakwater forms the northern edge of the
Exclusion Zone.
Pati Point Explosive
Ordnance Disposal
Range
Land site used by the Air Force to dispose of
ordnance. The Exclusion Zone extends partially
out over the nearshore waters of Guam off Pati
Point.
Notes: nm = nautical miles, NOAA = National Oceanic and Atmospheric Administration
2-17
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
2.5
OVERVIEW OF THE STRESSORS ANALYZED FOR EFFECTS DETERMINATIONS
For the purposes of this EFHA, the training and testing activities that encompass the Action were
deconstructed to derive potential stressors that may affect essential fish habitat. The stressors vary in
intensity, frequency, duration, and location within the Action Area. The stressors potentially affecting
essential fish habitat in this analysis are grouped into the following four categories:
•
•
•
•
Acoustic
Energy
Physical disturbance and strike
Contaminant
Table 2-7 describes the stressors in greater detail, including factors influencing how each stressor may
affect essential fish habitat.
2-18
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-7: Description of Stressors
Stressor
Acoustic
(sonar and other active
acoustic sources,
underwater explosives,
weapons firing, launch and
impact noise, aircraft
noise, and vessel noise)
Description of Stressor
Effects on species from acoustic sources are dependent on a number of factors,
including the type of sound received (non-impulse or impulse), the proximity of the
animal to the sound source, and the duration, frequency, and intensity of the sound.
Underwater sound propagation is highly dependent upon environmental
characteristics such as bathymetry, bottom type, water depth, temperature, and
salinity. The sound received at a particular location will be different than near the
source due to the interaction of many factors, including propagation loss; how the
sound is reflected, refracted, or scattered; the potential for reverberation; and
interference due to multi-path propagation.
Sonar and other active acoustic sources emit sound waves into the water to detect
objects, safely navigate, and communicate. Most systems operate within specific
frequencies (although some harmonic frequencies may be emitted at lower sound
pressure levels). Most sonar use is associated with anti-submarine warfare (ASW)
activities. Sonar use associated with mine warfare (MIW) would also contribute a
notable portion of overall acoustic sound.
Explosives used during training and testing activities include explosive ordnance,
including bombs, missiles, and naval gun shells; torpedoes; demolition charges; and
explosive sonobuoys. Depending on the activity, detonations would occur in the air,
near the water’s surface, or underwater (some torpedoes and sonobuoys). Demolition
charges could occur near the surface, in the water column, or on the seafloor. Most
detonations would occur in waters greater than 200 ft. (61 m) in depth, and greater
than 3 nm from shore, although MIW, demolition, and some testing detonations could
occur in shallow water closer to shore. Detonations associated with ASW would
typically occur in waters greater than 600 ft. (182.9 m) depth.
Noise associated with weapons firing and the impact of non-explosive practice
munitions (NEPM) could happen at any location within the Action Area but generally
would occur at locations greater than 12 nm from shore for safety reasons. These
training and testing events would occur in areas designated for anti-surface warfare
and similar activities. The firing of a weapon may have several components of
associated noise. Firing of guns could include sound generated by firing the gun
(muzzle blast), vibration from the blast propagating through a ship’s hull, and sonic
booms generated by the projectile flying through the air. Missiles and targets would
also produce noise during launch. In addition, the impact of NEPM at the water
surface can introduce noise into the water.
Fixed- and rotary-wing aircraft are used for a variety of training and testing activities
throughout the Action Area, contributing both airborne and underwater sound to the
ocean environment. Aircraft used in training and testing generally have reciprocating,
turboprop, or jet engines. Motors, propellers, and rotors produce the most noise, with
some noise contributed by aerodynamic turbulence. Aircraft sounds have more
energy at lower frequencies. Takeoffs and landings occur at established airfields as
well as on vessels at sea throughout the Action Area. Most aircraft noise would be
produced around air fields in the range complex. Military activities involving aircraft
generally are dispersed over large expanses of open ocean but can be highly
concentrated in time and location.
Vessels (including ships, small craft, and submarines) would produce low-frequency,
broadband underwater sound. Overall, naval traffic is often a minor component of
total vessel traffic (Mintz and Filadelfo 2011; Mintz and Parker 2006). Commercial
vessel traffic, which included cargo vessels, bulk carriers, passenger vessels, and oil
tankers (all over 65 ft. [20 m] in length), was heaviest near and between the major
shipping ports.
2-19
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-7: Description of Stressors (continued)
Stressor
Energy
(electromagnetic devices)
Physical disturbance and
strike
(vessels, in water devices,
military expended
materials)
Description of Stressor
Electromagnetic devices are used in towed or unmanned MIW systems that mimic the
electromagnetic signature of a vessel passing through the water. None of the devices
include any type of electromagnetic “pulse.” The devices work by emitting an
electromagnetic field and mechanically generated underwater sound to simulate the
presence of a ship. The sound and electromagnetic signature cause nearby mines to
detonate.
The static magnetic field generated by the electromagnetic devices is of relatively
minute strength. Typically, the maximum magnetic field generated would be
approximately 23 gauss (G). By comparison, magnetic field generated by a
refrigerator magnet is between 150 and 200 G. The strength of an electromagnetic
field decreases quickly with distance from the device. The magnetic field generated at
a distance of 4 m from the source is comparable to the earth’s magnetic field, which is
approximately 0.5 G.
Physical disturbances may occur in association with vessel movements, the use of inwater devices, and materials expended from vessels and aircraft.
Vessels used as part of the Action include ships (e.g., aircraft carriers, surface
combatants), support craft, small boats, and submarines, ranging in size from 5 to
over 300 m. Large Navy ships generally operate at speeds in the range of 10–15
knots, and submarines generally operate at speeds in the range of 8–13 knots. Small
craft (for purposes of this discussion, less than 40 ft. [12 m] in length), which are all
support craft, have variable speeds. Locations of vessel use in the Action Area varies
with the type of activity taking place, but greater activity would be expected near ports
than in other areas of the Action Area.
In-water devices as discussed in this analysis are unmanned vehicles, such as
remotely operated vehicles, unmanned surface vehicles and unmanned undersea
vehicles, and towed devices. These devices are self-propelled and unmanned or
towed through the water from a variety of platforms, including helicopters and surface
ships. In-water devices are generally smaller than most participating vessels, ranging
from several inches to about 15 m. These devices can operate anywhere from the
water surface to the benthic zone.
Military expended materials include: (1) all sizes of NEPM; (2) fragments from
explosive munitions; and (3) expended materials other than munitions, such as
sonobuoys, ship hulks, and expendable targets.
Activities using NEPM (e.g., small-, medium-, and large-caliber gun ammunitions,
missiles, rockets, bombs, torpedoes, and neutralizers), explosive munitions
(generating munitions fragments), and materials other than munitions (e.g., flares,
chaff, sonobuoys, decelerators/parachutes, aircraft stores and ballast, and targets)
have the potential to contribute to the physical disturbance and strike stressor.
2-20
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-7: Description of Stressors (continued)
Stressor
Contaminant
(explosives, explosion
byproducts, metals, and
chemicals)
Description of Stressor
Contaminant stressors associated with some training and testing activities could pose
indirect impacts to EFH through habitat degradation or alteration or an effect on prey
availability. Contaminant stressors include (1) explosives, (2) explosion byproducts
and unexploded ordnance, (3) metals, and (4) chemicals
Indirect impacts of explosives and unexploded ordnance to marine species via
degradation of sediment or water quality is possible in the immediate vicinity of the
ordnance. Explosion byproducts are not toxic to marine organisms at realistic
exposure levels (Rosen and Lotufo 2010). Relatively low solubility of most explosives
and their degradation products means that concentrations of these contaminants in
the marine environment are relatively low and readily diluted.
Metals are introduced into seawater and sediments as a result of training and testing
activities involving ship hulks, targets, ordnance, munitions, and other military
expended materials.
Several training and testing activities introduce potentially harmful chemicals into the
marine environment; principally, flares and propellants for rockets, missiles, and
torpedoes. Properly functioning flares missiles, rockets, and torpedoes combust most
of their propellants, leaving benign or readily diluted soluble combustion byproducts
(e.g., hydrogen cyanide). Operational failures allow propellants and their degradation
products to be released into the marine environment.
Notes: cm = centimeters, ft. = feet, m = meters, nm = nautical miles
2-21
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
This Page Intentionally Left Blank
2-22
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-8: Training Activities Occurring in the Action Area
Proposed Action
Range Activity
No. of activities
(per year)
Ordnance
(Number per year)
Location
Air Combat Maneuver (ACM)
4,800
None
Study Area > 12 nm from land: SUA
Air Defense Exercise (ADEX)
100
None
Study Area > 12 nm from land: SUA
4,800
None
Study Area > 12 nm from land: SUA
36
9,000 rounds
Study Area SUA > 12 nm from land
18
36 explosive missiles
Study Area SUA > 12 nm from land
5
40 rounds
Study Area SUA > 12 nm from land
12
24,000 rounds
Study Area SUA > 12 nm from land
15
15 explosive missiles
Study Area SUA > 12 nm from land
Anti-Air Warfare (AAW)
Air Intercept Control (AIC)
Gunnery Exercise (Air-to-Air) – Mediumcaliber
(GUNEX [A-A]) Medium-caliber
Missile Exercise (Air-to-Air) (MISSILEX [A-A])
Gunnery Exercise (Surface-to-Air) – Largecaliber
(GUNEX [S-A]) – Large-caliber
Gunnery Exercise (Surface-to-Air) – Mediumcaliber
(GUNEX [S-A]) – Medium-caliber
Missile Exercise (Surface-to-Air)
(MISSILEX [S-A])
2-23
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-8: Training Activities Occurring in the Action Area (continued)
Proposed Action
Range Activity
No. of activities
(per year)
Ordnance
(Number per year)
Location
None
MIRC; Rota Airport
Strike Warfare (STW)
Combat Search and Rescue
80
Amphibious Warfare (AMW)
Amphibious Rehearsal, No Landing – Marine
Air Ground Task Force
12
None
Study Area and Nearshore
Amphibious Assault
6
Blanks; Simunitions
MIRC; Tinian; Guam
Amphibious Raid
6
Blanks; Simunitions
MIRC; Tinian; Guam; Rota
Gunnery Exercise (Air-to-Surface) – Smallcaliber (GUNEX [A-S]) – Small-caliber
242
48,040 rounds
Study Area SUA > 12 nm from land
Gunnery Exercise (Air-to-Surface) – Mediumcaliber (GUNEX [A-S]) – Medium-caliber
295
36,650 (7,150
explosive)
Study Area SUA > 12 nm from land;
Transit Corridor
Missile Exercise (Air-to-Surface) – Rocket
(MISSILEX [A-S] – Rocket)
3
114 rockets (114
explosive)
Study Area SUA > 12 nm from land
Missile Exercise (Air-to-Surface) (MISSILEX
[A-S])
20
20 explosive missiles
Study Area SUA > 12 nm from land
Laser Targeting (at sea)
600
None
Study Area SUA > 12 nm from land
Bombing Exercise (Air-to-Surface)
(BOMBEX [A-S])
37
368 NEPM 184
explosive
Study Area > 50 nm from land
Torpedo Exercise (Submarine-to-Surface)
5
10 EXTORP
Study Area > 3 nm from land
Missile Exercise (Surface-to-Surface)
(MISSILEX [S-S])
12
12 Missiles explosive
Study Area > 50 nm from land
Anti-Surface Warfare (ASUW)
2-24
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-8: Training Activities Occurring in the Action Area (continued)
Proposed Action
Range Activity
No. of activities
(per year)
Ordnance
(Number per year)
Location
Gunnery Exercise (Surface-to-Surface) Ship
– Large-caliber (GUNEX [S-S] – Ship) Largecaliber
140
5,698 rounds
(500 explosive)
Study Area SUA > 12 nm from land;
Transit Corridor
Gunnery Exercise (Surface-to-Surface) Ship
– Small- and Medium-caliber (GUNEX [S-S] –
Ship) Small- and Medium-caliber
100
21,900 rounds
(900 explosive)
Study Area SUA > 12 nm from land;
Transit Corridor
2
28 explosive Bombs
42 explosive Missiles
800 explosive Largecaliber rounds
2 MK-48 explosive
4 explosive
Demolitions
Study Area > 50 nm from land and > 1,000
fathoms depth
Medium-caliber
10
2,100
(100 explosive)
Study Area SUA > 12 nm from land;
Transit Corridor
Small-caliber
40
36,000 rounds
Study Area > 3 nm from land; Transit
Corridor
40
200 G911 antiswimmer grenade
Study Area; MIRC
Anti-Surface Warfare (ASUW) (continued)
Sinking Exercise (SINKEX)
Representative ordnance. Actual ordnance
used will vary (typically less than shown).
Gunnery Exercise
(Surface-to-Surface)
Boat – Small and
Medium-caliber
(GUNEX [S-S] – Boat
Maritime Security Operations
(MSO)
2-25
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-8: Training Activities Occurring in the Action Area (continued)
Proposed Action
Range Activity
No. of activities
(per year)
Ordnance
(Number per year)
Location
Tracking Exercise –Helicopter (TRACKEX –
Helo)
62
None/
REXTORP
Study Area > 3 nm from land; Transit
Corridor
Torpedo Exercise – Helicopter
(TORPEX – Helo)
4
4 EXTORP
Study Area > 3 nm from land
11
None
Study Area > 3 nm from land
Tracking Exercise – Maritime Patrol Aircraft
(TRACKEX – Maritime Patrol Aircraft)
34
None/
REXTORP
Study Area > 3 nm from land
Torpedo Exercise – Maritime Patrol Aircraft
(TORPEX – Maritime Patrol Aircraft)
4
4 EXTORP
Study Area > 3 nm from land
CG/DDG-92
FFG-30
LCS-10
None/
REXTORP
Study Area > 3 nm from land
3
3 EXTORP
Study Area > 3 nm from land
Anti-Submarine Warfare (ASW)
Tracking Exercise – Maritime Patrol
Advanced Extended Echo Ranging
Sonobuoys
Tracking Exercise –Surface
(TRACKEX – Surface)
Torpedo Exercise – Surface
(TORPEX – Surface)
2-26
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-8: Training Activities Occurring in the Action Area (continued)
Proposed Action
Range Activity
No. of activities
(per year)
Ordnance
(Number per year)
Location
Tracking Exercise – Submarine
(TRACKEX – Sub)
12
None
Study Area > 3 nm from land; Transit
Corridor
Torpedo Exercise – Submarine (TORPEX –
Sub)
10
40 MK-48 EXTORP
Study Area > 3 nm from land
Joint Expeditionary Exercise
1
Note 1
Study Area; MIRC
Joint Multi-Strike Group Exercise
1
Note 1
Study Area; MIRC
Marine Air Ground Task Force Exercise
(Amphibious) – Battalion
4
Note 1
Study Area to nearshore; MIRC; Tinian;
Guam; Rota; Saipan; FDM
Special Purpose Marine Air Ground Task
Force Exercise
2
Note 1
Study Area to nearshore; MIRC; Tinian;
Guam; Rota; Saipan
480
None
Study Area
Anti-Submarine Warfare (ASW) (continued)
Major Training Events
Electronic Warfare (EW)
Electronic Warfare Operations (EW Ops)
2-27
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-8: Training Activities Occurring in the Action Area (continued)
Proposed Action
Range Activity
No. of activities
(per year)
Ordnance
(Number per year)
Location
3,200
25,600 cartridges
Study Area > 12 nm from land
Counter Targeting Chaff Exercise
(CHAFFEX) – Ship
40
240 cartridges
Study Area > 12 nm from land
Counter Targeting Chaff Exercise
(CHAFFEX) –Aircraft
3,200
25,600 cartridges
Study Area > 12 nm from land
Civilian Port Defense
1
Note 1
Mariana littorals; MIRC; Inner and Outer
Apra Harbor
Mine Laying
4
480 mine shapes
MIRC Warning Areas
Mine Neutralization – Explosive Ordnance
Disposal (EOD)
20
20 explosive charges
MIRC mine neutralization sites, 20 lb. NEW
maximum
(Piti site is 10 lb. NEW maximum)
Limpet Mine Neutralization System/Shock
Wave Generator
40
40 charges
Mariana littorals; Inner and Outer Apra
Harbor
Submarine Mine Exercise
16
n/a
Study Area; nearshore
Electronic Warfare (EW) (continued)
Counter Targeting Flare Exercise (FLAREX)
– Aircraft
Mine Warfare (MIW)
2-28
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-8: Training Activities Occurring in the Action Area (continued)
Proposed Action
Range Activity
No. of activities
(per year)
Ordnance
(Number per year)
Location
Airborne Mine Countermeasure – Mine
Detection
4
n/a
Study Area; nearshore
Mine Countermeasure Exercise – Towed
Sonar (AQS-20, LCS)
4
n/a
Study Area
Mine Countermeasure Exercise – Surface
(SMCMEX) Sonar (SQQ-32, MCM)
4
n/a
Study Area
Mine Neutralization – Remotely Operated
Vehicle Sonar (ASQ-235 [AQS-20], SLQ-48)
4
4 explosive
neutralizers
Study Area
Mine Countermeasure – Towed Mine
Detection
4
n/a
Study Area
Personnel Insertion/
Extraction
240
None
MIRC; Guam; Tinian; Rota
Underwater Demolition Qualification/
Certification
30
30 explosive charges
MIRC underwater demolition sites, 20 lb.
NEW maximum charge (except Piti 10 lb.
NEW maximum)
Mine Warfare (MIW) (continued)
Naval Special Warfare (NSW)
2-29
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-8: Training Activities Occurring in the Action Area (continued)
Proposed Action
Range Activity
No. of activities
(per year)
Ordnance
(Number per year)
Location
Surface Ship Sonar Maintenance
42
None
Study Area > 3 nm from land; Inner Apra
Harbor; Transit Corridor
Submarine Sonar Maintenance
48
None
Study Area > 3 nm from land; Inner Apra
Harbor; Transit Corridor
6
2,100 small-caliber
rounds
Study Area > 3 nm from land
12
4,000 blank rounds
Study Area
Submarine Navigation
8
None
Apra Harbor and Mariana littorals
Search and Rescue At Sea
40
None
Study Area
Precision Anchoring
18
None
Apra Harbor; Mariana Islands anchorages
Other
Small Boat Attack
Notes: (1) Exercise is composed of various activities accounted for elsewhere within Table 2-8
(2) Discussed as an embedded training activity to CHAFFEX/FLAREX in MIRC EIS/OEIS Appendix D (Air Quality Calculations and Record of NonApplicability).
(3) CHAFF = Chaff Exercise, EIS = Environmental Impact Statement, EOD = Explosive Ordnance Disposal, EXTORP = Exercise Torpedo, FDM = Farallon de
Medinilla, FLAREX = Flare Exercise, lb. = pounds, LCS = Littoral Combat Ship, MIRC = Mariana Islands Range Complex, n/a = Not Applicable, NEPM = Nonexplosive Practice Munitions, NEW = Net Explosive Weight, nm = nautical miles, OEIS = Overseas Environmental Impact Statement, REXTORP =
Recoverable Exercise Torpedo, SUA = Special Use Airspace
2-30
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-9: Proposed Naval Air Systems Command Testing Activities in the Action Area
Action
Range Activity
No. of activities
(per year)
Ordnance
(Number per year)
Location
Anti-Surface Warfare (ASUW)
Air-to-Surface Missile Test
8
8 harpoon missiles
(4 explosive)
Anti-Submarine Warfare Tracking Test –
Maritime Patrol Aircraft (Sonobuoys)
188
240 IEER
553 SUS
Action Area > 3 nm from land
Anti-Submarine Warfare Torpedo Test
40
40 EXTORP
Action Area > 3 nm from land
Broad Area Maritime Surveillance (BAMS)
Testing – MQ-4C Triton
10
None
Action Area
10
None
Action Area > 3 nm from land
Action Area > 50 nm from land
Anti-Submarine Warfare (ASW)
1
Electronic Warfare (EW)
Flare Test
1
Use of Improved Extended Echo Ranging (IEER) sonobuoys will decrease over time while being replaced by use of Multi-static Active Coherent (MAC)
sonobuoys. MAC buoys employ an electronic acoustic source in place of the explosive source used on the IEER buoys.
Notes: EIS = Environmental Impact Statement, EXTORP = Exercise Torpedo, IEER = Improved Extended Echo Ranging, MAC = Multi-static Active Coherent,
nm = nautical miles, OEIS = Overseas Environmental Impact Statement, SUS = Signal Underwater Sound
2-31
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-10: Proposed Naval Sea Systems Command Testing Activities in the Action Area
Action
Range Activity
No. of activities
(per year)
Ordnance
(Number per year)
Location
Life Cycle Activities
Ship Signature Testing
17
None
Action Area
Anti-Surface Warfare (ASUW)/Anti-Submarine Warfare (ASW) Testing
50
2,000 projectiles
Kinetic Energy Weapon Testing
MIRC > 12 nm from land
1 event total
5,000 projectiles
Torpedo Testing
2
20 torpedoes (8
explosive)
MIRC > 3 nm from land
Countermeasure Testing
2
56 torpedoes
Action Area
At-Sea Sonar Testing
20
None
Action Area
2-32
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-10: Proposed Naval Sea Systems Command Testing Activities in the Action Area (continued)
Action
Range Activity
No. of activities
(per year)
Ordnance
(Number per year)
Location
New Ship Construction
ASW Mission Package Testing
MCM Mission Package Testing
ASUW Mission
Package Testing
33
None
Action Area
32
48 neutralizers (24
explosive)
Action Area
Gun Testing –
Small-caliber
4
Gun Testing –
Medium-caliber
(30 mm)
4
Gun Testing –
Large-caliber
(57 mm)
4
Missile/
Rocket Testing
4
2,000 rounds
4,080 rounds (2,040
explosive)
Action Area; Warning Area > 12 nm from
land
5,600 rounds (3,920
in-air explosive)
32 missiles/
rockets (16 explosive)
Notes: MCM = Mine Countermeasure, MIRC = Mariana Islands Range Complex, mm = millimeters, nm = nautical miles
2-33
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 2-11: Proposed Office of Naval Research Testing Activities in the Action Area
Action
Range Activity
No. of activities
(per year)
Ordnance
(Number per year)
Location
Shipboard Protection Systems and Swimmer Defense Testing
Pierside Integrated Swimmer Defense
11
None
Inner Apra Harbor
1
n/a
Action Area
Office of Naval Research
North Pacific Acoustic Lab Philippine Sea
2018–19 Experiment (Deep Water)
Note: n/a = Not Applicable
2-34
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
3 ESSENTIAL FISH HABITAT
In 1996, the MSA was reauthorized and amended by the Sustainable Fisheries Act (Public Law 104-267).
The reauthorized MSA mandated numerous changes to the existing legislation designed to prevent
overfishing, rebuild depleted fish stocks, minimize bycatch, enhance research, improve monitoring, and
protect fish habitat. One of the most significant mandates in the MSA that came out of the
reauthorization was the EFH provision, which provides the means to conserve fish habitat and promote
sustainable fisheries and their stocks.
The EFH mandate requires that the regional Fishery Management Councils (FMCs), through federal
fishery management plans (FMPs), describe and identify EFH for each federally managed species;
minimize, to the extent practicable, adverse effects on such habitat caused by fishing; and identify other
actions to encourage the conservation and enhancement of such habitats. Congress defines EFH as
“those waters and substrate necessary to fish for spawning, breeding, feeding, or growth to maturity”
(16 U.S.C. §1802(10)). The term “fish” is defined in the MSA as “finfish, mollusks, crustaceans, and all
other forms of marine animals and plant life other than marine mammals and birds.” The regulations for
implementing EFH clarify that “waters” include all aquatic areas and their biological, chemical, and
physical properties, while “substrate” includes the associated biological communities that make these
areas suitable fish habitats (50 C.F.R. §600.10). Habitats used at any time during a species’ life cycle (i.e.,
during at least one of its lifestages) must be accounted for when describing and identifying EFH
(National Marine Fisheries Service 2002). Authority to implement the MSA is given to the Secretary of
Commerce through the National Marine Fisheries Service (NMFS).
The MSA requires federal agencies to consult with NMFS on activities that may adversely affect EFH or
when the NMFS independently learns of a federal activity that may adversely affect EFH. The MSA
defines an adverse effect as “any impact that reduces quality and/or quantity of EFH. Adverse effects
may include direct or indirect physical, chemical, or biological alterations of the waters or substrate and
loss of, or injury to, benthic organisms, prey species and their habitat, and other ecosystem
components, if such modifications reduce the quality and/or quantity of EFH. Adverse effects to EFH
may result from actions occurring within EFH or outside of EFH and may include site-specific or habitatwide impacts, including individual, cumulative, or synergistic consequences of actions” (50 C.F.R.
§600.810).
In addition to EFH designations, areas called Habitat Areas of Particular Concern (HAPCs) are also
designated by the regional FMCs. Designated HAPCs are discrete subsets of EFH that provide extremely
important ecological functions or are especially vulnerable to degradation (50 C.F.R. §600.805-600.815).
Regional FMCs may designate a specific habitat area as a HAPC based on one or more of the following
reasons (National Marine Fisheries Service 2002):
1.
2.
3.
4.
Importance of the ecological function provided by the habitat
The extent to which the habitat is sensitive to human-induced environmental degradation
Whether, and to what extent, development activities are, or will be, stressing the habitat type
Rarity of the habitat type
Categorization of an area as a HAPC does not confer additional protection or restriction to the
designated area.
The area encompassed by the Proposed Action (Study Area) extends through the jurisdiction of the
WPRFMC. As a result, training and testing activities that occur as part of the Proposed Action may have
3-1
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
the potential to affect EFH and HAPCs designated by this Council (Figure 3-1). Maps and figures of the
current designated EFH and HAPC locations in the MITT Study Area are located in this section.
Figure 3-1: Western Pacific Regional Fishery Management Council Jurisdiction within the Mariana Islands
Training and Testing Study Area
3.1
WESTERN PACIFIC REGIONAL FISHERY MANAGEMENT COUNCIL
The WPRFMC has authority over the fisheries based in, and surrounding, the State of Hawaii, the
Territory of American Samoa, the Territory of Guam, the CNMI, and the U.S. Pacific Remote Island Areas
(PRIA) of the Western Pacific Region (Figure 3-2). The PRIA comprise Baker Island, Howland Island, Jarvis
Island, Johnston Atoll, Kingman Reef, Wake Island, Palmyra Atoll, and Midway Atoll. The WPRFMC
developed a Fishery Ecosystem Plan (FEP) as an FMP, consistent with the MSA and the national
standards for fishery conservation and management (Western Pacific Regional Fishery Management
Council 2009). Since the 1980s, the Council has managed fisheries throughout the Western Pacific
3-2
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Region through separate species-based FMPs—the Bottomfish and Seamount Groundfish FMP (Western
Pacific Regional Fishery Management Council 1986a), the Crustaceans FMP (Western Pacific Regional
Fishery Management Council 1981), the Precious Corals FMP (Western Pacific Regional Fishery
Management Council 1979), the Coral Reef Ecosystems (CRE) FMP (Western Pacific Regional Fishery
Management Council 2001), and the Pelagic FMP (Western Pacific Regional Fishery Management
Council 1986b).
Figure 3-2: Western Pacific Regional Fishery Management Council Geographic Area
However, the WPRFMC is now moving towards an ecosystem-based approach to fisheries management
and is restructuring its management framework from species-based FMPs to place-based FEPs.
Recognizing that a comprehensive ecosystem approach to fisheries management must be initiated
through an incremental, collaborative, and adaptive management process, a multi-step approach is
being used to develop and implement the FEPs. To be successful, this will require increased
3-3
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
understanding of a range of issues including biological and trophic relationships, ecosystem indicators
and models, and the ecological effects of non-fishing activities on the marine environment.
The Mariana Archipelago FEP establishes the framework under which the WPRFMC will manage fishery
resources, and begin the integration and implementation of ecosystem approaches to management in
the Mariana Archipelago. This FEP does not establish any new fishery management regulations at this
time, but rather consolidates existing fishery regulations for demersal species. Specifically, this FEP
identifies as Management Unit Species (MUS) those species known to be present in waters around
Guam and the CNMI and incorporates all of the management provisions of the Bottomfish and
Seamount Groundfish FMP, the Crustaceans FMP, the Precious Corals FMP, and the Coral Reef
Ecosystem Fishery Management Plan (CRE FMP) that are applicable to the area. Although pelagic fishery
resources play an important role in the biological as well as the socioeconomic environment of these
islands, they will be managed separately through the Pacific Pelagic FEP.
The EFH designations were developed by the WPRFMC and approved by the Secretary of Commerce.
EFH designations for Bottomfish and Seamount Groundfish, Crustaceans, and Precious Corals were
partially approved by the Secretary on 3 February 1999, under Amendment 6 (64 Federal Register [F.R.]
19067-02). Disapproved sections include the bycatch provisions of Amendment 6 to the FMP for
Bottomfish and Seamount groundfish, as well as those for Amendment 8 to the Pelagic FMP. Also
disapproved were the criteria for identifying when overfishing would occur in the bottomfish, pelagics,
and crustacean fisheries.
3.1.1
3.1.1.1
BOTTOMFISH MANAGEMENT UNIT
Description and Identification of Essential Fish Habitat
Unlike the U.S. mainland, with its continental shelf ecosystems, Pacific islands are primarily volcanic
peaks with steep drop-offs and limited shelf ecosystems. The Bottomfish Management Unit Species
(BMUS) under the WPFRMC’s jurisdiction are found concentrated on the steep slopes of deepwater
banks. The 100-fathom isobath is commonly used as an index of bottomfish habitat. Adult bottomfish
are usually found in habitats characterized by a hard substrate of high structural complexity. The total
extent and geographic distribution of the preferred habitat of bottomfish is not well known. Bottomfish
populations are not evenly distributed within their natural habitat; instead, they are found dispersed in
a non-random, patchy fashion (Western Pacific Regional Fishery Management Council 2009).
There is regional variation in species composition, as well as a relative abundance of the MUS of the
deepwater bottomfish complex in the Western Pacific Region (Western Pacific Regional Fishery
Management Council 2009). In American Samoa, Guam, and the Northern Mariana Islands, the
bottomfish fishery can be divided into two distinct fisheries: a shallow- and a deep-water bottomfish
fishery, based on species and depth. The shallow-water (0–100 meters [m]) bottomfish complex
comprises groupers, snappers, and jacks in the genera Lethrinus, Lutjanus, Epinephelus, Aprion, Caranx,
Variola, and Cephalopholis (Table 3-1). The deep-water (100–400 m) bottomfish complex comprises
primarily snappers and groupers in the genera Pristipomoides, Etelis, Aphareus, Epinephelus, and
Cephalopholis (Table 3-1).
3-4
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 3-1: Essential Fish Habitat and Habitat Areas of Particular Concern Designations for the Mariana Archipelago Fishery Ecosystem Plan Management
Unit
Management Unit
Species Complex
Shallow-water species (0–50 fm): uku (Aprion
virescens), thicklip trevally (Pseudocaranx dentex),
giant trevally (Caranx ignoblis), black trevally
(Caranx lugubris), amberjack (Seriola dumerili),
taape (Lutjanus kasmira)
Bottomfish and
Seamount
Groundfish
Deep-water species (50–200 fm): ehu (Etelis
carbunculus), onaga (Etelis coruscans), opakapaka
(Pristipomoides filamentosus), yellowtail kalekale
(P. auricilla), kalekale (P. sieboldii), gindai (P.
zonatus), hapuupuu (Epinephelus quernus), lehi
(Aphareus rutilans)
Seamount groundfish species (50–200 fm):
armorhead (Pseudopentaceros richardsoni),
ratfish/butterfish (Hyperoglyphe japonica), alfonsino
(Beryx splendens)
EFH
Eggs and larvae: the water column
extending from the shoreline to the outer
limit of the EEZ down to a depth of 400 m
Juvenile/adults: the water column and
all bottom habitat extending from the
shoreline to a depth of 400 m
Eggs and larvae: the water column
extending from the shoreline to the outer
limit of the EEZ down to a depth of 400 m
Juvenile/adults: the water column and
all bottom habitat extending from the
shoreline to a depth of 400 m
Eggs and larvae: the (epipelagic zone)
water column down to a depth of 200 m
of all EEZ waters bounded by latitude
29°–35° N and longitude 171° E–179° W,
which is not within the Study Area
boundaries.
Juvenile/adults: all EEZ waters and
bottom habitat bounded by latitude 29°–
35° N and longitude 171° E–179° W
between 80 and 600 m, which is not
within the Study Area boundaries.
3-5
HAPC
All slopes and escarpments
between 40 and 280 m
All slopes and escarpments
between 40 and 280 m
No HAPC designated for
seamount groundfish
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 3-1: Essential Fish Habitat and Habitat Areas of Particular Concern Designations for the Mariana Archipelago Fishery Ecosystem Plan Management
Unit (continued)
Management Unit
Crustaceans
Species Complex
Spiny and slipper lobster:
spiny lobster (P. penicillatus, P. spp.),
ridgeback slipper lobster (Scyllarides haanii),
Chinese slipper lobster (Parribacus antarcticus)
Kona crab:
Kona crab (Ranina ranina)
Deepwater shrimp (Heterocarpus spp.)
EFH
Eggs and larvae: the water column from the
shoreline to the outer limit of the EEZ down to a
depth of 150 m
Juvenile/adults: all of the bottom habitat from the
shoreline to a depth of 100 m
Eggs and larvae: the water column and
associated outer reef slopes between 1550 and
700 m
Juvenile/adults: the outer reef slopes at depths
between 300 and 700 m
Precious
Corals
Coral Reef
Ecosystems
(CRE)
HAPC
No HAPC designated within
the Study Area.
No HAPC designated for
deepwater shrimp.
Little information is available on the CNMI precious coral fishery. The steep topography around the islands limits the available
habitat for precious coral. Since World War II no known precious coral harvests have occurred within the EEZ waters around CNMI.
Therefore, there is no known precious coral fishery in the Mariana Archipelago.
All Currently Harvested Coral Reef Taxa
(CHCRT)
All Potentially Harvested Coral Reef Taxa
(PHCRT)
Temperate species
Tropical species
Pelagic
Sharks
Squid
EFH for the Coral Reef Ecosystem MUS includes
the water column and all benthic substrate to a
depth of 100 m from the shoreline to the outer
limit of the EEZ
Eggs and larvae: the water column extending
from the shoreline to the outer limit of the EEZ
down to a depth of 200 m
Juvenile/adults: the water column extending
from the shoreline to a depth of 1,000 m
Includes all no-take Marine
Protected Areas identified
in the CRE-FMP, all Pacific
remote islands, as well as
numerous existing Marine
Protected Areas, research
sites, and coral reef
habitats throughout the
western Pacific
Water column down to
1,000 m that lies above
seamounts and banks
Notes: HAPC= Habitat Area of Particular Concern, CRE = Coral Reef Ecosystem, FMP= Fishery Management Plan, MITT= Mariana Islands Training and Testing, EEZ=
Exclusive Economic Zone, fm = fathoms, ft. = feet, CNMI = Commonwealth of the Northern Mariana Islands, m = meters, N = North, W = West, E = East
Source: Western Pacific Regional Fishery Management Council 2009
3-6
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
To reduce the complexity and the number of EFH designations required for individual species and life
stages, the WPFRMC has designated EFH for bottomfish assemblages pursuant to Section 600.805(b) of
62 F.R. 66551, which is based on ecological relationships between species and their preferred habitats.
The species complex designations include deep-slope bottomfish (shallow water and deep water) and
seamount groundfish complexes. The designation of these complexes is based on the ecological
relationships among species and their preferred habitat. These species complexes are grouped by the
known depth distributions of individual BMUS throughout the Western Pacific Region (Western Pacific
Regional Fishery Management Council 1986a).
Eggs and Larval Lifestages
The eggs and larvae of all BMUS are pelagic, floating at the surface until hatching and subject thereafter
to advection by the prevailing ocean currents. As snapper and grouper larvae are rarely collected in
plankton surveys, it is extremely difficult to study their distribution. Because of the existing scientific
uncertainty about the distribution of the eggs and larvae of bottomfish, the WPFRMC designated the
water column extending from the shoreline to the outer boundary of the Exclusive Economic Zone (EEZ)
and to a depth of 400 m as EFH for shallow-water and deep-water bottomfish eggs and larvae
throughout the Western Pacific Region (see Table 3-1).
Juvenile and Adult Lifestages
Given the uncertainty concerning the life histories and habitat requirements of many BMUS, the
WPFRMC designated EFH for adult and juvenile shallow-water and deep-water bottomfish as the water
column and bottom habitat extending from the shoreline to a depth of 400 m encompassing the steep
drop-offs and high-relief habitats that are important for bottomfish throughout the Western Pacific
Region (see Table 3-1).
Limited information is available for various larval, juvenile, and adult bottomfish genera of the shallowwater and deep-water complexes. Within the shallow-water complex, snappers form large aggregations
and groupers/jacks occur in pairs within large aggregations near areas of prominent relief. Spawning
coincides with lunar periodicity corresponding with new/full moon events. Groupers have been shown
to undergo small, localized migrations of several kilometers to spawn. Large jacks are highly mobile,
wide-ranging predators that inhabit the open waters above the reef or swim in upper levels of the open
sea and spawn at temperatures of 18–30 degrees Celsius (°C). Within the deep-water complex, snappers
aggregate near areas of bottom relief as individuals or in small groups. Snappers may be batch or serial
spawners, spawning multiple times over the course of the spawning season (spring and summer peaking
in November and December), exhibit a shorter, more well-defined spawning period (July–September), or
have a protracted spawning period (June–December, peaking in August). Some snappers display a
crepuscular periodicity (active during twilight hours) and migrate diurnally from areas of high relief
during the day at depths of 100–200 m to shallow (30–80 m), flat shelf areas at night. Other snapper
species exhibit higher densities on up-current side islands, banks, and atolls (Moffitt 1993).
Seamount Groundfish (All Lifestages)
The life histories and distributional patterns of species in the seamount groundfish management unit are
poorly understood. Data are lacking on the effects of oceanographic variability on migration and
recruitment of individual species. On the basis of the best available data, the WPFRMC designated EFH
for the adult life stage of the seamount groundfish complex as all waters and bottom habitat bounded
by latitude 29 degrees (°) North (N)–35° N and longitude 171° East (E)–179° West (W), which is not
within the Study Area boundaries, and within the depth range of 80–600 m. Essential Fish Habitat for
3-7
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
eggs, larvae, and juveniles is the epipelagic zone (surface to 200 m) of all waters bounded by latitude
29° N–35° N and longitude 171° E–179° W.
3.1.1.2
Habitat Areas of Particular Concern
On the basis of the known distribution and habitat requirements of adult bottomfish, the WPFRMC
designated all escarpments/slopes between 40 and 280 m throughout the Western Pacific Region,
including the Mariana Archipelago, as bottomfish HAPC (see Table 3-1). The designation is based on the
ecological function that these areas provide, the rarity of the habitat, and the susceptibility of these
areas to human-induced environmental degradation. In contrast, flat featureless bottom areas are
thought to provide low-value fishery habitat. However, the recent discovery of concentrations of
juvenile snappers in relatively shallow water and featureless bottom habitat indicates the need for more
research to help identify, map, and study nursery habitat for juvenile snapper.
No HAPC has been designated for species in the seamount groundfish management unit.
3.1.1.3
Figures and Maps
Figure 3-3 shows the EFH for all eggs and larval lifestage of bottomfish designated on Guam, Tinian, and
Farallon de Medinilla (FDM). Figure 3-4 shows the EFH for all juvenile and adult lifestage of bottomfishes
designated on FDM. Figures 3-5 and 3-6 show the EFH and HAPC designated on Guam and Tinian for all
juvenile and adult lifestages of bottomfish.
3-8
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-3: Essential Fish Habitat for All Eggs and Larval Lifestages of Bottomfish Designated on Guam, Tinian,
and Farallon de Medinilla
3-9
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-4: Essential Fish Habitat for All Juvenile and Adult Lifestages of Bottomfishes Designated on Farallon de
Medinilla
3-10
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-5: Essential Fish Habitat for All Juvenile and Adult Lifestages of Bottomfish and Habitat Areas of
Particular Concern Designated on Guam
3-11
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-6: Essential Fish Habitat for All Juvenile and Adult Lifestages of Bottomfish and Habitat Areas of
Particular Concern Designated on Tinian
3-12
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
3.1.2
3.1.2.1
CRUSTACEANS MANAGEMENT UNIT
Description and Identification of Essential Fish Habitat
To reduce the complexity and the number of EFH designations required for individual species and life
stages, the WPRFMC has divided EFH for crustaceans into three complexes: spiny and slipper lobster,
Kona crab, and deepwater shrimp. The designations are based on the ecological relationships among
species and their preferred habitat.
Spiny lobsters are found throughout the Indo-Pacific region. All spiny lobsters in the Western Pacific
Region belong to the family Palinuridae. The slipper lobsters belong to the closely related family
Scyllaridae. There are 839 species of crustaceans in the Marianas (Paulay et al. 2003). Thirteen species of
spiny lobster occur in the tropical and subtropical Pacific between 35° N and 35° South (S) (Western
Pacific Regional Fishery Management Council 2009). In the southwestern region of the North Pacific,
spiny lobsters are typically found in association with coral reefs. Coral reefs provide shelter as well as a
diverse and abundant supply of food. Adult spiny lobsters are typically found on rocky substrate in wellprotected areas, in crevices, and under rocks. In CNMI, spiny lobsters have not been found at depths
greater than 42 feet (ft.) (13 m). Spiny lobsters inhabit the rocky shelters in the windward surf zones of
oceanic reefs and move on to the reef flat at night to forage. Five species of spiny lobsters occur in the
Marianas; the Pronghorn spiny lobster (Panulirus penicillatus) is the most common species (Western
Pacific Regional Fishery Management Council 2001; Paulay et al. 2003). While slipper lobsters are
present in Guam and CNMI, the estimated annual biological catches are low; in 2013 it was 80 lb.
(Western Pacific Regional Fishery Management Council 2012).
The Kona crab is found in a number of environments, from sheltered bays and lagoons to surf zones, but
prefers sandy habitat in depths of 24–115 m (Smith 1993, Poupin 1996). The Kona crab spawns at least
twice during the spawning season; there are insufficient data to define the exact spawning season in the
MITT Study Area (Western Pacific Regional Fishery Management Council 2009).
The WPRFMC designated EFH for spiny lobster and Kona crab larvae as the water column from the
shoreline to the outer limit of the EEZ and to a depth of 150 m throughout the Western Pacific Region.
The EFH for juvenile and adult spiny lobster and Kona crab is designated as the bottom habitat from the
shoreline to a depth of 100 m throughout the Western Pacific Region.
There are three species of deep-water shrimp known to occur in the Study Area, Heterocarpus ensifer,
Heterocarpus laevigatus, and Heterocarpus longirostris. These species occur at varying depths, H. ensifer
at 366–550 m, H. laevigatus at 550–915 m, and H. longirostris at depths greater than 915 m (Moffitt and
Polovina 1987). The EFH for deepwater shrimp eggs and larvae is designated as the water column and
associated outer reef slopes between 550 and 700 m, and the EFH for juveniles and adults is designated
as the outer reef slopes at depths between 300 and 700 m (see Table 3-1).
3.1.2.2
Habitat Areas of Particular Concern
No HAPC has been designated for crustaceans in this Management Unit within the Study Area.
3.1.2.3
Figures and Maps
Figure 3-7 shows the EFH for all eggs and larval lifestages of crustaceans designated on Guam, Tinian,
and FDM. Figures 3-8 to 3-10 show the EFH for all juvenile and adult lifestages of crustaceans designated
on Guam, Tinian, and FDM.
3-13
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-7: Essential Fish Habitat for All Eggs and Larval Lifestages of Crustaceans Designated on Guam, Tinian,
and Farallon de Medinilla
3-14
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-8: Essential Fish Habitat for All Juvenile and Adult Lifestages of Crustaceans Designated on Guam
3-15
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-9: Essential Fish Habitat for All Juvenile and Adult Lifestages of Crustaceans Designated on Tinian
3-16
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-10: Essential Fish Habitat for All Juvenile and Adult Lifestages of Crustaceans Designated on Farallon de
Medinilla
3-17
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
3.1.3
CORAL REEF ECOSYSTEMS MANAGEMENT UNIT
In designating EFH for CRE MUS, the WPRFMC used an approach similar to one used by both the South
Atlantic and the Pacific Fishery Management Councils. This approach links the MUSs to specific habitat
“composites” (e.g., sand, live coral, seagrass beds, mangrove, open ocean) for each life history stage,
consistent with the water depth of the ecosystem to 100 m and to the limit of the EEZ.
The CRE FMP manages coral reef ecosystems surrounding the following U.S. Pacific Island areas: the
State of Hawai’i, the Territories of American Samoa and Guam, the CNMI, and the PRIAs of Johnston
Atoll, Kingman Reef, Palmyra and Midway Atolls, and Jarvis, Howland, Baker and Wake Islands (Western
Pacific Regional Fishery Management Council 2001). Under this plan, 80 coral reef species are managed
(Western Pacific Regional Fishery Management Council 2009).
Except for several of the major coral reef associated species, very little is known about the life histories,
habitat utilization patterns, food habits, or spawning behavior of most coral reef associated species. For
this reason, the WPRFMC, through the CRE FMP, designated EFH using a two-tiered approach based on
the division of MUS into the Currently Harvested Coral Reef Taxa (CHCRT) and Potentially Harvested
Coral Reef Taxa (PHCRT) categories. The categories are also consistent with the use of habitat
composites.
3.1.3.1
Currently Harvested Coral Reef Taxa Complex
In the first tier, EFH has been identified for species that are (a) currently being harvested in state and
federal waters and for which some fishery information is available and (b) likely to be targeted in the
near future based on historical catch data. Appendix B summarizes the habitat types used by CHCRT
species.
3.1.3.1.1
Description and Identification of Essential Fish Habitat
To reduce the complexity and the number of EFH identifications required for individual species and life
stages, the WPRFMC has designated EFH for species assemblages pursuant to 50 C.F.R. 600.815
(a)(2)(ii)(E). The designation of these assemblages or complexes is based on the ecological relationships
among species and their preferred habitat. These species complexes are grouped by the known depth
distributions of individual MUS. EFH for the Coral Reef Ecosystem MUS includes the water column and
all benthic substrate to a depth of 100 m from the shoreline to the outer limit of the EEZ (see Table 3-1).
3.1.3.1.2
Habitat Areas of Particular Concern
Because of the already-noted lack of scientific data, the WPRFMC considered HAPC as those locations
that are known to support populations of Coral Reef Ecosystem MUS and those that meet the NMFS
criteria for HAPC. The WPRFMC considered designating areas that are already protected—for example,
wildlife refuges—as HAPC, even though this is not one of the criteria NMFS uses to establish HAPC. The
Coral Reef Ecosystem MUS HAPCs for the Marianas identified in Table 3-2 have met at least one of the
four criteria (rarity of habitat, ecological function, susceptibility to human impact, likelihood of
developmental impacts), or the fifth criterion (i.e., protected areas) identified by the WPRFMC.
However, a great deal of life history work needs to be done in order to adequately identify the extent of
HAPCs and link them to particular species or life stages.
3.1.3.2
Figures and Maps
Figures 3-11 to 3-20 show the EFH for various species and lifestage of the CHCRT and HAPC designated
on Guam, Tinian, and FDM.
3-18
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-11: Essential Fish Habitat for Various Lifestages of the Currently Harvested Coral Reef Taxa-Coral Reef
Ecosystem) on Guam, Tinian, and Farallon de Medinilla
3-19
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-12: Essential Fish Habitat for All Juvenile and Adult Lifestages of the Currently Harvested Coral Reef
Taxa-Coral Reef Ecosystem on Guam
3-20
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-13: Essential Fish Habitat for All Juvenile and Adult Lifestages of Flagtails and Mullets (Currently
Harvested Coral Reef Taxa-Coral Reef Ecosystem) on Guam
3-21
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-14: Essential Fish Habitat for All Adult Lifestages of Rudderfishes (Currently Harvested Coral Reef TaxaCoral Reef Ecosystem) on Guam
3-22
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-15: Essential Fish Habitat for All Juvenile and Adult Lifestages of the Currently Harvested Coral Reef
Taxa-Coral Reef Ecosystem on Tinian
3-23
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-16: Essential Fish Habitat for All Juvenile and Adult Lifestages of Flagtails and Mullets (Currently
Harvested Coral Reef Taxa-Coral Reef Ecosystem) on Tinian
3-24
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-17: Essential Fish Habitat for All Adult Lifestages of Rudderfishes (Currently Harvested Coral Reef TaxaCoral Reef Ecosystem) on Tinian
3-25
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-18: Essential Fish Habitat for All Juvenile and Adult Lifestages of the Currently Harvested Coral Reef
Taxa-Coral Reef Ecosystem on Farallon de Medinilla
3-26
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-19: Essential Fish Habitat for All Juvenile and Adult Lifestages of the Flagtails and Mullets (Currently
Harvested Coral Reef Taxa-Coral Reef Ecosystem) on Farallon de Medinilla
3-27
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-20: Essential Fish Habitat for All Adult Lifestages of Rudderfishes (Currently Harvested Coral Reef TaxaCoral Reef Ecosystem) on Farallon de Medinilla
3-28
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
3.1.3.3
3.1.3.3.1
Potentially Harvested Coral Reef Taxa Complex
Description and Identification of Essential Fish Habitat
EFH has also been designated for the second tier, PHCRT. These taxa include thousands of species
encompassing almost all coral reef associated fauna and flora. However, there is very little scientific
knowledge about the life histories and habitat requirements of the thousands of species that compose
these taxa. In fact, a large percentage of these biota have not been described by science. Therefore, the
WPRFMC has used the precautionary approach in designating EFH for PHCRT so that enough habitat is
protected to sustain managed species. Appendix B summarizes the habitat types used by PHCRT species.
The designation of EFH for PHCRT in Guam and the Mariana Islands is the same as the EFH designation
for CHCRT (see Table 3-1).
3.1.3.3.2
Habitat Areas of Particular Concern
Because of the aforementioned lack of scientific data, the WPRFMC considered locations that are known
to support populations of Coral Reef Ecosystem MUS and meet NMFS criteria for HAPC. The designation
of HAPCs for PHCRT in Guam and the Mariana Islands is the same as the HAPC designations for CHCRT
(Table 3-2). However, a great deal of life history work needs to be done in order to adequately identify
the extent of HAPCs and link them to particular species or life stages.
3.1.3.3.3
Figures and Maps
Figure 3-21 shows the EFH for all lifestages of the PHCRT and HAPC designated on Guam, Tinian, and
FDM.
3-29
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-21: Essential Fish Habitat for All Lifestages of the Potentially Harvested Coral Reef Taxa-Coral Reef
Ecosystem and Habit Areas of Particular Concern Designated on Guam, Tinian, and Farallon de Medinilla
3-30
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 3-2: Coral Reef Ecosystem Habitat Areas of Particular Concern Criteria Designations in the Mariana
Archipelago
Guam
Cocos Lagoon
Orote Point Ecological Reserve
Area
Haputo Point Ecological
Reserve Area
Ritidian Point
Jade Shoals
CNMI
Saipan (Saipan Lagoon)
Rarity of
Habitat
Ecological
Function
Susceptibility
to Human
Impact
X
X
X
X
X
X
X
X
X
X
X
X
X
X
Likelihood of
Developmental
Impacts
Existing
Protective
Status
X
X
X
X
X
X
X
X
(Managaha
Marine
Conservation
Area)
Note: CNMI = Commonwealth of the Northern Mariana Islands
Source: Western Pacific Regional Fishery Management Council 2009
3.1.4
3.1.4.1
PELAGIC MANAGEMENT UNIT
Description and Identification of Essential Fish Habitat
The WPRFMC has used the best available scientific information to describe EFH in text and tables that
provide information on the biological requirements for each life stage (egg, larvae, juvenile, adult) of all
Pelagic Management Unit Species (PMUS). Careful judgment was used in determining the extent of the
EFH that should be designated to ensure that sufficient habitat in good condition is available to maintain
a sustainable fishery and the managed species’ contribution to a healthy ecosystem.
Pelagic fish occur in tropical and temperate waters of the Western Pacific Ocean. Geographical
distribution among the pelagic species is governed by seasonal changes in ocean temperature. These
species range from as far north as Japan, to as far south as New Zealand. Albacore tuna (Thunnus
alalunga), striped marlin (Tetrapurus audax), and broadbill swordfish (Xiphias gladius) have broader
ranges and occur from 50° N to 50° S (Western Pacific Regional Fishery Management Council 1998).
The pelagic species are typically found in epipelagic to pelagic waters; however, shark species can be
found in inshore benthic (bottom habitats), neritic (nearshore) to epipelagic (open ocean shallow zone),
and mesopelagic waters (open ocean zone with reduced light penetration). Gradients in temperature,
oxygen, or salinity can affect the suitability of a habitat for pelagic fish. Skipjack tuna (Katsuwonus
pelamis), yellowfin tuna (T. albacares), and Indo-Pacific blue marlin (Makaira nigricans) prefer warm
surface layers, where the water is well mixed and relatively uniform in temperature. Species such as
albacore tuna (Thunnus alalunga), bigeye tuna (Thunnus obesus), striped marlin (Kaijika audax), and
broadbill swordfish (Xiphias gladius) prefer cooler temperate waters associated with higher latitudes
and greater depths. Certain species are known to aggregate near the surface at night. However, during
the day, broadbill swordfish can be found at depths of 800 m, while bigeye tuna can be found around
275–550 m. Juvenile albacore tuna generally concentrate above 90 m with adults found in deeper
waters (90–275 m) (Western Pacific Regional Fishery Management Council 2009).
To reduce the complexity and the number of EFH identifications required for individual species and life
stages, the WPRFMC has designated EFH for pelagic species assemblages pursuant to Section 600.805(b)
3-31
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
of 62 F.R. 66551. The species complex designations for the PMUS are temperate species, tropical
species, sharks, and squid (Table 3-3). The designation of these complexes is based on the ecological
relationships among species and their preferred habitat. The marketable species complex has been
subdivided into tropical and temperate assemblages. The temperate species complex includes those
PMUS that are found in greater abundance in higher latitudes such as swordfish and bigeye, bluefin, and
albacore tuna.
Migration and life history patterns of most pelagic fish are poorly understood in the Pacific Ocean.
Additionally, very little is known about the distribution and habitat requirements of the juvenile life
stages of tuna and billfish prior to recruitment into fisheries. Seasonal movements of cooler-water tunas
such as the northern bluefin and albacore are more predictable and better defined than billfish
migrations. Tuna and related species tend to move toward the poles during the warmer months and
return to the equator during cooler months. Most pelagic species make daily vertical migrations,
inhabiting surface waters at night and deeper waters during the day. Spawning for pelagic species
generally occurs in tropical waters but may include temperate waters during warmer months. Very little
is known about the life history stages of species that are not targeted by fisheries in the Pacific such as
escolars or sname makerals (Western Pacific Regional Fishery Management Council 2009).
Because of the uncertainty about the life histories and habitat utilization patterns of many PMUS, the
WPRFMC has taken a precautionary approach by adopting a 1,000 m depth as the lower bound of EFH
for PMUS. Although many of the PMUS are epipelagic, some species are known to be present in the
mesopelagic zone (200–1,000 m). Bigeye tuna are abundant at depths in excess of 400 m and swordfish
have been tracked to depths of 800 m. Vertically migrating mesopelagic fishes and squids associated
with the deep scattering layer are important prey for PMUS and are most abundant above 1,000 m. The
EFH designation is also based on anecdotal reports from fishermen that PMUS aggregate over high relief
topographical features at water depths of 2,000 m or more. These reports are supported by research
that indicates seabed features with high relief, such as seamounts, exert a strong influence over the
water column adjacent to and above the seamount. Studies have shown that strong mixing often occurs
at the convergence of adjacent currents or water masses, which can take place at oceanic boundaries
along continental slopes, above seamounts and mid-ocean ridges, where other oceanic fronts occur
(e.g., at gyres), and in the mixed layer (Lalli and Parsons 1997; Western Pacific Regional Fishery
Management Council 2009). Mixing results in areas of high primary productivity, which in turn become
foraging ‘hotspots’ for pelagic species, including fishes in the Pelagic Management Unit.
3-32
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 3-3: Essential Fish Habitat and Habitat Area of Particular Concern designated by Western Pacific Regional
Fishery Management Council
Species Complex
EFH
HAPC
Temperate species
Albacore (Thunnus alalunga)
Bigeye tuna (Thunnus obesus)
Bluefin tuna (Thunnus thynnus)
Mackerel (Scomber spp.)
Pomfret (family Bramidae)
Striped marlin (Tetrapurus audax)
Swordfish (Xiphias gladius)
Tropical species
Black marlin (Makaira indica)
Blue marlin (Makaira nigricans)
Dogtooth tuna (Gymnosarda unicolor)
Frigate and bullet tunas (Auxis thazard, A. rochei)
Kawakawa (Euthynnus affinis)
Mahimahi (Coryphaena hippurus, C. equiselas)
Ono (Acanthocybium solandri)
Opah (Lampris spp.)
Sailfish (Istiophorus platypterus)
Skipjack (Katsuwonus pelamis)
Slender tuna (Allothunnus fallai)
Spearfish (Tetrapturus spp.)
Yellowfin (Thunnus albacares)
Eggs and larvae: the
(epipelagic zone) water
column down to a depth of 200
m from the shoreline to the
outer limit of the EEZ
Juvenile/adults: the water
column down to a depth of
1,000 m from the shoreline to
the outer limit of the EEZ
The water column from the
surface down to a depth of
1,000 m above all seamounts
and banks with summits
shallower than 2,000 m within
the EEZ
Sharks
Bigeye thresher shark (Alopias superciliosus)
Blue shark (Prionace glauca)
Thresher shark (Alopias vulpinus)
Longfin mako shark (Isurus paucus)
Oceanic whitetip shark (Carcharhinus longimanus)
Pelagic thresher shark (Alopias pelagicus)
Salmon shark (Lamna ditropis)
Shortfin mako shark (Isurus oxyrinchus)
Silky shark (Carcharhinus falciformis)
Squid
Diamondback squid (Thysanoteuthis rhombus)
Neon flying squid (Ommastrephes bartamii)
Purple flying squid (Sthenoteuthis oualaniensis)
Notes: EFH = Essential Fish Habitat, HAPC = Habitat Area of Particular Concern, EEZ = Exclusive Economic Zone, m = meters
Source: Western Pacific Regional Fishery Management Council 2009
The eggs and larvae of all teleost PMUS are pelagic. Eggs are slightly buoyant when first spawned, are
spread throughout the mixed layer, and are subject to advection by the prevailing ocean currents.
Because the eggs and larvae of the PMUS are found distributed throughout the tropical (and in summer,
the subtropical) epipelagic zone, EFH for these life stages has been designated as the epipelagic zone
(~200 m) from the shoreline to the outer limit of the EEZ. The EFH for juveniles and adults of PMUS is
3-33
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
the water column extending from the shoreline to a depth of 3,280 ft. (1,000 m). See Appendix B for
additional details on the life history and habitat utilization patterns of individual PMUS.
3.1.4.2
Habitat Areas of Particular Concern
The WPRFMC designated the water column to a depth of 1,000 m above all seamounts and banks within
the EEZ shallower than 2,000 m from the surface as HAPC for PMUS.
The relevance of topographic features deeper than 1,000 m is due to the influence they have on the
overlying mesopelagic zone. These deeper features (e.g., seamounts) are not designated as EFH or
HAPC, but the waters down to 1,000 m that are designated as HAPC can be influenced by topographic
features extending below 1,000 m. The 2,000 m depth contour captures the summits of most
seamounts and all banks within the EEZ waters under the WPRFMC’s jurisdiction. The basis for
designating these areas as HAPC is the ecological function provided, the rarity of the habitat type, the
susceptibility of these areas to human-induced environmental degradation, and proposed activities that
may stress the habitat type.
Because the PMUS are highly migratory, the areas outside the EEZ in the Western Pacific Region are
designated by the WPRFMC as “important habitat” because they provide essential spawning, breeding,
and foraging habitat.
3.1.4.3
Figures and Maps
Figure 3-22 shows the EFH for all lifestages of pelagic fishes designated on Guam, Tinian, and FDM.
3-34
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-22: Essential Fish Habitat for All Lifestages of Pelagic Fishes Designated on Guam, Tinian, and Farallon
de Medinilla
3-35
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
3.2
DESCRIPTION OF HABITATS
The Study Area covers a range of marine habitats which support a myriad of fish and invertebrate
communities. Waters of the Study Area include shoreline or littoral habitats between the mean high and
low water lines, bottom habitats below the mean high water line, and the overlying water column.
For littoral and bottom habitats, the habitat classification system described herein is a modified version
of the Classification of Wetlands and Deepwater Habitats of the United States (Cowardin et al. 1979).
The structure of the original classification system allows it to be used at any of several hierarchical
levels. The original classification employs 5 system names, 8 subsystem names, 11 class names, 28
subclass names, and an unspecified number of dominance types. The modified classification system
starts at the subsystem level (e.g., intertidal shores/subtidal bottoms) and focuses analysis on a
modified class level (e.g., soft shores/bottoms, hard shores/bottoms) and differentiates non-living
substrates from living structures on the substrate. Living structures on the substrate are termed
biogenic habitats, and include wetland plants, submerged aquatic vegetation (attached macroalgae and
rooted vascular plants), sedentary invertebrate beds, and reefs. As such, these classifications may or
may not overlap with the Coastal and Marine Ecological Classification Standard (Federal Geographic
Data Committee 2012) catalog of terms that provides a means for classifying ecological units using a
simple, standard format and common terminology. Therefore, Table 3-4 aligns the habitat groupings
used in this analysis with the Coastal and Marine Ecological Classification Standard Classifications.
Table 3-4: Coastal and Marine Ecological Classification Standard Crosswalk
MITT EIS/OEIS
Habitat Type and
Subtypes
Soft Shores
1
Beach
Tidal Delta/mudflats
and tidal riverine
and estuarine
streambeds
Relationship
to CMECS
CMECS Class/
Subclass
Confidence
Relationship
Notes
CMECS
Unconsolidated
Substrate =
Cowardin
Unconsolidated
Shore +
Unconsolidated
bottom. Shore is
considered in
the CMECS
Geoform
Component.
<
Unconsolidated
Substrate
Certain
=
Beach
Somewhat
Certain
<
Flat
3-36
Somewhat
Certain
MITT habitat
type = CMECS
ebb tidal delta
flat + flood tidal
delta flat + tidal
flat+ wind tidal
flat
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 3-4: Coastal and Marine Ecological Classification Standard Crosswalk (continued)
MITT EIS/OEIS
Habitat Type and
Subtypes
Rocky Shores
1
Relationship
to CMECS
CMECS Class/
Subclass
Confidence
Relationship
Notes
CMECS Rock
substrate =
Cowardin Rocky
Shore + Rock
Bottom. Shore is
considered in
the CMECS
Geoform
Component.
<
Rock Substrate
Certain
Vegetated Shores
1
=
Emergent Wetland
Certain
Salt/Brackish Marsh
≈
Emergent Tidal Marsh
Somewhat
Certain
Mangrove
Aquatic Beds
1
>
Tidal Mangrove Forest,
Tidal Mangrove
Shrubland
Somewhat
Certain
=
Aquatic Vegetation Bed
Certain
Seagrass
≈
Aquatic Vascular
Vegetation
Somewhat
Certain
Sargassum
<
Bethic Macroalgae
Somewhat
Certain
<
Unconsolidated
Substrate
Certain
Lagoons
≈
Lagoon
Somewhat
Certain
Abyssal Plain
≈
Abyssal Plain
Somewhat
Certain
Soft Bottoms
1
3-37
MITT Mangrove
= CMECS Tidal
Mangrove
Shrubland +
Tidal Mangrove
Forest. MITT
Mangrove has
no height
threshold.
MITT Seagrass
= CMECS
Freshwater and
Brackish Tidal
Aquatic
Vegetation +
Seagrass bed.
MITT Seagrass
has no salinity
threshold.
CMECS
Unconsolidated
Substrate =
Cowardin
Unconsolidated
Shore +
Unconsolidated
Bottom
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 3-4: Coastal and Marine Ecological Classification Standard Crosswalk (continued)
MITT EIS/OEIS
Habitat Type and
Subtypes
Mariana Trench
Hard Bottoms
1
Biotic/Reef
Relationship
to CMECS
≈
CMECS Class/
Subclass
Tectonic Trench
Confidence
Relationship
Notes
Somewhat
Certain
CMECS
Tectonic Trench
= General
description of
trenches,
Mariana Trench
is specific to
Study Area.
CMECS Rock
Substrate=
Cowardin Rocky
Shore + Rock
Bottom.
<
Rock Substrate
Certain
≈
Shallow/Mesophotic
Coral Reef Biota
Somewhat
Certain
Seamount (Level 1)
Seamount
Hydrothermal vents
>
>
Hydrothermal Vent
(Level 2), Hydrothermal
Vent Field
(Level 1 and 2)
Somewhat
Certain
MITT Seamount
= CMECS Guyot
+ Knoll +
Pinnacles. MITT
Seamounts does
not have shape
delimiters.
Somewhat
Certain
MITT
Hydrothermal
Vent does not
have a number
of vents
threshold.
Anthropogenic
Substrate =
includes classes
dependent on
the
anthropogenic
material;
however,
materials in the
Study Area vary
Artificial
Structures
<
Anthropogenic
Substrate
Somewhat
Certain
Artificial Reefs
≈
Artificial Reef
Somewhat
Certain
Shipwrecks
≈
Wreck (Level 2)
Somewhat
Certain
FADs
≈
Buoy (Level 2)
Somewhat
Certain
1
These habitat types were derived directly from Cowardin 1979.
Notes: CMECS = Coastal and Marine Ecological Classification Standard, Study Area = Mariana Islands Training
and Testing Study Area, FAD = Fish Aggregating Device
3-38
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
The ecological functions of the substrate and biogenic habitat for managed species and life stages are
implied by their presence, extent and quality within an area. Information documenting habitat presence
within broad geographic areas is widely available, whereas data on spatial extent and quality are sparse
and inconsistently classified. Establishing a proper baseline for impact assessment will be primarily
qualitative for habitats with sparse and inconsistent spatial data (noted in respective habitat section),
and quantitative in some areas.
3.2.1
WATER COLUMN
The flow and quality of water in the water column are key factors linking fish, habitat, and fishery
activities. Water column properties that may affect fisheries resources include temperature, salinity,
dissolved oxygen, total suspended solids, nutrients (nitrogen, phosphorus), and chlorophyll a (Western
Pacific Regional Fishery Management Council 2009). Other factors, such as depth, pH, water velocity and
movement, and water clarity, also affect the distribution of aquatic organisms.
Water column parameters referenced in EFH and HAPCs descriptions include waters (e.g., offshore,
nearshore, estuarine), vertical layers (e.g., epipelagic, benthic, thermocline), and salinity zones (e.g.,
mesohaline). Any reference to water bodies (e.g., all estuaries) implies the inclusion of all shore and
bottom habitats, unless selected habitats are specified (e.g., pelagic/demersal species; Appendix B).
Water types that characterize the Study Area vary along the continuum from estuaries at the mouths of
coastal rivers to offshore ocean waters. Salinity is often used to distinguish bodies of water; however,
salinity “boundaries” are not fixed but fluctuate depending on season, precipitation, winds, and global
climate changes (e.g., El Niño). The salinity of coastal, estuarine waters can vary dramatically from ~0 to
30 practical salinity units (psu) depending on several factors; however, on average salinity increases with
distance from shore. Ocean waters can be defined based on salinity as the water column seaward of
estuarine salinities (i.e., seaward of approximately 30 psu). On average, ocean waters have a salinity of
35 psu. Shallower, nearshore waters in the neritic zone (i.e., waters over the continental shelf) can have
salinities between 27 and 30 psu (Lalli and Parsons 1997). Salinity also varies with depth. In general,
salinity increases with depth.
Essential Fish Habitat designations may refer to ocean zones in defining the EFH. The pelagic zone is
generally regarded as extending from the low tide line seaward and includes the neritic zone and the
ocean zone. Offshore, ocean waters are defined herein as the water column seaward of the neritic zone
(Figure 3-23). Overlap occurs between the nearshore waters of the neritic zone and estuarine systems
where lower salinity plumes from riverine outflow enter continental shelf waters. Bays, inlets, sounds,
tidal creeks, and coastal rivers are characterized by estuarine waters. Offshore, nearshore, and estuarine
waters occur within all fishery management council regions.
3-39
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-23: Three-Dimensional Representation of a Continental Margin and Abyssal Zone
3.2.1.1
Currents, Circulation Patterns, and Water Masses
In ocean waters, large scale gyres and oceanic currents create physical and chemical dynamics that
influence the distribution of organisms. Ocean circulation in the Study Area is dominated by the east to
west motion of the North Equatorial Current (Starmer et al. 2008; Figure 3-24). The North Equatorial
Current is the southern current in the North Pacific Subtropical Gyre which occurs between the equator
and 50° N and is defined to the north by the North Pacific Current, to the east by the California Current,
to the south by the North Equatorial Current, and to the west by the Kuroshio Current (Tomczak and
Godfrey 2005). The North Pacific Subtropical Gyre, like all the ocean’s large subtropical gyres, has
extremely low rates of primary productivity (Valiela 1995) caused by a persistent thermocline (a distinct
layer of water in which temperature changes more rapidly with depth than it does above or below) that
prevents the vertical mixing of water. Thermocline layers are present in the water column at varying
depths throughout the world’s oceans; however, in most areas, particularly nearshore, they are broken
down seasonally, allowing nutrient rich waters below the thermocline to replenish surface waters and
fuel primary production.
Surface currents are horizontal movements of water primarily driven by the drag of the wind over the
sea surface. Wind-driven circulation dominates in the upper 330 ft. (100 m) of the water column and
therefore drives circulation over continental shelves (Hunter et al. 2007). Surface currents of the Pacific
Ocean include equatorial, circumpolar, eastern boundary, and western boundary currents. In the Study
Area, there are persistent trade winds from the east-northeast which generate wind driven waves and
circulation patterns (Starmer et al. 2008). The overall flow in the Study Area is northwestward; however,
3-40
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
very little is known about the oceanic circulation around the islands in the Study Area and the impact
that the eddies that the islands create has upon the circulation of the open ocean (Wolanski et al. 2003).
Similar to cold fronts and warm fronts in the atmosphere, an oceanic front is the boundary between two
water masses with distinct differences in temperature and salinity (i.e., density). An oceanic front is
characterized by rapid changes in water properties over a short distance.
Figure 3-24: Surface Circulation of the Pacific Ocean and Outline of the North Pacific Subtropical Gyre
3.2.1.2
Water Column Characteristics and Processes
The characteristics of the water column are defined by the temperature, salinity, and density of waters
in the region. The physical and chemical properties of the water column affect primary production in the
region.
3-41
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Sea surface temperature varies considerably across the Pacific Ocean with warmest waters near the
equator and coldest waters at the poles. Sea surface temperature also varies seasonally with warmest
temperatures occurring in August/September and coldest in February/March in the Study Area (Pickard
and Emery 1990). Annual changes of a few degrees can also occur at the latitudes in the Study Area, and
diurnal changes in isolated areas of the open ocean are also known to occur. Spatial and temporal
variations in sea surface temperate are greater near the coast than in the open ocean due to factors
such as riverine outflow, shallower waters, and longitudinal currents that transport warmer or cooler
waters along the coast (Pickard and Emery 1990). Sea surface temperatures are also affected by
atmospheric conditions (e.g., winds), which can lead to seasonal upwelling (Tomczak and Godfrey 2005).
In the Study Area, sea surface temperature averages 82 degrees Fahrenheit (°F) (28°C) with little
seasonal variation (Pacific Regional Integrated Sciences and Assessment Program 2012; Figure 3-25).
In the open ocean portion of the Study Area, the water column contains a well-mixed surface layer
extending to a depth of approximately 400 ft. (125 m). Immediately below the mixed layer is a zone
where the temperature declines rapidly with depth, known as the thermocline. In low latitudes, which
include the Study Area, water temperature at the bottom of the thermocline ranges from 40 to 50°F
(5 to 10°C). The temperature of deeper waters (> 1,600 ft. [500 m]) is relatively constant (Pickard and
Emery 1990). Unlike in more temperate climates, the thermocline is relatively stable, rarely breaking
down and allowing the nutrient-rich, deeper waters to mix with surface waters. This constitutes what
has been defined as a “significant” surface duct (a mixed layer of constant temperature extending from
the sea surface to approximately 100 ft. [30 m] or more), which influences the transmission of sound in
the water.
3-42
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-25: Sea Surface Temperature Showing the Seasonal Variation in the Mariana Islands Training and Testing Study Area
3-43
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Seawater is made up of a number of components including gases, nutrients, dissolved compounds,
particulate matter (solid compounds such as sand, marine organisms, and feces), and trace metals
(Garrison 1998). Seawater characteristics are primarily determined by temperature and the gases and
solids dissolved in it.
Seawater is primarily composed of dissolved salts. Chloride, sodium, calcium, potassium, magnesium,
and sulfate make up 98 percent of the solids in seawater, with chloride and sodium making up
85 percent of that total (Garrison 1998, Lalli and Parsons 1997). Sea surface salinity within the Study
Area ranges from 33 to 35 parts per thousand, with lower salinities closer to shore (National Oceanic
and Atmospheric Administration 2009; United Nations Educational Scientific and Cultural Organization
2009).
The density of seawater varies with salinity and temperature (Libes 1992), which leads to stratification
(i.e., arranged in layers) of the water column. In open areas, there are typically three density layers in
the water column: a surface layer (0–600 ft. [0–200 m]), an intermediate layer (600–5,000 ft. [200–1,500
m]), and a deep layer (below 5,000 ft. [1,500 m]) (Castro and Huber 2007).
Nutrients are chemicals or elements necessary to for primary production (i.e., the conversion of
inorganic material into organic material by photosynthetic organisms). Basic nutrients in seawater
include dissolved nitrogen, phosphates, and silicates. Dissolved inorganic nitrogen occurs in ocean
waters as nitrate, nitrite, and ammonia, with nitrates as the dominant form. The nitrate concentration of
the coastal waters within the Study Area is low ranging from approximately 0.54–0.33 micrograms per
liter. Growth and production of primary producers (e.g., phytoplankton) is often limited by the
availability of a nutrient (e.g., nitrate) (Lalli and Parsons 1997). Overall nutrients in the Study Area tend
to increase in concentration with increasing water depth (U.S. Environmental Protection Agency 2010).
Although not a primary nutrient, the availability of iron can affect primary production in the marine
environment. Iron is introduced to the marine environment primarily in the form of sediments carried
by rivers and wind driven transport from continents, as well as from volcanic eruptions (Langmann et al.
2010). Iron is a limiting factor for growth of phytoplankton in high nutrient, low chlorophyll surface
water (Coale et al. 1998; Coale et al. 1996; Martin and Gordon 1988).
3.2.1.3
Bathymetry
This section provides a description of the bathymetry (water depth) of the Study Area. Given that the
bathymetry of an area reflects the topography (surface features) of the seafloor, it is an important factor
for understanding the potential impacts of Navy training and testing activities on the seafloor, the
propagation of underwater sound, and species diversity. The discussion of bathymetry includes a
general overview of the marine geology in the Study Area and a description of the bathymetry of Navy
training and testing areas.
The contour of the ocean floor as it descends from the shoreline has an important influence on the
distribution of organisms, as well as the structure and function of marine ecosystems (Madden et al.
2009). The Study Area is located at the intersection of the Philippine and Pacific crustal plates, atop what
is believed to be the oldest seafloor on the planet dating to the Jurassic era. The collision of the two
plates has resulted in the subduction of the Pacific Plate beneath the Philippine Plate forming the
Mariana Trench. The Mariana Trench is over 1,410 miles (mi.) (2,269 kilometers [km]) long and 71 mi.
(114 km) wide. The deepest point on Earth, known as Challenger Deep, is in the Mariana Trench at a
depth of approximately 35,400 ft. (10,800 m) (Lutz and Falkowski 2012). Challenger Deep is located
3-44
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
338 mi. (544 km) southwest of Guam in the southwestern extremity of the Mariana Trench (U.S.
Department of the Navy 2010).
The seafloor of the Study Area region is characterized by the Mariana Trench, the Mariana Trough,
ridges, numerous seamounts (or guyots), hydrothermal vents, and volcanic activity (Figure 3-26). Two
volcanic arcs, the West Mariana Ridge (a remnant volcanic arc) and the Mariana Ridge (an active
volcanic arc) are separated by the Mariana Trough. The Mariana Trough formed when the oceanic crust
in this region began to spread between the ridges 4 million years ago. The Mariana Trough is spreading
at a rate of less than 0.4 in. (1 cm) per year in the northern region and at rates up to 1.2 in. (3 cm) per
year in the center of the trough. The Mariana archipelago is located on the Mariana Ridge, 99–124 mi.
(159–200 km) west of the Mariana Trench subduction zone. The Mariana archipelago comprises 15
volcanic islands: Guam, Rota, Tinian, Saipan, FDM, Aguijan, Anatahan, Sarigan, Guguan, Alamagan,
Pagan, Agrigan, Asuncion, Maug, and Farallon de Pajaros. Approximately 497 mi. (795 km) separate
Guam from Farallon de Pajaros.
The islands north of FDM are located on an active volcanic ridge and were formed between 1.3 and 10
million years ago. The six southern islands (Guam to FDM) are on the old Mariana fore-arc ridge and
formed about 43 million years ago (Eocene). The young, active volcanic ridge is approximately 16–22 mi.
(26–35 km) west of the southern ridge. The islands on the southern ridge consist of a volcanic core
covered by thick coralline limestone (up to several hundreds of meters). The subsidence of the original
volcanoes in the southern islands allowed for the capping of the volcanoes by limestone deposits.
Limestone covers the northern half of Guam (limestone plateau height: 295–590 ft. (90–180 m) above
mean sea level) while volcanic rock and clay are exposed on the southern half of the island. Tinian
consists of rocky shoreline cliffs and limestone plateaus with no apparent volcanic rock. Similar to
Tinian, the uplifted limestone substrate of FDM is bordered by steep cliffs (The Environmental Company
2004).
In contrast, volcanoes north of FDM have not subsided below sea level, do not have limestone caps, and
remain active. The islands of Anatahan, Guguan, Alamagan, and Pagan are affected by two active
volcanoes, and the islands of Agrigan, Asuncion, and Farallon de Pajaros have documented volcanic
activity spanning from 1883 to 1967. Ruby Volcano and Esmeralda Bank are submarine volcanoes found
west of Saipan and Tinian. Ruby Volcano erupted in 1966 and then again in 1995 as the surrounding area
experienced submarine explosions, fish kills, a sulfurous odor, bubbling water, and volcanic tremors
Smithsonian National Museum of Natural History 1995). Ruby Volcano, also known as Ruby Seamount, is
25 mi. (40 km) northwest of Saipan and is estimated to be approximately 200 ft. (61 m) below sea level
(U.S. Department of the Navy 2010). The summits of the Esmeralda Bank are from 140 to 460 ft. (43 to
140 m) beneath the sea surface as reported in U.S. Department of the Navy 2010.
3-45
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-26: Seafloor Surrounding the Mariana Islands
3-46
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
3.2.1.4
Water Column Essential Fish Habitat
The list of managed species and life-stages for which water column areas are referenced in the EFH or
HAPCs descriptions are compiled in Appendix B, and a summary of water column EFH and HAPCs
designated by the WPRFMC is provided in Table 3-5.
Table 3-5: Water Column Essential Fish Habitat and Habit Areas of Particular Concern References within the
Mariana Islands Training and Testing Study Area
Water
Column
Parameters
Waters
Vertical
layers
EFH or
HAPC
Habitat Area
Occurrence
in Study
Area
Offshore
EFH
Nearshore
EFH
Estuarine
EFH
All EEZ waters
All EEZ waters above the
thermocline
Less than or equal to 100 m
EFH
Less than or equal to 150 m
EFH
Less than or equal to 400 m
EFH
Between 550 and 700 m
EFH
Less than or equal to 600 m
EFH
Less than or equal to1,000 m
EFH/HAPC
Less than or equal to 3,500 m
Notes: (1) The habitats listed may or may not be represented in the available Geographic
Information Systems data. (2) EFH = Essential Fish Habitat, HAPC = Habitat Area of
Particular Concern, EEZ = Exclusive Economic Zone, m = meters
3.2.2
SUBSTRATES
The fundamental descriptor of substrates as either soft or hard is a key factor in structuring biogenic
habitats (Nybakken 1993). The substrate type is also referenced in the EFH or HAPC designations for
species/life stages, which are compiled in Appendix B. A summary of the types of substrates designated
as EFH and HAPC is provided in Table 3-6. Seafloor features (e.g., seamounts, banks, slopes,
escarpments) are included among the types of substrate and are identified on the habitat maps where
geospatial information is available. Bottom substrates in the Study Area are shown in Figures 3-27
through 3-31.
3-47
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 3-6: Substrate Essential Fish Habitat and Habit Areas of Particular Concern References within the Mariana
Islands Training and Testing Study Area
EFH or
HAPC
Habitats
Occurrence
in the Study
Area
Rocky Shelf
Non-Rocky Shelf
Canyon
HAPC
Soft Substrate
EFH
Coral Reef/Hard Substrate
EFH
Patch Reefs
EFH
Surge Zone
EFH
Deep-slope Terraces
EFH
Banks
HAPC
Seamounts
HAPC
Continental Slope/Basin
Notes: (1) The habitats listed may or may not be represented in the
available Geographic Information system data. (2) EFH = Essential Fish
Habitat, HAPC = Habitat Area of Particular Concern
3-48
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-27: Bottom Substrate around Guam
3-49
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-28: Bottom Substrate in Apra Harbor
3-50
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-29: Bottom Substrate around Saipan
3-51
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-30: Bottom Substrate around Tinian
3-52
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-31: Bottom Substrate around Farallon de Medinilla
3-53
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
3.2.2.1
Soft Shores
Soft shores include all wetland habitats having three characteristics: (1) unconsolidated substrates with
less than 75 percent aerial coverage of stones, boulders, or bedrock; (2) less than 30 percent aerial
coverage of vegetation other than pioneering plants; and (3) any of the following water regimes:
irregularly exposed, regularly flooded, irregularly flooded, seasonally flooded, temporarily flooded,
intermittently flooded, saturated, or artificially flooded (Cowardin et al. 1979). Soft shores include
beaches, tidal flats and deltas, and stream beds of the tidal riverine and estuarine systems.
Intermittent or intertidal riverine channels and intertidal estuarine channels are classified as
streambeds. Intertidal flats, also known as tidal flats or mudflats, consist of loose mud, silt, and fine sand
with organic-mineral mixtures that are regularly exposed and flooded by the tides (Karleskint et al.
2006). Muddy, fine sediment is deposited in sheltered inlets and estuaries where wave energy is low
(Holland and Elmore 2008). Mudflats are typically unvegetated, but may be covered with mats of green
algae and benthic diatoms (single-celled algae), or sparsely vegetated with low-growing aquatic plants.
The muddy, intertidal habitat occurs most often as part of a patchwork of intertidal habitats that may
include rocky shores, tidal creeks, sandy beaches, salt marshes, and mangroves.
Beaches form through the interaction of waves and tides, as particles are sorted by size and deposited
along the shoreline (Karleskint et al. 2006). Wide flat beaches with fine-grained sands occur where wave
energy is limited. Narrow, steep beaches of coarser sand form where energy and tidal ranges are high
(Speybroeck et al. 2008). Three zones characterize beach habitats: (1) dry areas above the mean high
water, (2) wrack line (line of organic debris left on the beach by the action of tides) at the mean high
water mark, and (3) a high-energy intertidal zone.
On the island of Guam, the majority of the coastline is comprised of rocky intertidal regions.
Interspersed among this rocky shoreline are 58 beaches composed of calcareous or volcanic sands
(Eldredge 1983). The west coast of Saipan contains well developed fine-sand beaches protected by the
Saigon and Tanapag Lagoons (Scott 1993). All other beaches on Saipan consist of coral-algal-mollusk
rubble. The island of Tinian has 13 beaches (10 located on the west coast and 3 on the east coast). These
beaches are not well developed (except Tinian Harbor on the southwest coast, and Unai Dankulo along
the east coast) and are comprised mainly of medium to coarse grain calcareous sands, gravel, and coral
rubble (Eldredge 1983; Kolinski et al. 2001). On Rota, the rare beaches are found scattered among
limestone patches and are composed of rubble and sand (Eldredge 1983). The coastal area of FDM
contains two small intertidal beaches on the northeastern and western coastlines that are inundated at
high tide.
3.2.2.2
Hard Shores
Hard shores include aquatic environments characterized by bedrock, stones, or boulders that, singly or
in combination, cover 75 percent or more of the substrate and where vegetation covers less than 30
percent (Cowardin et al. 1979). Water regimes are restricted to irregularly exposed, regularly flooded,
irregularly flooded, seasonally flooded, temporarily flooded, and intermittently flooded. Rocky intertidal
shores are areas of bedrock that alternate between periods of submergence at high tide and exposure
to air at low tide. Extensive rocky shorelines can be interspersed with sandy areas, estuaries, or river
mouths.
Environmental gradients between hard shorelines and subtidal habitats are determined by wave action,
depth and frequency of tidal inundation, and stability of the substrate. Where wave energy is extreme,
only rock outcrops may persist. In areas where wave energy is lower, a mixture of rock sizes will form
3-54
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
the intertidal zone. Boulders scattered in the intertidal and subtidal areas provide substrate for attached
macroalgae and sessile invertebrates.
Hard shores are the dominant marine habitat on all islands within the Study Area. This is due to the
volcanic origin of all of the islands (Eldredge 1983). Coastlines within the Study Area are generally lined
with rocky intertidal areas, steep cliffs and headlands, and the occasional sandy beach or mudflat
(Eldredge 1983). Erosion of rocky coastlines by the sea in the Study Area has produced wave-cut cliffs
(created by undercutting the shoreline and causing large sections to fall into the sea). Erosion has also
resulted in sea-level benches comprised of volcanic rock or limestone along the coastline of some
islands (Eldredge 1979, 1983). Large block and boulders often buttress the foot of steep cliffs in the
Study Area.
3.2.2.3
Soft Bottoms
Soft bottoms include all wetland and deepwater habitats with at least 25 percent cover of particles
smaller than stones (10–24 in. [25–60 cm]), and a vegetative coverage less than 30 percent (Cowardin et
al. 1979). Water regimes are restricted to subtidal, permanently flooded, intermittently exposed, and
semi-permanently flooded. Soft bottom forms the substrate of channels, shoals, subtidal flats, and other
features of the bottom. Sandy channels emerge where strong currents connect estuarine and ocean
water columns. Shoals form where sand is deposited along converging, sediment-laden currents forming
capes. Subtidal flats occur between the soft shores and the channels or shoals.
Soft bottom substrates in coastal regions of the Study Area are not common. This is due to the fact that
the intertidal and subtidal regions are often characterized by limestone pavement interspersed with
coral colonies and submerged boulders (Kolinski et al. 2001). Shorelines are often rocky with
interspersed sand beaches or mud flats (Eldredge 1983; Pacific Basin Environmental Consultants 1985).
One type of soft bottom habitat that occurs in the Study Area is lagoons. A lagoon can be described as a
semi-enclosed bay found between the shoreline and the landward edge of a fringing reef or barrier reef
(National Centers for Coastal Ocean Science and National Oceanic and Atmospheric Administration
2005). Lagoons typically contain three distinct zones: freshwater zone, transitional zone, and saltwater
zone (Thurman 1997). Most tropical reef-associated lagoons are not brackish and lack significant
freshwater input. The bottoms of the lagoons are mostly sandy and can be flat, rippled, or filled with
sand mounds created by burrowing organisms. Coral rubble, coral mounds, seagrass, and algae are
found within the lagoons. Coral mounds tend to be more abundant in the outer lagoons and are widely
scattered or absent in the inner lagoons (National Centers for Coastal Ocean Science and National
Oceanic and Atmospheric Administration 2005; Pacific Basin Environmental Consultants 1985).
Lagoons of coastal Guam are associated with Apra Harbor (Inner Harbor, Outer Harbor, and Sasa Bay),
Cocos Lagoon, and numerous embayments along the western coastline. Apra Harbor is the only deep
lagoon on Guam and is the busiest port in the Mariana Islands. The Outer Harbor is enclosed by the
Glass Breakwater. Sasa Bay, located on the edge of the Outer Harbor, is a shallow coastal lagoon
populated with patchy corals (Scott 1993). The Inner Apra Harbor is a human-made lagoon created by
dredging in the 1940s. Cocos Lagoon, a shallow lagoon (40 ft. [12.2 m] deep), is located on the southern
tip of Guam and is encompassed by a series of barrier and fringing reefs (Paulay et al. 2002). The
majority of the substrate in Apra Harbor is sand, as depicted in Figure 3-28; however, there are
intermittent patches of harder substrates (shoals and reefs) within the harbor.
3-55
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
The western coastline of Saipan is lined with sandy beaches protected by a barrier reef which forms
Tanapag and Saipan Lagoons (Scott 1993). Tanapag Lagoon is a typical high-island barrier reef lagoon.
Tanapag Lagoon is located on the northwestern coast of Saipan. Also, on the western coastline of
Saipan, the barrier reefs form two additional lagoons, creating the largest lagoon system in the Mariana
Islands, Garapan Lagoon and Chalan Kanoa Lagoon (Environmental Services Duenas & Associates 1997).
The western side of Tinian has limited lagoon development near the harbor, whereas Rota does not
have any well developed lagoon formations (Pacific Basin Environmental Consultants 1985). Offshore of
FDM, at a depth of approximately 65 ft. (19.8 m), the sandy soft bottom seafloor slopes abruptly
downward toward the abyssal plain (U.S. Department of the Navy 2005). Most of the other islands in the
Marianas also have sandy slopes below the fore reef, typically starting at 100–130 ft. (30.48–39.62 m),
with some variations (U.S. Department of the Navy 2005). See Figures 3-27, 3-28, 3-29, 3-30, and 3-31
for information on the distribution of soft bottom habitats as derived by satellite imagery by National
Oceanic and Atmospheric Administration (NOAA), near Guam, Apra Harbor, Saipan, Tinian, and FDM,
respectively.
In the open ocean portion of the Study Area, soft bottom habitat is located in the Mariana Trough. The
Mariana Trough is comprised of a large relatively flat abyssal plain with water depths ranging from
approximately 11,500–13,100 ft. (3,505.2–3,992.9 m) (Thurman 1997). Very little data regarding the
Mariana Trough within the Study Area has been obtained. However, in general abyssal plains can be
described as large and relatively flat regions covered in a thick layer of fine silty sediments with the
topography interrupted by occasional mounds and seamounts (Kennett 1982; Thurman 1997). The
abyssal plain and similar deepwater areas were originally thought to be devoid of life; however recent
research has shown that these areas are host to thousands of species of invertebrates and fish ("The
Mariana Trench - Biology - Part 1" 2003).
3.2.2.4
Hard Bottoms
Hard, rocky bottom includes all subtidal habitats with substrates having an areal cover of stones,
boulders, or bedrock 75 percent or greater and vegetative cover of less than 30 percent (Cowardin et al.
1979). Generic hard bottom could be any naturally occurring material on the bottom that is sufficiently
solid and stationary (e.g., hard consolidated mud) to support sedentary, attached macroalgae or
invertebrates (e.g., barnacles, anemones, hard corals). As such, hard bottom substrate forms the
foundation of attached macroalgae beds, sedentary invertebrate beds, and reefs.
Subtidal rocky bottom occurs as extensions of intertidal rocky shores and as isolated offshore outcrops.
The shapes and textures of the larger rock assemblages and the fine details of cracks and crevices are
determined by the type of rock, the wave energy, and other local variables (Davis 2009). Maintenance of
rocky reefs requires wave energy sufficient to sweep sediment away (Lalli and Parsons 1993) or OS areas
lacking a significant sediment supply; therefore, rocky reefs are rare on broad coastal plains near
sediment-laden rivers and are more common on high-energy shores and beneath strong bottom
currents, where sediments cannot accumulate. The shapes of the rocks determine, in part, the type of
community that develops on a rocky bottom (Witman and Dayton 2001).
Islands within the Study Area (Guam to FDM) support reefs as do islands north of FDM (Anatahan,
Sarigan, Guguan, Alamagan, Maug, and Farallon de Pajaros). Reefs are also found on OS banks including
Galvez bank located 12 mi. (19.3 km) south of Guam, Santa Rosa Reef located 25 mi. (40.2 km) southsouthwest of Guam, Arakane Bank located 200 mi. (321.9 km) west-northwest of Saipan, Tatsumi Reef
located 1.2 mi. (1.93 km) southeast of Tinian, Pathfinder Bank located 170 mi. (273.6 km) west of
Anahatan, and Supply Reef located 11.5 mi. (18.5 km) northwest of Maug Island (Starmer 2005). The
3-56
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
degree of reef development depends on a number of environmental controls including the age of the
islands; volcanic activity; the availability of favorable substrates and habitats; weathering caused by
groundwater discharge, sedimentation, and runoff accentuated by the overgrazing of feral animals; and
varying levels of exposure to wave action, trade winds, and storms (Eldredge 1983; Paulay 2003; Randall
1985, 1995; Randall et al. 1984; Starmer 2005). See Figures 3-27, 3-28, 3-29, 3-30, and 3-31 for
information on the distribution of hard bottom habitats in the open ocean, near Guam, Apra Harbor,
Saipan, Tinian, and FDM, respectively.
Within the open ocean portion of the Study Area, two types of hard bottom habitat are seamounts and
flat-topped seamounts known as guyots. Generally, seamounts tend to be conical in shape and volcanic
in origin, although some seamounts are formed by vertical tectonic activity along converging plate
margins (Rogers 1994). Both volcanic and tectonic seamounts are present in the open ocean portion of
the Study Area. Seamount and guyot topography is a striking contrast to the surrounding flat,
sediment-covered abyssal plain. Seamounts and guyots can affect local ocean circulation causing
upwelling, which can supply nutrients to surface waters (Rogers 1994; Lalli and Parsons 1997).
Figure 3-26 shows the locations of both seamounts and guyots in the Study Area. Refer to biological
resources chapters of the MITT EIS/OEIS for more information on species inhabiting seamounts.
Deep-sea hydrothermal vents occur in areas of crustal formation near mid-ocean ridge systems
(Humphris 1995). A number of hydrothermal vents have been located in the Study Area, and it is likely
that more exist. Evidence of active hydrothermal venting has been identified in the vicinity of more than
12 submarine volcanoes and at two sites along the back-arc spreading center off to the west of the
Mariana Islands (Embley et al. 2004; Kojima 2002). Hydrothermal vents located in the Mariana Trough
experience high levels of site specific species due to their geographic isolation from other vent systems.
At least 8 of the 30 identified genera known to occur only in the western Pacific hydrothermal vent
systems are found in the Mariana Trough (Hessler and Lonsdale 1991; Paulay 2003). Hydrothermal vents
at Esmeralda Bank, one of the active submarine volcanoes in the Study Area, span an area of
0.08 square mile (0.207 square kilometer) on the seafloor and expel water with temperatures exceeding
172°F (77.8°C) (Stuben et al. 1992). West of Guam and on the Mariana Ridge, there are three known
hydrothermal vent fields: Forecast Vent site (13°24’N, 143°55’E, depth 4,750 ft. [1,447.8 m]), TOTO
Caldera (12°43’N, 143°32’E), and the 13° N Ridge (13°05’N, 143°41’E) (Kojima 2002). Refer to biological
resources chapters of the MITT EIS/OEIS for more information on species inhabiting hydrothermal vents.
3.2.2.5
Artificial Structures
Artificial habitats are human-made structures that provide habitat for marine organisms. Artificial
habitats occur in the marine environment either designed with the intention of being used as habitat
(e.g., artificial reefs), designed with the intention of functioning as something other than habitat (e.g.,
fish aggregating devices, which are floating objects moored at specific locations in the ocean to attract
fishes that live in the open ocean), or are unintentional (e.g., shipwrecks). Artificial structures function
as hard bottom by providing structural attachment points for algae and sessile invertebrates, which in
turn support a community of animals that feed, seek shelter, and reproduce there (National Oceanic and
Atmospheric Administration 2007).
Artificial habitats in the Study Area include artificial reefs, shipwrecks, and fish aggregating devices.
Artificial reefs are designed and deployed to supplement the ecological services provided by coral or
rocky reefs. Artificial reefs range from simple concrete blocks to highly engineered structures. Vessels
that sink to the seafloor, including shipwrecks within the Study Area, are colonized by the common
3-57
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
encrusting and attached marine organisms that attach to hard bases. Over time, the wrecks become
functioning ecosystems.
Many shipwrecks are found within the Study Area, including grounded vessels and military wreckage.
Vessels have probably wrecked upon the shores of the Mariana Islands since Spanish galleons sailed to
these islands during the seventeenth century. There are abundant WWII-era remains (including sunken
ships, airplanes, and tanks) along the shores of the Mariana Islands that resulted from the battles of
Guam, Saipan, and Tinian (Commonwealth of the Northern Mariana Islands 2001). Surrounding Guam
there are 63 documented shipwrecks dating between 1520 and 1941 (Carrell et al. 1991). However, only
the locations of about 60 known wrecks, obstructions, or occurrences (e.g., shipwrecks, aircraft, and
military equipment) have been determined (Figure 3-32), including one World War II-era amphibious
tractor in Agat Bay and 31 submerged wrecks, obstructions, or occurrences in the Guam Commercial
Harbor (work and fishing boats; barges; tugs; landing craft utility vessels; a British passenger ship (“C S
Scotia”); WWII Japanese freighters or transport ships (“Tokai Maru,” “Kitsugawa Maru,” and “Nichiyu
Maru”); and three Japanese planes from World War II commonly referred to as Val, Jake, and Hufe
(Carrell et al. 1991; Lotz 1998).
Most artificial reefs intended as habitat in marine waters have been placed and monitored by individual
state programs; national and state databases indicating the locations of artificial reefs are not available
(National Oceanic and Atmospheric Administration 2007). In the Study Area, there are dedicated
artificial reefs found in two locations: Agat Bay, Guam and Apra Harbor, Guam. In 1969, 357 tires were
tied together and scattered over a 5,000-square-foot (ft.2) (4,645-square-meter [m2]) area in Cocos
Lagoon (Eldredge 1979). In the early 1970s, a second reef consisting of 2,500 tires was also placed in
Cocos Lagoon (Eldredge 1979). These tire reefs have disintegrated and no longer serve as artificial reefs.
In 1977, a 52.5 ft. (16.0 m) barge was modified to enhance fish habitat and was sunk in 60 ft. (18.3 m) of
water in Agat Bay. In Apra Harbor, the “American Tanker” was sunk in 1944 at the entrance of the
harbor to act as a breakwater. In 1944, the 76th Naval Construction Battalion (SEABEES) built the Glass
Breakwater which forms the north and northwest sides of Apra Harbor (Thompson 2002). The enormous
seawall is made of 1,200 acre-feet (148,000 cubic meters [m3]) of soil and coral extracted from Cabras
Island (Thompson 2002). The Glass Breakwater is the largest artificial substrate in the Marianas.
Currently, Guam and the Northern Mariana Islands maintain several fish aggregating devices within
20 nm of the shoreline (Chapman 2004; Guam Department of Agriculture Division of Aquatic and
Wildlife 2004). Figures 3-33 and 3-34 show the locations of the fish aggregating devices surrounding
Guam, Tinian, and Saipan. Lost fish aggregating devices are replaced normally within 2 weeks (Chapman
2004). Fish aggregating device sites may change frequently; the U.S. Coast Guard is responsible for
keeping track of these changes. Fish aggregating device buoys, with long chains, may be considered a
safety hazard if the buoys become disconnected.
3-58
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-32: Known Shipwrecks and Other Obstructions within 12 Nautical Miles of Guam, Rota, Tinian, and
Saipan
3-59
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-33: Fish Aggregating Devices Surrounding Guam
3-60
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-34: Fish Aggregating Devices around Tinian and Saipan
3-61
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
3.2.3
BIOGENIC HABITATS
Living structures on the substrate are termed biogenic habitats, and include wetland shores, attached
macroalgae beds, submerged rooted vegetation beds, and coral reefs (refer to relevant sections for
detailed description). The differences between biogenic habitats reflect a basic continuum of resilience
and recovery from disturbance; attached macroalgae recover quickly from the least disturbance (Mach
et al. 2007), whereas reef structures take a very long time to recover from a relatively high level of
disturbance (Fox and Caldwell 2006). The biogenic habitats also correspond to the EFH or HAPC
descriptors for species/life stages (see Table 3-1). The biogenic habitats are classified by water (e.g.,
open ocean, continental shelf, nearshore) to refine their location within a fishery management council
region (Table 3-7).
Table 3-7: Biogenic Habitats in Fishery Management Council Area and Their Essential Fish Habitat Synonyms
Descriptor
Habitats
Vegetated Shores
Submerged Rooted
Vegetation Beds
Attached Macroalgae Beds
Reefs
Mangrove
Seagrass beds
Coral reefs
Notes: The habitats listed may or may not be represented in
the available Geographic Information System data.
3.2.3.1
Vegetated Shores
Vegetated shorelines are characterized by erect, rooted, herbaceous hydrophytes, excluding mosses and
lichens that grow above the water line (Cowardin et al. 1979). This vegetation is present for most of the
growing season in most years. These wetlands are usually dominated by perennial plants. All water
regimes are included except subtidal and irregularly exposed. Vegetated shorelines in the Study Area are
formed by mangrove plant species.
Mangroves are a group of woody plants that have adapted to brackish water environments in the
tropics and subtropics (Ruwa 1996). Mangroves provide critical ecosystem services in their role as
primary producers, including contributions to the decomposition of matter (Bouillon 2009), sediment
stabilization (Ruwa 1996), nursery habitat (Mitsch et al. 2009), and providers of habitat for commercially
important species (e.g., fish, shrimp, and crabs) (Aburto-Oropeza et al. 2008; Hogarth 1999). Nearshore
fisheries associated with mangroves are generally more productive than those not associated with
mangroves due to the nutrient storage in the plants and the physical complexity of the habitat that
mangroves provide for fish and their prey (Ruwa 1996).
Mangroves provide important nursery habitat for many species of fish and invertebrates. Conservation
of mangrove habitats is important due to the use of these areas as nurseries for commercial fish species
and coral reef fish species (Laegdsgaard and Johnson 1995). Additionally, researchers have found that
coral reef fish were twice as abundant on reefs adjacent to mangrove forests compared to reefs without
mangroves (Roach 2004).
Mangrove forests are native to the Study Area; however, they are only present on the islands of Guam
and Saipan (Figure 3-35). The mangroves of Guam are the most extensive and diverse, totaling
approximately 170 acres (ac.) (68 hectares [ha]) (Scott 1993); however, a recent Landsat survey
3-62
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
documented only 84.5 ac. (34.2 ha) (Bhattarai and Giri 2011). Guam has 10 species of mangroves,
including Rhizophora mucronata, Rhizophora apiculata, Avicennia marina, Bruguiera gymnorhiz,
Lumnitzera littorea, Nypa fructicans, Xylocarpus moluccensis, Heritiera littoralis, Heritiera tiliaceus, and
Acrostichum aureum (Guam Department of Agriculture 2005). The mangrove forests on Saipan are
dominated by a single species, Bruguiera gymnorhiza.
Figure 3-35: Distribution of Seagrass and Mangrove Communities in the Mariana Islands Training and Testing
Study Area: (a) Guam, (b) Apra Harbor, and (c) Tinian and Saipan
3.2.3.2
Submerged Rooted Vegetation Beds
Submerged rooted vegetation form “meadows” or “beds” where they dominate the intertidal or shallow
subtidal zone of estuarine or nearshore waters (Fonseca et al. 1998). The plants grow in soft bottom
substrate receiving 15–22 percent or more of surface light intensity (Fonseca et al. 1998; Kemp et al.
2004) depending on “bio-optical” properties of the water (Biber et al. 2007).
Seagrasses are unique among flowering plants in their ability to grow submerged in shallow marine
environments. Except for some species that inhabit the rocky intertidal zone, seagrasses grow in
shallow, subtidal, or intertidal sediments, and can extend over a large area to form seagrass beds
(Garrison 2004; Phillips and Meñez 1988). They provide suitable nursery habitat for commercially
important organisms (e.g., crustaceans, fish, and shellfish) and also are a food source for some
protected species (e.g., sea turtles) (Heck et al. 2003). The structure of seagrass beds can prevent
coastal erosion, promotes nutrient cycling through the breakdown of detritus (Dawes 1998; South
Atlantic Fishery Management Council 1998), and improves water quality. Seagrasses also contribute a
3-63
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
high level of primary production to the marine environment, which supports high species diversity and
biomass (Spalding et al. 2003).
Seagrass beds are distributed within the Study Area (Figures 3-36 to 3-40). Seagrasses that occur in the
coastal areas of the Study Area from the southern Mariana Islands include Enhalus acoroides, Halodule
uninervis, and Halophila minor (Tsuda et al. 1977). Both Guam and Saipan have extensive seagrass
meadows surrounding the coastlines (National Centers for Coastal Ocean Science and National Oceanic
and Atmospheric Administration 2005), including extensive beds in Agat Bay (including the Agat Unit of
the War in the Pacific National Historical Park) (Daniel and Minton 2004), south of Apra Harbor, Agana
Bay, and Cocos Lagoon on Guam (Daniel and Minton 2004; Eldredge et al. 1977). According to NOAA
satellite surveys, there are no seagrass beds in Apra Harbor (Figure 3-37); however, smaller beds of
seagrasses may be present in this area. The NOAA satellite surveys do not show seagrass beds around
Tinian (Figure 3-38). However, a literature review provided information that Tinian possesses seagrass
beds along the northeastern, eastern, the southwestern, and northwestern coastlines (Kolinski et al.
2001, U.S. Department of the Navy 2003), and that seagrasses were largely absent from Tinian’s north
and south coasts (Kolinski et al. 2001). Seagrasses are more scattered on the island of Saipan (Figure 339), with seagrass beds reported along Tanapag Beach (along the northwest coast) and in Puerto Rico
Mudflats (northwest shoreline, north of Tanapag Beach) (Scott 1993, Tsuda et al. 1977). There is no
record of seagrasses for the islands north of Saipan (Tsuda 2009), which is also documented in the NOAA
satellite surveys for FDM (Figure 3-40).
3-64
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-36: Marine Vegetation Surrounding Guam
3-65
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-37: Marine Vegetation in Apra Harbor
3-66
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-38: Marine Vegetation Surrounding Tinian
3-67
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-39: Marine Vegetation Surrounding Saipan
3-68
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-40: Marine Vegetation Surrounding Farallon de Medinilla
3-69
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
3.2.3.3
Attached Macroalgae Beds
Attached, non-vascular plants (i.e., macroalgae) form “meadows” or “beds” where they dominate
intertidal shores or subtidal bottoms. Green, red, and brown algae represent basic taxonomic groups of
macroalgae species, with some species (e.g., kelp, seaweed) growing attached to substrate. As a general
rule, algae can grow down to bottom areas receiving 1 percent or more of surface light intensity (Wetzel
2001).
In the Study Area there are 31 species of brown algae (Phaeophyta) (Lobban and Tsuda 2003). Most
species are attached to the seafloor in coastal waters, and include species such as Sargassum ilicifolium,
Sargassum obtusifolium, and Sargassum polycystum (Lobban and Tsuda 2003). Green algae
(Chlorophyta) are found in areas with a wide range of salinity, such as bays and estuaries, and are eaten
by various organisms, including zooplankton (small animals that float in the water), snails, and
herbivorous fish. In the Study Area, the green algae, Caulerpa racemosa and Caulerpa lentillifera, are
harvested for human consumption.
Red alga (Rhodophyta) occurs in coastal waters of the Study Area, primarily in reef environments and
intertidal zones. Some species of red algae that occur in the Study Area include Erythrotrichia carnea
and Yamadaella caenomyce (Lobban and Tsuda 2003). In the Study Area, the species Gracilaria tsudae
had previously been harvested for human consumption until being implicated in the deaths of three
individuals in 1991 (Tsuda 2009). Many Rhodophyta species support coral reefs by trapping loose
sediments, and cementing coral fragments to provide the base structures for coral growth and a living
protective cover (Castro and Huber 2000). Coralline algae secrete calcium carbonate to build a hard shell
around its cell walls. There are both encrusting forms, which grow as a crust over hard structures such as
rocks and the shells of organisms like clams and snails, and upright forms of coralline algae (Kennedy
2012). Some species of red crustose coralline algae in the Study Area include Hydrolithon onkodes,
Lithophyllum pygmaeum, and Pneophyllum conicum (Minton et al. 2009). The percentage cover of red
coralline algae is estimated from surveys to be less than 20 percent for Guam and Tinian and increases
to approximately 31–50 percent on portions of the southwestern side of Saipan (Minton et al. 2009).
3.2.3.4
Coral Reefs and Communities
The Mariana nearshore environment is characterized by extensive coral bottom and coral reef areas.
There are fewer reef-building hard coral species and genera in the northern compared to the southern
Mariana Islands: 159 species and 43 genera of hard coral species in the northern islands versus 256
species and 56 genera in the southern islands (Randall 2003; Abraham et al. 2004). In general, the coral
reefs of the Marianas have a lower coral diversity compared to other reefs in the northwestern Pacific
(e.g., Palau, Philippines, Australian Great Barrier Reef, southern Japan, and Marshall Islands) but a higher
diversity than the reefs of Hawaii. Corals reported in Guam are typically found on shallow reefs and
upper forereefs (or outer portion of the reef, closest to open ocean) at depths less than 245 ft. (74.7 m),
and deeper forereef habitats within the photic zone that allows for coral growth (> 245 ft. [> 74.7 m]
water depth) (Randall 2003).
Coral Communities and Reefs of Guam
On the island of Guam, most northern shorelines are karstic (layer or layers of soluble bedrock, usually
carbonate rock such as limestone or dolomite) and bordered by limestone cliffs. In a few areas, the
shorelines consist of volcanic substrates. On windward shores, reefs are narrow and have steep
forereefs. Narrow reef flats or shallow fringing reefs (approximately 325–3,250 ft. [99.06–990.6 m]
wide) are characteristic of leeward and more protected coastlines. Reefs also occur in lagoonal habitats
3-70
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
in Apra Harbor and Cocos Lagoon. Reef organisms also occur on eroded limestone substrates including
submerged caves and crevices, and large limestone blocks fallen from shoreline cliffs (Paulay 2003).
Reefs in the southern half of Guam have always been subject to more naturally-occurring sedimentation
than in the northern half of the island because of the difference of erosional products (volcanic in the
south versus limestone in the north) (Richmond and Davis 2002). Coral cover and diversity are currently
higher on reefs located along the northeastern coast of Guam (Richmond and Davis 2002). Historical
surveys suggest that diversity was actually higher in the south before anthropogenic impacts severely
impacted those reefs. The National Centers for Coastal Ocean Science (NCCOS)/NOAA (2005) survey of
shallow water benthic habitats of Guam determined that the overall coral cover around Guam ranged
from 10 to 90 percent. Most of the reefs surrounding Guam have a coral cover ranging from 10 to 50
percent. NCCOS/NOAA (2005) delineates four of the areas of Guam where coral cover ranges from 50 to
90 percent: an area off Mergagan Point on the northeastern end of the island, an area off Pagat Point on
the eastern side of the island, an area immediately south of Togacha Bay also on the eastern side of the
island, and Apra Harbor.
The reefs near populated areas of Guam, Saipan, Tinian, and Rota receive most of the human impacts
from coastal development, population growth, fishing, and tourism. These threats can result in coral
death from coastal runoff (Downs et al. 2009), reduced growth rates caused by a decrease in the pH of
the ocean from pollution (Cohen et al. 2009), reduced tolerance to global climate change (Carilli et al.
2010), and increased susceptibility to bleaching (which are often tied to atypically high sea temperatures
[Brown 1997; Glynn 1993; van Oppen and Lough 2009]). Human-made noise may impact coral larvae by
masking the natural sounds that serve as cues to orient them towards suitable settlement sites (Vermeij
et al. 2010).
Exposure to runoff from land from development projects can also affect local reef communities. Erosion
rates in the Ugum Watershed on Guam doubled from 1975 to 1993 as a result of road construction and
development projects. The discharge of cleaning chemicals has also occurred, with subsequent impacts
on local coral populations (Wilkinson 2002). Exposure to oil runoff from land, and natural seepage is
another threat to marine invertebrates. Additional information on the biology, life history, and
conservation of marine invertebrates (ESA-listed species, species of concern, and candidate species) can
be found on the website maintained by the NMFS.
Apart from a few exceptions, coral reefs in the Pacific Ocean are confined to the warm tropical and
subtropical waters between 30° N and 30° S. Over 400 scleractinian (stony corals) and hydrozoan coral
species (hydrocorals), representing 22 families and 108 genera, have been identified from Guam and the
Mariana Islands (Randall 2003). Of this total number, 377 are scleractinian species that occur within 20
families and 99 genera, and 26 are hydrozoan species that occur within 2 families and 9 genera. About
70 percent of the coral fauna (281 species) contain zooxanthellae in their tissues and about 30 percent
(122 species) are azooxanthellate, although several genera contain both azooxanthellate and
zooxanthellae species (Randall 2003). Azooxanthellate corals obtain energy from detritus, zooplankton,
and nekton they capture from the surrounding water. Since azooxanthellate corals do not depend on
sunlight or a symbiotic existence with zooxanthellae, they can be found in deeper waters (National
Marine Fisheries Service 2010).
Deep-sea coral communities are prevalent throughout the Mariana Islands chain, and often form
offshore reefs. Much like shallow-water corals, deep-sea corals are fragile, slow growing, and can
survive for hundreds of years. In the Mariana Islands, gorgonians, while occurring at all depths, are the
3-71
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
most commonly found corals in deep-sea communities. Gorgonian diversity and abundance increase
below 30 m (98.4 ft.), especially in steep, cavernous, and current-swept areas, so that about 20 species
are known between 30 and 60 m (98.4 and 196.9 ft.) (Pauley et al. 2003). Several of the gorgonian
species listed have been encountered at diving depths only in caverns along the southern Orote
Peninsula of Guam, especially the Blue Hole; these species are otherwise restricted to deeper water. In
contrast, the much richer deep-water fauna remains poorly known. Gorgonians, the soft coral genera
Siphonogorgia and Dendronephthya, and black corals become much more diverse and abundant below
60 m (196.9 ft.). Dredging and tangle net surveys (Eldredge 2003) have already revealed about 70
species of arborescent octocorals at 60–400 m (196.9–1,312.3 ft.) and many others surely remain to be
collected.
There is evidence that overall coral reef habitat has declined in the Study Area, and this is used as a
proxy for population decline in many species. Natural and human-induced disturbances affecting the
reefs of Guam have caused a significant decline of coral cover and recruitment since the 1960s
(Richmond 1994). Coral cover on many fore reef slopes on Guam has decreased from over 50 percent to
less than 25 percent (Birkeland 1997). There are several reefs of Guam where coral cover remains high,
including Apra Harbor, Agat Bay, Orote Ecological Reserve Area, and Haputo Ecological Reserve Area.
Species that are particularly susceptible to bleaching, disease, and other threats are more susceptible to
further decline; therefore, population decline is based on both the percentage of destroyed reefs and
the percentage of critical reefs that are likely to be destroyed within 20 years (Wilkinson 2004).
Because of its depth (51 m), the Apra Harbor lagoon is unique to the MIRC Study Area (Paulay et al.
1997). It provides habitat for unique and diverse benthic fauna: for example, most of the sponges and
ascidians (sea squirt) found in Apra Harbor—48 species of sponges and 52 species of ascidians—are
unique to Apra Harbor. Some of the species (1 sponge and 16 ascidians) were introduced via ship traffic.
Indigenous species generally occupy natural substrates while introduced and cryptogenic species
(species whose origins cannot be verified) generally occupy artificial substrata (e.g., wharf walls,
concrete revetments, moorings, and navigational buoys) (Paulay et al. 1997).
Corals are found in the Outer Apra Harbor where they thrive on shoals and fringing reefs. Detailed
surveys and benthic habitat maps for specific locations within Apra Harbor were produced for an
environmental assessment (Department of the Navy 2007), and are depicted in Figures 3-41 to 3-44.
Porites rus is the dominant coral species on the shoals in the center of the harbor outside Sasa Bay
(Western Shoals, Jade Shoals, and Middle Shoals) (Figures 3-41 and 3-42). Other coral species associated
with these shoals include Porites lobata, P. annae, P. cylindrica, Millepora dichotoma, Acropora formosa,
and P. damicornis (Paulay et al. 1997). Coral cover on the shoals range from 50 to 90 percent (Paulay
2003).
Along the southern boundary of Apra Harbor between Orote Point and Gabgab Beach including east and
west of ammunition pier or “Kilo Wharf,” coral cover on fringing reefs is high (Figure 3-43). These areas
support high coral cover (close to 100 percent cover) consisting mainly of P. rus (> 90 percent of the
cover) and other stony corals including P. lichen, P. lobata, Platygyra pini, Leptoseris spp., Lobophyllia
corymbosa, and Acanthastrea echinata (Smith 2004). Reefs located further in the harbor (excluding the
Inner Apra Harbor) have been severely impacted by freshwater runoff, sedimentation, and polluted
discharges (Richmond and Davis 2002). Corals in the Inner Apra Harbor (including P. rus and P.
damicornis) encrust sheet pilings, rocks, and concrete debris (Smith 2007).
3-72
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
In Inner Apra Harbor, corals occur in Abo Cove and the inner portion of the entrance channel to the
Inner Apra Harbor (Department of the Navy 2005, U.S. Naval Base Guam 2013). In the entrance channel
to the Inner Apra Harbor, corals consist of P. rus and P. cylindrica (Department of the Navy 2005). Corals
are also found on sheet piles in the entrance channel of the Inner Apra Harbor and the outer reaches of
the Inner Apra Harbor (Smith 2007).
Figure 3-41: Benthic Habitats of the Sasa Bay
3-73
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-42: Benthic Habitats of San Luis Beach
3-74
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-43: Benthic Habitats of Kilo Wharf
3-75
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-44: Benthic Habitats of Glass Breakwater
Coral Communities and Reefs of Tinian
Barrier reefs, fringing reefs, and a broad shelf area (1,000 m wide) are found off the Tinian Harbor. The
largest amount of coral cover is probably found along the outer edges of the reef (fore reef and terrace)
(Starmer et al. 2002). Fringing and fore reefs (less than 200 m wide) occur immediately next to the
western shoreline of Tinian. Corals are found on the fore reef and insular shelf seaward of the fore reef.
From Unai Masalok to Puntan Masalok, no fringing reefs are found. Furthermore, there are no fringing
reefs from Puntan Masalok to Puntan Carolinas (southernmost point of Tinian). Fringing reefs reoccur
past Puntan Carolinas. NCCOS/NOAA (2005) determined that the overall coral cover around Tinian
ranged from 10 to 50 percent (Figure 3-45).
3-76
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-45: Coral Coverage Surrounding Tinian
3-77
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Coral cover ranges from 14 to 59 percent on coral reefs at Kammer Beach and Two Coral Head,
respectively (Quinn and Kojis 2003). Dominant coral species in terms of cover are Goniastrea retiformis
at Kammer Beach, and P. rus at Two Coral Head. Coral cover is much higher at Two Coral Head
compared to Kammer Beach due to fewer coral predator-resistant species (Quinn and Kojis 2003).
Unai Chulu, Unai Babui, and Unai Dankulo are three beach areas and nearshore reefs within the MIRC
Study Area that have been evaluated for amphibious landing activities (Marine Research Consultants
1999). Unai Chulu and Unai Babui are located on the northwestern side of Tinian and Unai Dankulo on
the east side of the island, north of Puntan Masalok. At Unai Chulu, within 20 m seaward of the
shoreline, the reef flat substrate includes sand, rubble, and outcrops of a fossil reef. Live cover in the
inner reef flat is mostly composed of turf algae. The few coral species of the genus Porites located in this
area of the reef form circular, flattopped, and lobate colonies. In the middle of the reef flat, echinoids
have bioeroded the reef substrate, and corals (small branching and encrusting colonies) are more
abundant when compared to the inner reef flat. The fringing reef is exposed to wave action, resulting in
few coral colonies. Seaward of the fringing reef, the reef front forms a spur-and-groove system
(alternating channels and ridges that are perpendicular to the fringing reef). Spurs are 1–2 m wide and
the grooves are approximately 5 m wide. Abundant coral cover was observed within the spurs. Seaward
of the spur-and-groove system is a deep reef front terrace. The reef morphology off Unai Babui is similar
to that of Unai Chulu except that the spur-and-groove system was more developed at Unai Babui.
Surveys from 2008 found that overall taxa at Unai Babui was 4.3 times higher on the reef slope than the
reef flat, with corals accounting for most of the observed difference (Minton et al. 2009). The surveys
also found that the taxa richness at Unai Chulu and Unai Dangkolo were higher on the reef slope than
the reef flat. Unai Dangkolo reef flat had the highest coral density of all of the reefs surveyed in the
study (Minton et al. 2009).
A fringing reef borders the Unai Dankulo white carbonate beach (National Centers for Coastal Ocean
Science and National Oceanic and Atmospheric Administration 2005). Macroalgae (10–50 percent cover)
populate the reef flat. Corals (10–50 percent cover) are a main constituent of the fore reef and insular
shelf (National Centers for Coastal Ocean Science and National Oceanic and Atmospheric Administration
2005). Surveys conducted in 1994, however, report that the inner reef flat supports an extensive (50–70
percent coral cover) and diverse reef community (25 coral species). On the reef front, there is a spurand-groove system down to a depth of 10 m, seaward of which the benthos is comprised of carbonate
pavement. Both the spur-and-groove system and the fore reef pavement are densely populated by
corals (36 species of corals). The recent benthic habitat mapping of the CNMI by NCCOS/NOAA (2005)
reflects the change in reef flat composition. Since NCCOS/NOAA (2005) show relatively abundant coral
cover on the reef front, the fore reef has possibly retained some of its pre-typhoon, pre-December 1997
characteristics. The impacts of corallivorous predators have most likely altered the coral composition
and cover on the fore reef (Quinn and Kojis 2003).
Coral Communities and Reefs of Farallon de Medinilla
In contrast with the other southern Mariana Islands, FDM does not include fringing or fore reefs. Rather,
it has a relatively wide insular shelf (400–1,800 m wide) that supports limited coral cover along all sides
except the western side of the island (National Centers for Coastal Ocean Science and National Oceanic
and Atmospheric Administration 2005) (Figure 3-46). In 2004, 81 species of corals were observed on
reefs at FDM (Department of the Navy 2005). Overall, the northwestern nearshore area (eroded
submerged cliff face and reef terrace) of the island supports the highest diversity of marine
invertebrates and fishes on FDM (Department of the Navy 2005). Most of the coastline of FDM is
bordered by steep karstic cliffs which for the most part extend 6–9 m below the waterline (Department
3-78
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
of the Navy 2005). Cliffs on the western shoreline extend more than 20 m below the waterline. There
are numerous underwater caves along the FDM shoreline. Boulders dislodged from the cliffs border the
base of the cliffs. Seaward of the cliff face is a reef terrace that is 30–50 m wide and 10–25 m deep
beyond which is a sandy slope zone. On parts of the western side of the island, a vertical wall undercut
by caves and ledges delimits the seaward edge of the reef terrace and intersects with the sandy slope
habitat. At the southern end of the island, a 2 m deep “finger reef” extends 200 m southward. The edges
of the finger reef are vertical walls that drop down to a 30 m depth. The reef terrace consists of a
spur-and-groove system on the eastern (windward) side of the island where the island forms an isthmus
separating the lower narrow third of the island from the wider upper two-thirds of the island
(Department of the Navy 2005).
Near the cliff edge on the reef terrace of the eastern side of FDM, there is estimated to be less than
5 percent coral cover (Department of the Navy 2005). Further offshore, there is estimated to be 10–20
percent coral cover composed of encrusting Porites and head coral forming Pocillopora. Coral cover on
the boulders is estimated to be 25–30 percent and comprised of Pocillopora, Porites, Montipora, and
Millepora. Coral cover on the ridges of the spur-and-groove system off the island isthmus on the
windward side ranges is estimated to be from 15 to 25 percent, and is composed of Porites and
Pocillopora. There are large aggregations of the long-spined urchin Echinotrix diadema (hundreds to
thousands of individuals) seen both on the eastern and western sides of the island, and high coral cover
is found on boulders along the reef terrace on the leeward side of the island (50–70 percent, mostly
Pocillopora coral heads). Most of the branching colonies of Pocillopora sp. on the leeward side have
broken branches (Department of the Navy 2005).
Since 1971, FDM has been a target site for live-fire military exercises (ship-to-shore gunfire, aerial
gunnery and bombing) (Smith et al. 2013). The majority of the ordnance found underwater at FDM
during reef assessments conducted since 1999 occur at the northwestern end of the island (Smith et al.
2013). Some ordnance is imbedded in the seafloor suggesting a miss and other appears to have rolled
off the island; however, in both cases, no damage to the health of the coral reef ecosystem has been
observed (Smith et al. 2013).
Assessments of the near shore marine and fisheries resources at FDM have been conducted between
1999 and 2012. The surveys conducted through 2004 were performed by a Navy contract biologist, with
assistance from a NOAA, U.S. Fish and Wildlife Service, and CNMI representative. Support was also
provided by Navy Explosive Ordnance Disposal (EOD) personnel. All surveys since 2004 have been
performed by Navy marine ecologists, with support from Navy EOD personnel. Based upon the
observations from the surveys, fish stocks around FDM are robust and healthy (Smith et al. 2013). In
fact, based upon subjective estimates of size, total numbers and health, the fish stocks around FDM are
probably among the best in the entire archipelago. Sea turtle sightings have remained relatively
constant between 1999 and 2012; both green sea turtles and hawksbill turtles have been regularly
sighted. During the 2004 survey, it was noted that many of the corals with branching or plating type
growth forms sustained significant breakage. Some members of the 2004 survey team suggested this
could be the result of bombing/training activities. However, based upon observations at other locations,
bombing levels in previous and subsequent years, plus observations made during the 2005, 2006, and
2007 surveys, it is clear that the damage observed in 2004 was a result of a direct hit on FDM by
Typhoon Ting Ting shortly before the 2004 survey was conducted (Department of the Navy 2004). This
site showed complete recovery by the 2010 survey (Smith et al. 2013). In conclusion, the near shore
marine natural resources at FDM are thriving; the island in fact, is serving as a de-facto preserve due to
the restricted fishing access (see Riegl et al. 2008 for comparable results at Vieques, Puerto Rico).
3-79
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 3-46: Coral Communities Surrounding Farallon de Medinilla
3-80
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
4 ASSESSMENT OF IMPACTS
The overall approach to analysis in this EFHA included the following general steps:
1.
2.
3.
4.
Identification of habitats designated as EFH and HAPCs for analysis
Analysis of habitat-specific impacts for individual stressors
Analysis of habitat-specific impacts for combined stressors
Consideration of mitigations to reduce any potential impacts
Navy training and testing activities in the Proposed Action may produce one or more stimuli that cause
stress on a habitat designated as EFH. Each proposed Navy activity was examined to determine its
potential stressors (Table 4-1). Not all stressors affect every habitat, nor do all proposed Navy activities
produce stressors (Table 4-2). The potential direct, indirect, and cumulative impacts of the Proposed
Action were analyzed based on the presence of these potential stressors within the designated habitat.
First, a preliminary analysis was conducted to determine the habitats designated as EFH that could be
potentially impacted and their associated stressors. The term stressor is broadly used in this document
to refer to an agent, condition, or other stimulus that causes stress to an organism or alters physical,
socioeconomic, or cultural resources. Secondly, each resource was analyzed for potential impacts from
individual stressors, followed by an analysis of the combined impacts of all stressors related to the
Proposed Action. Mitigation measures are discussed in detail in Chapter 5.
In this phased approach, the initial analyses were used to develop each subsequent step so the analysis
focused on relevant issues that warranted the most attention. The systematic nature of this approach
allowed the Proposed Action with the associated stressors and potential impacts to be effectively
tracked throughout the process. This approach provides a comprehensive analysis of applicable
stressors and potential impacts. Each step is described in more detail below.
4.1
POTENTIAL IMPACTS TO ESSENTIAL FISH HABITAT
This section evaluates how and to what degree the activities described in Section 2.4 (Description of the
Action Area) could impact EFH and HAPC in the Fishery Management Council region of the Study Area. A
stressor is analyzed for a designated habitat if it has the potential to alter the quality or quantity of that
habitat (e.g., water column, benthic habitat). The stressors that could potentially impact one or more
EFH and HAPCs in the Study Area are shown in Table 4-1.
4-1
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 4-1: List of Stressors Analyzed
Components and Stressor Categories for Essential Fish Habitat
Acoustic Stressors
Sonar and other active acoustic sources
Vessel noise
Explosives
Swimmer defense airguns
Weapons firing, launch, and impact noise
Energy Stressors
Electromagnetic devices
Physical Disturbance and Strike Stressors
Vessels
In-water devices
Military expended materials
Seafloor devices
Contaminant Stressors
Explosives, explosive byproducts, unexploded ordnance
Metals
Chemicals other than explosives
Other Materials
The stressors vary in intensity, frequency, duration, and location within the Study Area. The data
available for these parameters are limited to what is presented in Section 2.4 (Description of the Action
Area). The specific analysis of the training and testing activities considers the stressor “footprints” and
their coincidence with designated EFH and HAPCs within the Fishery Management Council boundaries.
The duration of impacts is based on either the duration of stressor or recovery of the habitat:
•
•
•
•
Temporary – stressor duration or recovery in hours, days, or weeks
Short Term – stressor duration or recovery in less than 3 years
Long Term – stressor duration or recovery in more than 3 years but less than 20 years
Permanent – stressor duration or recovery in more than 20 years
Minimal effects could be those that are limited in duration and that allow the affected area to recover
before measurable long-term or permanent impacts to EFH occur, or those that may result in relatively
small and insignificant long-term or permanent impacts to EFH and its ecological functions.
The conclusions for spatial and temporal impacts on EFH and HAPCs are encapsulated in text boxes at
the end of the training and testing activities sections under each substressor. The managed species life
stages that could be impacted are listed by habitat descriptors in Appendix B. The analysis will be
separated by: (1) potential impacts on the biological components of the water column, (2) potential
impacts on benthic substrate, and (3) potential impacts on biogenic habitats. Because HAPCs are subsets
of the specific management unit EFH (see Table 3-1), impacts to the water column, benthic substrate,
and biogenic habitats, also cover impacts to HAPC. If an impact to a specific HAPC is anticipated that is
not covered in the analysis, then a separate analysis and conclusion will be conducted. Physical impacts
to the water column habitat are analyzed in the contaminant stressor analysis.
4-2
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 4-2: Stressors by Warfare and Testing Area
Warfare Area/Testing Area
Acoustic
Stressors
Energy
Stressors
Physical
Disturbance and
Strike Stressors
Contaminant
Stressors
Training Activities
Anti-Air Warfare
Amphibious Warfare
Strike Warfare
Anti-Surface Warfare
Anti-Submarine Warfare
Electronic Warfare
Mine Warfare
Naval Special Warfare
Major Training Events
Other Training Activities
Anti-Surface Warfare
Anti-Submarine Warfare
Shipboard Protection Systems and Swimmer
Defense Testing
New Ship Construction
Life Cycle Activities
Office of Naval Research Testing
Testing Activities
4.1.1
ACOUSTIC STRESSORS
This section analyzes the potential impacts of acoustic stressors on EFH and HAPCs resulting from
training and testing activities within the Study Area. Acoustic sources were divided into two categories,
impulsive and non-impulsive. Impulsive sounds feature a very rapid increase to high pressures, followed
by a rapid return to static pressure. For both non-impulsive and impulsive stressors, water column EFH
and HAPC within the Study Area may be temporarily impacted through an increase in the ambient sound
levels. While the level of ambient sound in the water column will return to normal immediately
following the completion of the training or testing exercise, thus resulting in only a temporary impact to
water column EFH, federally managed fish and invertebrate species may be affected during this period
within the vicinity of the stressor as a result of this brief alteration of the ambient noise level.
The analysis of the potential effects to fish and invertebrates as a result of impacts to the water column
habitats designated as EFH is limited to physical injury or mortality within the immediate vicinity of
where the stressor may occur. Hearing loss, auditory masking, physiological stress, and behavioral
reactions to impulsive stressors beyond the range of physical impacts are assumed but not quantified,
and are included with the physical impacts. If there is no physical injury or mortality anticipated, the
impact on water column EFH is assessed qualitatively. The qualitative assessment of hearing loss,
auditory masking, physiological stress, and behavioral reactions is based on the hearing and vocalization
capacities of fish and invertebrates.
Impulsive sounds are often produced by processes involving a rapid release of energy or mechanical
impacts (Hamernik and Hsueh 1991). Explosions and airgun impulses are examples of impulsive sound
4-3
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
sources. Non-impulsive sounds lack the rapid rise time and can have durations longer than those of
impulsive sounds. Sonar pings and underwater transponders are examples of non-impulsive sound
sources. The terms “impulsive” and “non-impulsive” were selected for use because they were deemed
more technically accurate and less confusing than the terms “explosive” and “acoustic” used in previous
documentation.
Fish Hearing and Vocalization
All fish have two sensory systems to detect sound in the water: the inner ear, which functions very much
like the inner ear in other vertebrates, and the lateral line, which consists of a series of receptors along
the fish’s body (Popper and Schilt 2008). The inner ear generally detects relatively higher-frequency
sounds, while the lateral line detects water motion relative to the fish at low frequencies (below a few
hundred Hertz) (Hastings and Popper 2005). Although hearing capability data only exist for fewer than
100 of the 32,000 fish species, current data suggest that most species of fish detect sounds from 50 to
1,000 Hertz (Hz), with few fish hearing sounds above 4 kHz (Popper 2008). It is believed that most fish
have their best hearing sensitivity from 100 to 400 Hz (Popper 2003). Additionally, some clupeids (shad
in the subfamily Alosinae) possess ultrasonic hearing (i.e., able to detect sounds above 100,000 Hz)
(Astrup 1999).
The inner ears of fish are directly sensitive to acoustic particle motion rather than acoustic pressure.
Although a propagating sound wave contains both pressure and particle motion components, particle
motion is most significant at low frequencies (less than a few hundred Hertz) and closer to the sound
source. However, a fish’s gas-filled swim bladder can enhance sound detection by converting acoustic
pressure into localized particle motion, which may then be detected by the inner ear. Fish with swim
bladders generally have more sensitive and higher-frequency hearing than fish without swim bladders
(Popper and Fay 2010). Some fish also have specialized structures such as small gas bubbles or gas-filled
projections that terminate near the inner ear. These fish have been called “hearing specialists,” while
fish that do not possess specialized structures have been referred to as “generalists” (Popper et al.
2003). In reality many fish species possess a continuum of anatomical specializations that may enhance
their sensitivity to pressure (versus particle motion), and thus higher frequencies and lower intensities
(Popper and Fay 2010).
Past studies indicated that hearing specializations in marine fish were quite rare (Amoser and Ladich
2005). However, more recent studies show there are more fish species than originally investigated by
researchers, such as deep-sea fish, that may have evolved structural adaptations to enhance hearing
capabilities (Buran et al. 2005; Deng et al. 2011). Marine fish families Holocentridae (squirrelfish and
soldierfish), Pomacentridae (damselfish), Gadidae (cod, hakes, and grenadiers), and Sciaenidae (drums,
weakfish, and croakers) have some members that can potentially hear sound up to a few kilohertz.
There is also evidence, based on the structure of the ear and the relationship between the ear and the
swim bladder, that at least some deep-sea species, including myctophids, may have hearing
specializations and thus be able to hear higher frequencies (Deng et al. 2011; Popper 1977; Popper
1980), although it has not been possible to do actual measures of hearing on these fish from great
depths.
Several species of reef fish tested show sensitivity to higher frequencies (i.e., over 1,000 Hz). The
hearing of the shoulderbar soldierfish (Myripristis kuntee) has an auditory range extending toward 3 kHz
(Coombs and Popper 1979), while other species tested in this family have been demonstrated to lack
this higher frequency hearing ability (e.g., Hawaiian squirrelfish [Adioryx xantherythrus] and saber
4-4
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
squirrelfish [Sargocentron spiniferum]). Some damselfish can hear frequencies of up to 2 kHz, but with
best sensitivity well below 1 kHz (Egner and Mann 2005; Kenyon 1996; Wright et al. 2005, 2007).
Sciaenid research by Ramcharitar et al. (2006) investigated the hearing sensitivity of weakfish (Cynoscion
regalis). Weakfish were found to detect frequencies up to 2 kHz. The sciaenid with the greatest hearing
sensitivity discovered thus far is the silver perch (Bairdiella chrysoura), which has responded to sounds
up to 4 kHz (Ramcharitar et al. 2004). Other species tested in the family Sciaenidae have been
demonstrated to lack this higher frequency sensitivity.
It is possible that the Atlantic cod (Gadus morhua, Family: Gadidae) is also able to detect high-frequency
sounds (Astrup and Mohl 1993). However, in Astrup and Mohl’s (1993) study it is feasible that the cod
was detecting the stimulus using touch receptors that were overdriven by very intense fish-finding sonar
emissions (Astrup 1999; Ladich and Popper 2004). Nevertheless, Astrup and Mohl (1993) indicated that
cod have high frequency thresholds of up to 38 kHz at 185–200 dB referenced to (re) 1 micropascal
(µPa), which likely only allows for detection of odontocete’s clicks at distances no greater than 33–98 ft.
(10–30 m) (Astrup 1999).
Experiments on several species of the Clupeidae (e.g., herrings, shads, and menhadens) have obtained
responses to frequencies between 40 and 180 kHz (Astrup 1999); however, not all clupeid species tested
have demonstrated this very-high-frequency hearing. Mann et al. (1998) reported that the American
shad can detect sounds from 0.1 to 180 kHz with two regions of best sensitivity: one from 0.2 to 0.8 kHz,
and the other from 25 to 150 kHz. This shad species has relatively high thresholds (about 145 dB re
1 µPa), which should enable the fish to detect odontocete clicks at distances up to about 656 ft. (200 m)
(Mann et al. 1997). Likewise, other members of the subfamily Alosinae, including alewife (Alosa
pseudoharengus), blueback herring (Alosa aestivalis), and Gulf menhaden (Brevoortia patronus), have
upper hearing thresholds exceeding 100–120 kHz. In contrast, the Clupeidae bay anchovy (Anchoa
mitchilli), scaled sardine (Harengula jaguana), and Spanish sardine (Sardinella aurita) did not respond to
frequencies over 4 kHz (Gregory and Clabburn 2003; Mann et al. 2001). Mann et al. (2005) found
hearing thresholds of 0.1–5 kHz for Pacific herring (Clupyea pallasii).
Two other groups to consider are the jawless fish (Superclass: Agnatha – lamprey) and the cartilaginous
fish (Class: Chondrichthyes – the sharks, rays, and chimeras). While there are some lampreys in the
marine environment, virtually nothing is known about their hearing capability. They do have ears, but
these are relatively primitive compared to the ears of other vertebrates, and it is unknown whether they
can detect sound (Popper and Hoxter 1987). While there have been some studies on the hearing of
cartilaginous fish, these have not been extensive. However, available data suggest detection of sounds
from 20 to 1,000 Hz, with best sensitivity at lower ranges (Casper et al. 2003; Casper and Mann 2006;
Casper and Mann 2009; Myrberg 2001). It is likely that elasmobranchs only detect low-frequency sounds
because they lack a swim bladder or other pressure detector.
Most of the other marine species investigated to date lack higher-frequency hearing (i.e., greater than
1,000 Hz). This notably includes sturgeon species tested to date that could detect sound up to 400 or
500 Hz (Lovell et al. 2005) and Atlantic salmon that could detect sound up to about 500 Hz (Hawkins and
Johnstone 1978; Kane et al. 2010).
Bony fish can produce sounds in a number of ways and use them for a number of behavioral functions
(Ladich 2008). Over 30 families of fish are known to use vocalizations in aggressive interactions, whereas
over 20 families are known to use vocalizations in mating (Ladich 2008). Sound generated by fish as a
4-5
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
means of communication is generally below 500 Hz (Slabbekoorn et al. 2010). The air in the swim
bladder is vibrated by the sound-producing structures (often muscles that are integral to the swim
bladder wall) and radiates sound into the water (Zelick et al. 1999). Sprague and Luczkovich (2004)
calculated that silver perch can produce drumming sounds ranging from 128 to 135 dB re 1 µPa. Female
midshipman fish apparently use the auditory sense to detect and locate vocalizing males during the
breeding season (Sisneros and Bass 2003).
Invertebrate Hearing and Vocalization
Very little is known about sound detection and use of sound by aquatic invertebrates (Budelmann
1992a, b; Montgomery et al. 2006; Popper et al. 2001). Organisms may detect sound by sensing either
the particle motion or pressure component of sound, or both. Aquatic invertebrates probably do not
detect pressure since many are generally the same density as water and few, if any, have air cavities
that would function like the fish swim bladder in responding to pressure (Budelmann 1992b; Popper et
al. 2001). Many aquatic invertebrates, however, have ciliated “hair” cells that may be sensitive to water
movements, such as those caused by currents or water particle motion very close to a sound source
(Budelmann 1992a, b; Mackie and Singla 2003). This may allow sensing of nearby prey or predators or
help with local navigation.
Aquatic invertebrates that can sense local water movements with ciliated cells include cnidarians,
flatworms, segmented worms, urochordates (tunicates), mollusks, and arthropods (Budelmann 1992a,
b; Popper et al. 2001). The sensory capabilities of corals and coral larvae are largely limited to detecting
water movement using receptors on their tentacles (Gochfeld 2004) and exterior cilia (Vermeij et al.
2010). Some aquatic invertebrates have specialized organs called statocysts for determination of
equilibrium and, in some cases, linear or angular acceleration. Statocysts allow an animal to sense
movement and may enable some species, such as cephalopods and crustaceans, to be sensitive to water
particle movements associated with sound (Hu et al. 2009; Kaifu et al. 2008; Montgomery et al. 2006;
Popper et al. 2001). Because any acoustic sensory capabilities, if present at all, are limited to detecting
water motion, and water particle motion near a sound source falls off rapidly with distance, aquatic
invertebrates are probably limited to detecting nearby sound sources rather than sound caused by
pressure waves from distant sources.
Both behavioral and auditory brainstem response studies suggest that crustaceans may sense sounds up
to 3 kHz, but best sensitivity is likely below 200 Hz (Goodall et al. 1990; Lovell et al. 2005; Lovell et al.
2006). Most cephalopods (e.g., octopus and squid) likely sense low-frequency sound below 1,000 Hz,
with best sensitivities at lower frequencies (Budelmann 1992b; Mooney et al. 2010; Packard et al. 1990).
A few may sense higher frequencies up to 1,500 Hz (Hu et al. 2009). Squid did not respond to toothed
whale ultrasonic echolocation clicks at sound pressure levels (SPLs) ranging from 199 to 226 dB re 1 μPa,
likely because these clicks were outside of squid hearing range (Wilson et al. 2007). However, squid
exhibited alarm responses when exposed to broadband sound from an approaching seismic airgun with
received levels exceeding 145–150 dB re 1 micropascal squared second (μPa2-s) root mean square
(McCauley et al. 2000).
Aquatic invertebrates may produce and use sound in territorial behavior, to deter predators, to find a
mate, and to pursue courtship (Popper et al. 2001). Some crustaceans produce sound by rubbing or
closing hard body parts together, such as lobsters and snapping shrimp (Au and Banks 1998; Latha et al.
2005; Patek and Caldwell 2006). The snapping shrimp chorus makes up a significant portion of the
ambient noise budget in many locales (Au and Banks 1998; Cato and Bell 1992). Each click is up to
215 dB re 1 µPa, with a peak around 2–5 kHz (Au and Banks 1998; Heberholz and Schmitz 2001). Other
4-6
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
crustaceans make low-frequency rasping or rumbling noises, perhaps used in defense or territorial
display, that are often obscured by ambient noise (Patek and Caldwell 2006; Patek et al. 2009).
Reef noises, such as fish pops and grunts, sea urchin grazing (around 1.0–1.2 kHz), and snapping shrimp
noises (around 5 kHz) (Radford et al. 2010), may be used as a cue by some aquatic invertebrates. Nearby
reef noises were observed to affect movements and settlement behavior of coral and crab larvae (Jeffs
et al. 2003; Radford et al. 2007; Stanley et al. 2010; Vermeij et al. 2010). Larvae of other crustacean
species, including pelagic and nocturnally emergent species that benefit from avoiding predators
associated with coral reefs, appear to avoid reef noises (Simpson et al. 2011). Detection of reef noises is
likely limited to short distances (less than 330 ft. [100 m]) (Vermeij et al. 2010).
4.1.1.1
Non-Impulsive Stressors
Sonar and other non-impulsive sound sources (e.g., vessel noise) emit sound waves into the water to
detect objects, safely navigate, transiting, and communicate. This section analyzes the potential impacts
of these acoustic sources on EFH and HAPC resulting from training and testing activities within the Study
Area. Unlike explosives and other impulsive stressors, only water column EFH and HAPC within the Study
Area may be temporarily impacted by non-impulsive sound effects. The analysis of impacts on the water
column environment for fish and invertebrates is limited to physical injury or mortality where those
impacts may occur. Hearing loss, auditory masking, physiological stress, and behavioral reactions to
impulsive stressors beyond the range of physical impacts are assumed but not quantified, and are
included with the physical impacts. If there is no physical injury or mortality anticipated, the impact on
water column EFH is assessed qualitatively.
4.1.1.1.1
Sonar and Other Active Acoustic Sources
Most active systems operate within specific frequencies although some harmonic frequencies may be
emitted at lower SPLs. Sonar use associated with ASW would emit the most non-impulsive sound
underwater during training and testing activities. Sonar use associated with MIW would also contribute
a notable portion of overall non-impulsive sound. Other sources of non-impulsive noise include acoustic
communications and other sound sources used in testing. General categories of sonar systems are
described in Section 2.2.1 (Sonar and Other Active Acoustic Sources). The hours of usage of each
acoustic source class proposed are shown in Table 4-3.
Underwater sound propagation is highly dependent upon environmental characteristics such as
bathymetry, bottom type, water depth, temperature, and salinity. The sound received at a particular
location will be different than near the source due to the interaction of many factors, including
propagation loss; how the sound is reflected, refracted, or scattered; the potential for reverberation;
and interference due to multi-path propagation.
4-7
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 4-3: Sonar and Other Active Acoustic Source Classes for the Proposed Action
(Annual Hours or Number of Items)
Source Class
Category
Airguns (AG) Used
during swimmer
defense and diver
deterrent activities
Anti-Submarine
Warfare (ASW)
Tactical sources used
during anti-submarine
warfare training and
testing activities
Low-Frequency (LF)
Sources that produce
signals less than 1 kHz
High-Frequency (HF)
and Very HighFrequency (VHF):
Tactical and nontactical sources that
produce signals
greater than 10 kHz
but less than 180 kHz
Mid-Frequency (MF)
Tactical and nontactical sources that
produce signals from 1
to 10 kHz
Annual
Source Use
for Training
(hours
except as
noted)
Annual
Source Use
for Testing
(hours
except as
noted)
Source
Class
Description
AG*
Up to 60 in. airguns
(e.g., Sercel Mini-G)
0
308
ASW1
Mid-frequency Deep Water Active
Distributed System (DWADS)
0
144
ASW2*
Mid-frequency Multistatic Active
Coherent sonobuoy (e.g.,
AN/SSQ-125)
160
500
ASW3
Mid-frequency towed active acoustic
countermeasure systems (e.g.,
AN/SLQ-25)
3,574
361
ASW4*
Mid-frequency expendable active
acoustic device countermeasures (e.g.,
MK 3)
11
0
LF4
Low-frequency sources equal to
180 dB and up to 200 dB
0
123
LF5
Low-frequency sources less than
180 dB
0
11
LF6
Low-frequency sonars currently in
development (e.g., anti-submarine
warfare sonars associated with the
Littoral Combat Ship)
0
40
HF1
Hull-mounted submarine sonars (e.g.,
AN/BQQ-10)
100
13
HF4
Mine detection, classification, and
neutralization sonar (e.g., AN/SQS-20)
716
344
HF5
Active sources (greater than 200 dB)
not otherwise binned
0
336
HF6
Active sources (equal to 180 dB and up
to 200 dB) not otherwise binned
1,036
137
MF1
Hull-mounted surface ship sonars (e.g.,
AN/SQS-53C and AN/SQS-60)
1,856
16
MF2
Hull-mounted surface ship sonars (e.g.,
AN/SQS-56)
596
29
MF3
Hull-mounted submarine sonars (e.g.,
AN/BQQ-10)
191
1
3
4-8
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 4-3: Sonar and Other Active Acoustic Source Classes for the Proposed Action
(Annual Hours or Number of Items) (continued)
Source Class
Category
Source
Class
Description
Annual
Source Use
for Training
(hours
except as
noted)
Mid-Frequency (MF)
Tactical and nontactical sources that
produce signals from 1
to 10 kHz
(continued)
MF4
Helicopter-deployed dipping sonars
(e.g., AN/AQS-22 and AN/AQS-13)
144
70
MF5*
Active acoustic sonobuoys (e.g.,
DICASS)
1,908
680
MF6*
Active underwater sound signal
devices (e.g., MK 84)
0
33
MF8
Active sources (greater than 200 dB)
not otherwise binned
0
123
MF9
Active sources (equal to 180 dB and
up to 200 dB) not otherwise binned
0
47
MF10
Active sources (greater than 160 dB,
but less than 180 dB) not otherwise
binned
0
231
MF11
Hull-mounted surface ship sonars
with an active duty cycle greater than
80%
308
16
MF12
High duty cycle – variable depth
sonar
472
184
M3
Mid-frequency acoustic modems
(greater than 190 dB)
0
112
SD1
High-frequency sources with short
pulse lengths, used for the detection
of swimmers and other objects for the
purpose of port security.
0
2,341
TORP1*
Lightweight torpedo (e.g., MK 46, MK
54, or Surface Ship Defense System)
11
104
TORP2*
Heavyweight torpedo (e.g., MK 48)
50
12
Acoustic Modems (M)
Transmit data
acoustically through the
water
Swimmer Detection
Sonar (SD) Used to
detect divers and
submerged swimmers
Torpedoes (TORP)
Source classes
associated with active
acoustic signals
produced by torpedoes
Annual
Source Use
for Testing
(hours
except as
noted)
* These sources are measured by items, not hours.
Notes: dB = decibels, DICASS = Directional Command Activated Sonobuoy, in.3 = cubic inches, kHz = kilohertz
A very simple estimate of sonar transmission loss can be calculated using the spherical spreading law,
TL = 20 log10r, where r is the distance from the sound source and TL is the transmission loss in decibels.
While a simple example is provided here for illustration, the Navy Acoustic Effects Model takes into
account the influence of multiple factors to predict acoustic propagation (U.S. Department of the Navy
2012). The simplified estimate of spreading loss for a ping from a hull-mounted tactical sonar with a
representative source level of 235 dB re 1 µPa is shown in Figure 4-1. The figure shows that sound levels
drop off significantly near the source, followed by a more steady reduction with distance. Most
non-impulsive sound sources used during training and testing have sound source levels lower than this
example.
4-9
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 4-1: Estimate of Spreading Loss for a 235 Decibels Referenced to 1 Micropascal Sound Source Assuming
Simple Spherical Spreading Loss
Most use of active acoustic sources involves a single unit or several units (ship, submarine, aircraft, or
other platform) employing a single active sonar source in addition to sound sources used for
communication, navigation, and measuring oceanographic conditions. Anti-submarine warfare activities
may also use an acoustic target or an acoustic decoy.
Anti-Submarine Warfare Sonar Systems
Sonar used in ASW are deployed on many platforms and are operated in various ways. Anti-submarine
warfare active sonar is usually mid-frequency (1–10 kHz) because mid-frequency sound balances
sufficient resolution to identify targets and distance within which threats can be identified.
•
•
•
•
•
Ship tactical hull-mounted sonar contributes the largest portion of overall non-impulsive sound.
Duty cycle can vary from about a ping per minute to continuously active. Sonar can be
wide-ranging in a search mode or highly directional in a track mode.
A submarine‘s mission revolves around its stealth; therefore, a submarine’s mid-frequency sonar
is used infrequently because its use would also reveal a submarine’s location.
Aircraft-deployed, mid-frequency, ASW systems include omnidirectional dipping sonar
(deployed by helicopters) and omnidirectional sonobuoys (deployed from various aircraft),
which have a typical duty cycle of several pings per minute.
Acoustic decoys that continuously emulate broadband vessel sound or other vessel acoustic
signatures may be deployed by ships and submarines.
Torpedoes use directional high-frequency sonar when approaching and locking onto a target.
Practice targets emulate the sound signatures of submarines or repeat received signals.
Anti-submarine warfare activities for all platforms typically would occur within and adjacent to existing
OPAREAs beyond 3 nm, with the exception of sonar dipping activities conducted by helicopters closer to
shore. In addition, hull-mounted sonars may occasionally be used in port during system maintenance
4-10
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
and testing. Most ASW activities involving submarines or submarine targets would occur in waters
greater than 600 ft. (183 m) deep due to safety concerns about running aground at shallower depths.
Most events usually occur over a limited area and are completed in less than 1 day, often within a few
hours. Multi-day ASW events requiring coordination of movement and effort between multiple
platforms with active sonar over a larger area occur less often, but constitute a large portion of the
overall non-impulsive underwater noise that would be produced by Navy activities.
Mine Warfare Sonar Systems
Sonar used to locate mines and other small objects is typically high frequency, which provides higher
resolution. Mine detection sonar is deployed at variable depths on moving platforms to sweep a
suspected mined area (towed by ships, helicopters, or unmanned underwater vehicles). Mid-frequency
hull-mounted sonar can also be used in an object detection mode known as “Kingfisher” mode. Mine
detection sonar use would be concentrated in areas where practice mines are deployed, typically in
water depths less than 200 ft. (61 m). Most events usually occur over a limited area and are completed
in less than 1 day, often within a few hours.
Other Active Acoustic Sources
Active sound sources used for navigation and obtaining oceanographic information (e.g., depth,
bathymetry, and speed) are typically directional, have high duty cycles, and cover a wide range of
frequencies, from mid frequency to very high frequency. These sources are similar to the navigation
systems on standard large commercial and oceanographic vessels. Sound sources used in
communications are typically high frequency or very high frequency. These sound sources could be used
by vessels during most activities and while transiting throughout the Study Area.
Potential Impacts on Biological Components of the Water Column
Sonar and other active acoustic sources would not disturb the substrate, but they could affect the
pelagic water column as a habitat for fish and invertebrates. Potential impacts on the water column
habitat from active acoustic sources would mainly include impacts on species occupying the water
column and their prey, including fish and invertebrates. These impacts could include injury or death,
hearing loss, auditory masking, and physiological stress or behavioral reactions for those species.
Potential direct injuries from non-impulsive sound sources, such as sonar, are unlikely because of the
relatively lower peak pressures and slower rise times than potentially injurious sources such as
explosives. Non-impulsive sources also lack the strong shock wave such as that associated with an
explosion. Therefore, direct injury is not likely to occur from exposure to non-impulsive sources such as
sonar, or subsonic aircraft noise for the reasons discussed below. The theories of sonar-induced acoustic
resonance, bubble formation, neurotrauma, and lateral line system injury were studied under
experimental conditions.
Two reports examined the effects of mid-frequency sonar-like signals (1.5–6.5 kHz) on larval and
juvenile fish of several species (Jørgensen et al. 2005; Kvadsheim and Sevaldsen 2005). In the first study,
Kvadsheim and Sevaldsen (2005) showed that intense sonar activities in herring spawning areas affected
less than 0.3 percent of the total juvenile stock. The second study, Jørgensen et al. (2005) exposed larval
and juvenile fish to various sounds in order to investigate potential effects on survival, development,
and behavior. The study used herring (Clupea harengus) (standard lengths 2–5 cm), Atlantic cod (Gadus
morhua) (standard length 2 and 6 cm), saithe (Pollachius virens) (4 cm), and spotted wolffish (Anarhichas
minor) (4 cm) at different developmental stages. The researchers placed the fish in plastic bags 10 ft.
4-11
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
(3 m) from the sound source and exposed them to between four and 100 pulses of 1-second duration of
pure tones at 1.5, 4, and 6.5 kHz. The fish in only two groups out of the 82 tested exhibited any adverse
effects. These two groups were both composed of herring, a hearing specialist, and were tested with
SPLs of 189 dB re 1 µPa, which resulted in a post-exposure mortality of 20–30 percent. In the remaining
80 tests, there were no observed effects on behavior, growth (length and weight), or the survival of fish
that were kept as long as 34 days post exposure. While statistically significant losses were documented
in the two groups impacted, the researchers only tested that particular sound level once, so it is not
known if this increased mortality was due to the level of the test signal or to other unknown factors.
High SPLs may cause bubbles to form from micronuclei in the blood stream or other tissues of animals,
possibly causing embolism damage (Ketten 1998). Fish have small capillaries where these bubbles could
be caught and lead to the rupturing of the capillaries and internal bleeding. It has also been speculated
that this phenomena could also take place in the eyes of fish due to potentially high gas saturation
within the fish’s eye tissues (Popper and Hastings 2009).
As reviewed in Popper and Hastings (2009), Hastings (1990; 1995) found ‘acoustic stunning’ (loss of
consciousness) in blue gouramis (Trichogaster trichopterus) following an 8-minute exposure to a 150 Hz
pure tone with a peak SPL of 198 dB re 1 µPa. This species of fish has an air bubble in the mouth cavity
directly adjacent to the animal’s braincase that may have caused this injury. Hastings (1990; 1995) also
found that goldfish exposed to 2 hours of continuous wave sound at 250 Hz with peak pressures of 204
dB re 1 µPa, and fathead minnows exposed to 0.5 hour of 150 Hz continuous wave sound at a peak level
of 198 dB re 1 µPa did not survive.
The only study on the effect of exposure of the lateral line system to continuous wave sound (conducted
on one freshwater species) suggests no effect on these sensory cells by intense pure tone signals
(Hastings et al. 1996).
The most familiar effect of exposure to high-intensity sound is hearing loss, meaning an increase in the
hearing threshold. This phenomenon is called a noise-induced threshold shift, or simply a threshold shift
(Miller 1974). A temporary threshold shift (TTS) is a temporary, recoverable loss of hearing sensitivity
over a small range of frequencies related to the sound source to which the fish was exposed. A TTS may
last several minutes to several weeks and the duration is related to the intensity of the sound source
and the duration of the sound (including multiple exposures). A permanent threshold shift (PTS) is
non-recoverable, results from the destruction of tissues within the auditory system, and can occur over
a small range of frequencies related to the sound exposure. As with TTS, the animal does not become
deaf but requires a louder sound stimulus (relative to the amount of PTS) to detect a sound within the
affected frequencies; however, in this case, the affect is permanent.
Permanent hearing loss has yet to be documented in fish. The sensory hair cells of the inner ear in fish
can regenerate after they are damaged, unlike in mammals where sensory hair cell loss is permanent
(Lombarte and Popper 1994; Smith et al. 2006). As a consequence, any hearing loss in fish may be as
temporary as the timeframe required to repair or replace the sensory cells that were damaged or
destroyed (Smith et al. 2006).
While some marine fish may be able to detect mid-frequency sounds, most marine fish are hearing
generalists and have their best hearing sensitivity below mid-frequency sonar. Behavioral responses, if
they occur, would be brief, and unlikely to have any substantial costs. Kvadsheim and Sevaldsen (2005)
reported no behavioral reaction of herrings to low- and mid-frequency sonar. Sustained auditory
4-12
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
damage is not expected. Sensitive life stages (juvenile fish, larvae, and eggs) very close to the sonar
source may experience injury or mortality, but area-wide effects would likely be minor. For these
reasons, the use of mid-frequency sonar would not significantly affect fish or invertebrate populations.
Doksæter et al. (2009) investigated the potential behavioral effects of sonar on Atlantic herring (a
clupeid). The reactions of free-swimming herring to sonar transmissions at 1–2 kHz and 6–7 kHz were
compared with the playback of recorded killer whale (Orcinus orca) feeding sounds. Received SPLs for
the lower frequency range were 127–197 dB re 1 µPa, and for the higher range were 139–209 dB re
1 µPa. The killer whale feeding sounds ranged from approximately 800 Hz to 20 kHz and source levels of
150–160 dB re 1 µPa at 1 m. The reactions of the herring, which were generally located between 10 and
50 m in the water column, were monitored by two upward-looking echosounders. No vertical or
horizontal fleeing reactions to the sonar transmissions were observed as the vessels passed multiple
times over the stock of herring. By contrast, the killer whale feeding sounds induced both vertical and
horizontal fleeing reactions in the herring. The authors concluded that the operation of sonar resulted in
no effect on the behavior of the herring stock; therefore, there would be no large-scale adverse effects
to the herring stock.
Since high-frequency sound attenuates quickly in the water, high levels of sound would be restricted to
areas near the source. Most species would probably not hear these sounds and would therefore
experience no disturbance; even for fish able to hear sound at high frequencies, only short-term
exposure would occur, and effects would be transitory and of little biological consequence. Although
some species may be able to produce sound at higher frequencies (greater than 1 kHz), vocal marine fish
largely communicate below the range of mid-frequency levels used by most sonars. Further, most
marine fish species are not expected to be able to detect sounds in the mid-frequency range of the
operational sonars. The fish species that are known to detect mid-frequencies (including most clupeids)
do not have their best sensitivities in the range of the operational sonars. Thus, these fish can only hear
mid-frequency sounds when sonars are operating at high energy levels or the fish are in proximity to the
sonars. Considering the low-frequency detection of most marine species and the limited time of
exposure due to the moving sound sources, most mid-frequency active sonar used in the Study Area
would not have the potential to substantially mask key environmental sounds.
While not likely for mid-frequency active sonars, the low-frequency active sonars may have a greater
ability to mask biologically important sounds due to their operational frequency range coinciding with
range detectable and use for communication by most marine fish species. However, low-frequency
active usage is rare and most low-frequency active operations are conducted in deeper waters. The
majority of fish species, including those that are the most highly vocal, exist on the continental shelf and
within nearshore, estuarine areas. Fish within a few tens of kilometers around low-frequency active
sonar could experience brief periods of masking while the system is used, with effects most pronounced
closer to the source. However, overall effects would be temporary and infrequent.
Exposure of many fish species to sonars and other acoustic sources has the potential to result in stress
to the animal and may also elicit alterations in normal behavior patterns (e.g., swimming, feeding,
resting, spawning, etc.). Such impacts may have the potential to affect the long-term growth and
survival of an individual. However, due to the temporary and infrequent nature of sonar use in the Study
Area, the resulting stress on fish is not likely to impact the health of resident populations, because
behavioral changes are not expected to have lasting effects on the survival, growth, or reproduction of
fish species.
4-13
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
In summary, sonar use could affect prey species (including fish and invertebrates) by masking
ecologically important sounds, inducing stress, altering behaviors, or changing hearing thresholds which
could affect water column habitat. Hearing specialists are more likely to be impacted than generalists
due to their ability to detect both low- and mid-frequency sounds. However, any such effects would be
temporary and infrequent as a vessel operating mid-frequency sonar transits an area. There is no
information available to suggest that exposure to non-impulsive acoustic sources results in mortality.
Training Activities
Training activities involving the use of sonar could occur in multiple locations including offshore waters,
inland waters such as Inner Apra Harbor, and while pierside. The annual hours of sonar and other active
acoustic sources from Navy training activities are listed in Table 4-3.
Training activities involving the use of non-impulsive acoustic stressors may reduce the quality of water
column EFH and HAPC through the increase in ambient noise levels. This potential reduction would be
localized to the area of the training activity and be only temporary in duration. The quality of the water
column as EFH and HAPC would be restored to normal levels immediately following the completion of
the training activities. There is no anticipated effect of non-impulsive acoustic sources, including sonar,
on benthic substrates and biogenic habitats designated as EFH or on HAPCs.
Testing Activities
Testing activities involving the use of sonar could occur in multiple locations including offshore waters,
inland waters such as bays, and while pierside. The annual hours of sonar and other active acoustic
sources from Navy testing activities are listed in Table 4-3.
Testing events involving the use of non-impulsive acoustic stressors may reduce the quality of water
column EFH and HAPC through the increase in ambient noise levels. This potential reduction would be
localized to the area of the testing event and be only temporary in duration. The quality of the water
column as EFH and HAPC would be restored to normal levels immediately following the completion of
the testing events. There is no anticipated effect of non-impulsive acoustic sources, including sonar, on
benthic substrates and biogenic habitats designated as EFH or on HAPCs.
4.1.1.1.2
Vessel Noise
Naval vessels would produce low-frequency, broadband underwater sound. In the EEZ, Navy ships are
estimated to contribute roughly 1 percent of the total energy due to large vessel broadband noise
(Mintz and Filadelfo 2011; Mintz and Parker 2006).
Vessel movements involve transit to and from ports to various locations within the Study Area, and
many ongoing and proposed training and testing activities within the Study Area involve maneuvers by
various types of surface ships, boats, and submarines (collectively referred to as vessels). Operations
involving vessel movements occur intermittently and are variable in duration, ranging from a few hours
up to 2 weeks. Additionally, a variety of smaller craft will be operated within the Study Area.
Potential impacts on the water column habitat from vessel movements would mainly include impacts on
prey species, including fish and invertebrates. Vessel movements have the potential to expose fish and
invertebrates to sound and general disturbance, which could result in short-term behavioral or
physiological responses (e.g., avoidance, stress, increased heart rate). While vessel movements have the
potential to expose fish and invertebrates occupying the water column to sound and general
disturbance, potentially resulting in short-term behavioral or physiological responses, such responses
4-14
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
would not be expected to compromise the general health or condition of individual fish or populations
of invertebrates.
Based on the information above, water column EFH and HAPC would not be adversely affected by the
vessel noise generated from Navy training and testing activities. There is no anticipated effect of vessel
noise, on benthic substrates and biogenic habitats designated as EFH or on HAPCs.
4.1.1.2
Impulsive Stressors
Underwater explosions, swimmer defense airguns, and weapons firing noise all produce a rapid pressure
rise and high peak pressure (see relevant section below for supporting details). This section analyzes the
potential impacts of these explosive and impulsive sources on EFH and HAPC resulting from training and
testing activities within the Study Area. Unlike non-impulsive stressors, all habitats within the Study Area
may be physically impacted by impulsive sound effects. The analysis of impacts on the water column
environment presented below is limited to physical injury or mortality for prey species, such as fish and
invertebrates. Section 4.1.1.1 (Non-Impulsive Stressors) describes the non-lethal impacts of sound on
fish and invertebrates.
4.1.1.2.1
Explosives
Explosive detonations are associated with high-explosive ordnance, including bombs, missiles,
torpedoes, and naval gun shells; mines and charges; explosive sonobuoys; anti-swimmer grenades, and
ship shock trial charges. Most explosive detonations during training and testing would be at or below
the water surface, although charges associated with mine neutralization could occur near the ocean
bottom. While most detonations would occur in waters greater than 200 ft. (61 m) in depth, mine
neutralization events would typically occur in shallower waters (less than 200 ft. [61 m]). Training and
testing activities using explosions generally would not occur within 3 nm of shore, with the exception of
civilian port defense and designated underwater detonation areas in nearshore waters.
In general, explosive events would consist of a single explosion or multiple explosions over a short
period. During training, all large, high-explosive bombs would be detonated near the surface over deep
water. Bombs with high-explosive ordnance would be fused to detonate on contact with the water, and
it is estimated that 99 percent of them would explode within 5 ft. of the ocean surface (U.S. Department
of the Navy 2005). Table 4-4 shows parameters of some ordnance detonated during training and testing
activities.
4-15
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 4-4: Representative Ordnance, Net Explosive Weights, and Detonation Depths
Ordnance
M3A2 anti-swimmer concussion grenades
76-millimeter round
Sonobuoy charge
Airborne mine neutralization system
(AMNS) charges
Hellfire AGM 114 rocket
5 in. Naval gunfire
Underwater mine neutralization charges
Maverick missile
MK-20 bomb
MK-82 bomb
MK-83 bomb
Explosive ordnance detonation (EOD)
charges
MK-48 torpedo
MK-84 bomb
Net Explosive Weight (lb.)
0.5
2
5
5
8
8
20
100
110
192
416
5, 10, 75, 600
650
945
Detonation Depth
At or just below water’s surface
1 ft. (0.3 m)
Throughout the water column
Subsurface
At or just below water’s surface
1 ft. (0.3 m)
Throughout the water column
At or just below water’s surface
2–3 ft. (0.6–0.9 m)
2–3 ft. (0.6–0.9 m)
2–3 ft. (0.6–0.9 m)
Throughout the water column
Subsurface
2–3 ft. (0.6–0.9 m)
Notes: ft. = foot/feet, m = meter(s), lb. = pound(s)
Underwater explosions create a cavity filled with high-pressure gas, which pushes the water out against
the opposing external hydrostatic pressure. At the instant of explosion, a certain amount of gas is
instantaneously generated at high pressure and temperature, creating a bubble. In addition, the heat
causes a certain amount of water to vaporize, adding to the volume of the bubble. This action
immediately begins to force the water in contact with the blast front in an outward direction. This
intense pressure wave, called a “shock wave,” passes into the surrounding medium and travels faster
than the speed of sound. Noise associated with the blast is also transmitted into the surrounding
medium as acoustic waves. As the pressure waves generated by the explosion travel, they will interact
with the surface and seafloor, lose energy, and be perceived as acoustic waves.
The detonation depth of an explosive is important because of the propagation effect known as surfaceimage interference. For sources located near the sea surface, a distinct interference pattern arises from
reflection from the water's surface. As the source depth or the source frequency decreases, these two
paths increasingly, destructively interfere with each other, reaching total cancellation at the surface
(barring surface reflection scattering loss). Since most explosive sources used in military activities are
munitions that detonate essentially upon impact, the effective source depths are quite shallow and,
therefore, the surface-image interference effect can be pronounced.
Potential Impacts on the Physical Components of the Water Column
An explosion detonated near the surface would not disturb the substrate, but the shock wave could
affect the pelagic water column as a habitat for fish and invertebrates. The expanding gases can set up a
pulsating bubble whose recurring pressure waves also may contribute significantly to damage. Many
animals, especially smaller animals, are unlikely to survive if they are present in the region of bulk
cavitation. Cavitation occurs when shock waves, which are generated by the underwater detonation of
an explosive charge, propagate to the surface and are reflected back into the water as rarefaction (or
negative pressure) waves. These rarefaction waves cause a state of tension to occur within a large
region of water. Since water cannot ordinarily sustain a significant amount of tension, it cavitates and
the surrounding pressure drops to the vapor pressure of water. The region in which this occurs is known
4-16
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
as the cavitation region, and includes all water cavitating at any time after the detonation of the
explosive charge. The upper and lower boundaries form what is referred to as the cavitation envelope
(U.S. Department of the Navy 2008). A water hammer pulse is generated when the upper and lower
layers of the cavitation region rejoin (close).
Concern about potential fish mortality associated with the use of at-sea explosives led military
researchers to develop mathematical and computer models that predict safe ranges for fish and other
animals from explosions of various sizes (Goertner 1982; Goertner et al. 1994; Yelverton et al. 1975).
Young (1991) provides equations that allow estimation of the potential effect of underwater explosions
on fish possessing swim bladders using a damage prediction method developed by Goertner (1982).
Young’s parameters include the size of the fish and its location relative to the explosive source, but are
independent of environmental conditions (e.g., depth of fish and explosive shot frequency). An example
of such model predictions is shown in Table 4-5, which lists estimated explosive-effects ranges using
Young’s (1991) method for fish possessing swim bladders exposed to explosions that would typically
occur during training exercises. The 10 percent mortality range is the distance beyond which 90 percent
of the fish present would be expected to survive. It is difficult to predict the range of more subtle effects
causing injury but not mortality (Continental Shelf Associates Inc. 2004).
Table 4-5: Estimated Explosive Effects Ranges for Fish with Swim Bladders
Training Operation and Type
of Ordnance
10% Mortality Range (ft.)
NEW (lb.)
Depth of
Explosion (ft.)
1 oz. Fish
1 lb. Fish
30 lb. Fish
MK-103 Charge
0.002
10
40
28
18
AMNS Charge
3.24
20
366
255
164
20
30
666
464
299
Mine Neutralization
20 lb. NEW UNDET Charge
Missile Exercise
Hellfire
8
3.3
317
221
142
100
3.3
643
449
288
8
1
244
170
109
MK-20
109.7
3.3
660
460
296
MK-82
192.2
3.3
772
539
346
MK-83
415.8
3.3
959
668
430
MK-84
945
3.3
1,206
841
541
Maverick
Firing Exercise with IMPASS
HE Naval Gun Shell, 5-inch
Bombing Exercise
Notes: ft. = foot/feet, HE = high explosive, IMPASS = Integrated Maritime Portable Acoustic Scoring and Simulation, lb. =
pound(s), NEW = Net Explosive Weight, oz. = ounce, UNDET = Underwater Detonation
Fish not killed or driven from a location by an explosion might change their behavior, feeding pattern, or
distribution. Changes in behavior of fish have been observed as a result of sound produced by
explosives, with effect intensified in areas of hard substrate (Wright 1982). Fish which ascend too
quickly, a typical response to fear or to avoid negative stimuli, might experience an increase in the
volume of gas-filled organs due to the reduction in ambient pressure. The resulting inflation might
render the fish unable to immediately return to its normal habitat depth because the expanded organs
make the buoyancy of the fish too great to overcome by swimming downward. Stunning from pressure
waves could also temporarily immobilize fish, making them more susceptible to predation.
4-17
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
The few studies of marine invertebrates (crustaceans and mollusks) exposed to explosions show a range
of impacts, from mortality close to the source to no observable effects. Limited studies of crustaceans
have examined mortality rates at various distances from detonations in shallow water (Aplin 1947;
Chesapeake Biological Laboratory 1948; Gaspin et al. 1976). Similar studies of mollusks have shown
them to be more resistant than crustaceans to explosive impacts (Chesapeake Biological Laboratory
1948; Gaspin et al. 1976). Other invertebrates found in association with mollusks, such as sea
anemones, polychaete worms, isopods, and amphipods, were observed to be undamaged in areas near
detonations (Gaspin et al. 1976). Using data from these experiments, Young (1991) developed curves
that estimate the distance from an explosion beyond which at least 90 percent of certain marine
invertebrates would survive, depending on the weight of the explosive (Figure 4-2). In deeper waters
where most detonations would occur near the water surface, most benthic marine invertebrates would
be beyond the 90 percent survivability ranges shown above, even for larger explosives (up to source
class E12 [601–1,000 lb. NEW]).
Source: Young 1991
Figure 4-2: Prediction of Distance to 10 Percent Mortality of Marine Invertebrates Exposed to an Underwater
Explosion
The number of fish or invertebrates affected by an underwater explosion would depend on the
population density in the vicinity of the blast, as well as factors discussed above such as NEW, depth of
the explosion, and fish size. For example, if an explosion occurred in the middle of a dense school of
menhaden, herring, or other schooling fish, a large number of fish could be killed. Individually, such
explosions represent minimal mortality in terms of the total population of such fish in the Study Area.
The cumulative effect of multiple explosions over a period of time could have greater than minimal
impacts on fish or invertebrate populations, but this is very difficult to quantify without density and
biomass estimates of fish within the impact footprint.
The worst case scenario for explosive impacts on fish and invertebrates in the water column is based on
information from Table 4-4 (representative explosive munitions), Table 4-5 (10 percent mortality range
for 30 lb. (13.6 kg) fish, and Figure 4-2 (10 percent mortality range for crab). The range to less than 10
4-18
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
percent mortality is very similar for both a 30 lb. (13.6 kg) fish and a shrimp. The total impact area
assumes no overlap in footprints, which is unlikely considering the point of targets in training and testing
activities (e.g., hit the target). Such calculations provide one of the variables necessary in determining
the level impact. A determination of population level impacts requires more information on the density
and biomass of managed species and life-stages in the Study Area than is currently available.
Potential Impacts to Benthic Substrates and Biogenic Habitats
Mine neutralization training and underwater demolition qualification/certification activities would
involve explosions on or near the seafloor, which could affect benthic substrates and biogenic habitats.
Table 4-6 lists training and testing activities that include seafloor explosions, along with the location of
the activity and the associated explosives charges. Primarily soft-bottom habitat would be utilized for
underwater detonations. Cobble, rocky reef, and other hard bottom habitat may be scattered
throughout the area, but those areas would be avoided during training to the maximum extent
practicable.
Table 4-6: Training and Testing Activities that Include Seafloor Explosions
Activity
Training
Mine Neutralization
(Explosive
Ordnance Disposal)
Underwater
Demolition
Qualification/
Certification
Explosive
Charge (lb.
1
NEW )
Underwater
Detonations
1–20 lb.
20
MIRC mine neutralization sites
1–20 lb.
30
MIRC underwater demolition sites
1–20 lb.
24
Study Area
Location
Testing
MCM Mission
Package Testing
1
NEW is the actual weight in pounds of explosive mixtures or compounds
Notes: NEW = Net Explosive Weight, lb. = pound(s), MIRC = Mariana Islands Range Complex, MCM = Mine
Countermeasure Exercise
The determination of effect for training and testing activities on the seafloor is based on the largest netweight charge for each training activity which is 20 lb. (9.1 kg) NEW explosions. Explosions produce high
energies that would be partially absorbed and partially reflected by the seafloor. Hard bottoms would
mostly reflect the energy (Berglind, et al. 2009), whereas a crater would be formed in soft bottom
(Gorodilov & Sukhotin 1996). The area and depth of the crater would vary according to depth, bottom
composition, and size of the explosive charge. The relationship between crater size and depth of water
is non-linear, with relatively small crater sizes in the shallowest water, followed by a spike in size at
some intermediate depth, and a decline to an average flat-line at greater depth (Gorodilov & Sukhotin
1996; O'Keeffe & Young 1984).
In general, training and testing activities that include seafloor detonations occur in water depths ranging
from 6 ft. (1.8 m) to about 100 ft. (30 m). Based on Gorodilov & Sukhotin (1996), the depth (h) and
radius (R) of a crater from an underwater explosion over soft bottom is calculated using the charge
4-19
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
radius (r0) 4 multiplied by a number determined by solving for h or R along a non-linear relationship
between [depth of water/r0] and [h or R/r0]. The area of impacted substrate for each 20 lb. (9.1 kg)
underwater explosion on the seafloor would be approximately 366 ft.2 (34 m2). The radii of craters are
expected to vary little among unconsolidated sediment types. On sediment types with non-adhesive
particles (everything except clay), the impacts should be temporary as these areas should recover;
craters in clay may persist for years (O'Keeffe and Young 1984). The production of craters in soft bottom
could uncover subsurface hard bottom, altering marine substrate types.
Hard substrates reflect more energy from bottom detonations than do soft bottoms (Keevin and
Hempen 1997). The amount of consolidated substrate (i.e., bedrock) converted to unconsolidated
sediment by surface explosions varies according to material types and degree of consolidation (i.e.,
rubble, bedrock). Because of a lack of accurate and specific information on hard bottom types, the
impacted area is assumed to be equal to the area of soft bottom impacted. Potential exists for fracturing
and damage to hard-bottom habitat if underwater detonations occur over that type of habitat.
Training Activities
Under the Proposed Action, an estimated 50 underwater explosions per year would occur on or near the
seafloor within the Study Area, as identified in Table 4-6. Underwater explosions near the seafloor
would occur in the MIRC mine neutralization sites. Underwater explosives placed on or near the seafloor
would range from 1 to 20 lb. (0.4 to 9.1 kg) NEW. Figures 4-3 and 4-4 show the mine neutralization sites
in relation to vegetation and coral coverage.
Detonations on the seafloor would result in approximately 18,300 ft.2 (1,700 m2) of disturbed benthic
habitat per year in the Study Area (Table 4-7). Underwater explosions near the seafloor would primarily
occur in the nearshore portions of the Study Area (see Figure 2-3) at appropriate mine counter measure
training sites. Training activities that include bottom-laid underwater explosions are infrequent (only
about 50 explosions per year), and are likely to occur in the same general area. Additionally, the
designated mine counter measure training sites mainly consist of soft bottom substrates that are
expected to recover to their previous structure. The effects of training activities that use underwater
explosives on any hard substrate are determined to be permanent although individually minimal
throughout the Study Area, and in areas where habitat data are available (e.g., Apra Harbor). There are
no known hard substrates in the mine neutralization area.
4
Pounds per cubic inches of TNT (1.64 grams/cubic centimeters) x number of pounds, then solving for radius in the geometry of
a spherical volume
4-20
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 4-3: Mine Neutralization and Beach Landing Sites in Relation to Marine Vegetation
4-21
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 4-4: Mine Neutralization Sites and Beach Landing Sites in Relation to Coral
4-22
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 4-7: Bottom Detonations for Training and Testing Activities under Proposed Action
Net Explosive
1
Weight (lb.)
Impact
2
Footprint ft.
2
(m )
Number of
Charges
Total Impact
2
2
Area ft. (m )
20
366 (34)
20
7,320 (680)
20
366 (34)
30
10,980 (1,020)
-
-
50
18,300 (1,700)
Mine Countermeasure Mission
Package Testing Activities
5
145 (13)
24
3,480(310)
Total
-
-
24
3,480(310)
Activity
Training
Mine Neutralization (Explosive
Ordnance Disposal [EOD])
Underwater Demolition
Qualification/Certification
Total
Testing
1
Analysis assumes the largest charge, in terms of net explosive weight, for the training activity.
Notes: lb. = pounds, ft.2 = square feet, m2 = square meters
Training activities that include bottom-laid underwater explosions are infrequent (only about 50
explosions per year), and are likely to occur in the same general area, which are mainly soft bottom
habitats. The recovery for habitats in areas of repeated detonations would be expected to be prolonged.
The effects of training activities that use underwater explosives on any hard substrate are determined to
be permanent although individually minimal throughout the Study Area, and in areas where habitat data
are available (e.g., Apra Harbor). There are no known hard substrates in the mine neutralization area.
Therefore, the effects on soft bottom substrate are determined to be short term, individually and
cumulatively minimal.
Training activities using explosives that could potentially affect water column EFH and HAPC would be
conducted throughout the Study Area. The activity areas for training and testing activities are shown in
Figure 2-1, and the impact footprints presented in Table 4-8 represents the zone of greater than
10 percent mortality of shrimp or 30 lb. fish (refer to Section 4.1.1.2.1, Explosives, for details on
methods).
If all the explosives listed in Table 4-8 were detonated such that their mortality zone did not overlap
(very unlikely), the sum of potential temporary impacts per year on offshore water column EFH and
HAPC could be as much as 19,320 m3 impacted. This would be a small percentage of the total available
water column EFH available in the Study Area. Following the detonations, the water column EFH and
HAPC would be expected to return to its previous condition.
4-23
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 4-8: Explosions in the Water Column from Training Activities (Excluding Explosion on or near the Bottom),
and Their Impact on Water Column Essential Fish Habitat
Training
Explosive Category
2 1
Number of Explosions
Impact Footprint (m )
E1 (0.1 lb.–0.25 lb. NEW)
8,100
2,555
E2 (> 0.26 lb.–0.5 lb. NEW)
106
42
E3 (> 0.5 lb.–2.5 lb. NEW)
380
258
E4 (> 2.5 lb.–5 lb. NEW)
156
134
E5 (> 5 lb.–10 lb. NEW)
684
738
E6 (> 10 lb.–20 lb. NEW)
60
82
E7 (> 20 lb.–60 lb. NEW)
0
20
E8 (> 60 lb.–100 lb. NEW)
12
28
E9 (> 100 lb.–250 lb. NEW)
4
13
E10 (> 250 lb.–500 lb. NEW)
8
32
E11 (> 500 lb.–650 lb. NEW)
2
9
E12 (> 650 lb.–1,000 lb. NEW)
184
920
E13 (> 1,000 lb.–1,740 lb. NEW)
0
0
1
The impact footprint represents the zone of less than 10 percent mortality of shrimp or 30 lb. (14-kilogram)
fish; largest NEW of the explosives category was used in the calculations.
Notes: NEW = Net Explosive Weight, lb. = pound(s), m2 = square meter(s)
Given the small amount of water column habitat affected using a very unlikely worst case scenario,
and the quick recovery time, the effects of underwater explosives on water column EFH and HAPC is
determined to be temporary, and individually and cumulatively minimal throughout the Study Area.
Testing Activities
Under the Proposed Action, there would be 24 underwater detonations (explosive neutralizers) used
during mine countermeasure mission package testing activities. The maximum NEW of each detonation
would be 5 lb., which could impact an area of 145 ft.2 (13.5 m2). Underwater explosions associated with
testing activities would disturb approximately 3,480 ft.2 (323.3 m2) per year of substrate in the Study
Area.
Testing activities that include bottom-laid underwater explosions are infrequent (only about 24
explosions per year), and the percentage of area affected is small (less than 1 percent of the total Study
Area). Soft bottom substrates of disturbed areas would be expected to recover their previous structure,
with the fastest recovery occurring in areas with high waves and tidal energies. The recovery for habitats
in areas of repeated detonations would be expected to be slightly longer than those areas with high
waves and tidal energies. The effects of testing activities with underwater explosives on any hard
substrate are determined to be permanent although individually minimal throughout the Study Area,
and in areas where habitat data are available (e.g., Apra Harbor). Therefore, underwater explosions
would be limited to local and short-term impacts on benthic habitat in the Study Area.
4-24
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Testing activities that include bottom-laid underwater explosions are infrequent (only about 24
explosions per year), and the percentage of the Study Area affected is small (less than 1 percent of the
total Study Area). Additionally, detonations are likely to occur in the same general area, which would
further decrease the total area affected. The recovery for habitats in areas of repeated detonations
would be expected to be prolonged. The effects of testing activities with underwater explosives on any
hard substrate are determined to be permanent although individually minimal throughout the Study
Area). The effects on soft bottom substrate are determined to be short term, individually and
cumulatively minimal.
Testing activities using explosives that detonate at or near the surface could potentially affect water
column EFH and would be conducted throughout the Study Area. The activity areas for testing activities
are shown in Figure 2-1, and the impact footprints presented in Table 4-9 represents the zone of greater
than 10 percent mortality of shrimp or 30 lb. (14 kg) fish (refer to Section 4.1.1.2.1, Explosives, for
details on methods).
If all the munitions listed in Table 4-9 were detonated such that their zone of mortality did not overlap
(very unlikely), the sum of potential temporary impacts per year on offshore water column EFH and
HAPC, could be as much as 5,776 m3 impacted. This would be a small percentage of the total available
water column EFH available in the Study Area. Following the detonations, the water column EFH and
HAPC would be expected to return to its previous condition.
Given the small amount of water column habitat affected using a very unlikely worst case scenario, and
the quick recovery time, the testing effects of underwater explosives on water column EFH is
determined to be temporary, and minimal throughout the Study Area.
4-25
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 4-9: Explosions in the Water Column from Testing Activities (Excluding Explosion on or near the Bottom),
and Their Impact on Water Column Essential Fish Habitat
Testing
Munitions Category
Number of
Explosions
Impact
2 1
Footprint (m )
E1 (0.1 lb.–0.25 lb. NEW)
2,040
644
E2 (>0.26 lb.–0.5 lb. NEW)
0
0
E3 (>0.5 lb.–2.5 lb. NEW)
553
375
E4 (> 2.5 lb.–5 lb. NEW)
264
226
E5 (> 5 lb.–10 lb. NEW)
0
0
E6 (> 10 lb.–20 lb. NEW)
16
22
E7 (> 20 lb.–60 lb. NEW)
0
0
E8 (> 60 lb.–100 lb. NEW)
4
9
E9 (> 100 lb.–250 lb. NEW)
0
0
E10 (> 250 lb.–500 lb. NEW)
4
160
E11 (> 500 lb.–650 lb. NEW)
4
17
E12 (> 650 lb.–1,000 lb. NEW)
0
0
E13 (> 1,000 lb.–1,740 lb. NEW)
0
0
1
The impact footprint represents the zone of less than 10 percent mortality of shrimp
or 30 lb. (14-kilogram) fish, largest NEW of the explosives category was used in the
calculations.
Notes: NEW = Net Explosive Weight, m2 = square meter(s), lb. = pound(s)
4.1.1.2.2
Swimmer Defense Airguns
Swimmer defense airguns would be used for integrated swimmer defense testing at pierside locations.
Pierside integrated swimmer defense testing involves a limited number of impulses from a small airgun
in Inner Apra Harbor. Airguns would be fired a limited number of times during each activity at an
irregular interval as required for the testing objectives.
Underwater impulses would be generated using a small (60-cubic-inch) airgun, which is essentially a
stainless steel tube charged with high-pressure air via a compressor. An impulsive sound is generated
when the air is almost instantaneously released into the surrounding water, an effect similar to popping
a balloon in air. Generated impulses would have short durations, typically a few hundred milliseconds.
The root-mean-squared SPL and SPL at a distance 1 m from the airgun would be approximately 200–210
dB re 1 µPa and 185–195 dB re 1 µPa2-s.
Impulses from airguns lack the strong shock wave and rapid pressure increase, as would be expected
from explosive sources that can cause primary blast injury or barotraumas to fish and invertebrates.
There is little evidence that airguns can cause direct injury to adult fish, with the possible exception of
injuring small juvenile, larval fish, or other invertebrates nearby (approximately 16 ft. [4.9 m]).
Therefore, small juvenile, larval fish, or other invertebrates within a few meters of the airgun may be
injured or killed. In addition, fish that are able to detect the airgun impulses may exhibit alterations in
natural behavior.
4-26
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
It is unlikely that swimmer defense airguns would affect biogenic habitats as they would be used only in
Navy ports (Inner Apra Harbor), which do not support large areas of biogenic habitat. There would be no
anticipated impact from swimmer defense airguns on abiotic habitats, because the pressure wave
generated by the airguns would not be strong enough to cause disruptions.
Swimmer defense airguns are not expected to cause direct trauma to marine fish and invertebrates or
permanently affect water column EFH or HAPC. Abiotic substrate and associated seagrass or sedentary
invertebrate beds should be unaffected because the pressure wave generated by the swimmer defense
airgun testing would not be strong enough to disrupt abiotic substrates, would not be used in locations
near these habitats, and would only last for a very limited duration.
4.1.1.2.3
Weapons Firing, Launch, and Impact Noise
Noise associated with weapons firing and non-explosive impact could happen at any location within the
Study Area but generally would occur at locations greater than 12 nm from shore. Testing activities
involving weapons firing noise would be those events involved with testing weapons and launch
systems. These activities would also take place throughout the Study Area.
The firing of a weapon may have several components of associated noise. Firing of guns could have
acoustic effects from sound generated by firing the gun (muzzle blast), vibration from the blast
propagating through a ship’s hull, and sonic booms generated by the projectile flying through the air
(Table 4-10). Missiles and targets would produce noise during launch. In addition, impact of NEPM can
introduce sound into the water.
Table 4-10: Representative Weapons Noise Characteristics
Noise Source
Sound Level
In-Water
Naval Gunfire Muzzle Noise (5 in./54-caliber)
Approximately 200 dB re 1 µPa directly under
gun muzzle at 5 ft. below water surface
Airborne
Naval Gunfire Muzzle Noise (5 in./54-caliber)
178 dB re 20 µPa directly below the gun muzzle
above the water surface
Hellfire Missile Launch from Aircraft
149 dB re 20 µPa at 4.5 m
7.62 mm M-60 Machine Gun
90 dBA re 20 µPa at 50 ft.
0.50-caliber Machine Gun
98 dBA re 20 µPa at 50 ft.
Notes: dB = decibels; dBA = decibels, A-weighted; ft. = feet; µPa = micropascal; re = referenced to; mm =
millimeters; in. = inches; m = meters
4.1.1.2.4
Naval Gunfire Noise
Firing a ship deck gun produces a muzzle blast in air that propagates away from the muzzle in all
directions, including toward the water surface. Most sound enters the water in a narrow cone beneath
the sound source (within 13 degrees of vertical). In-water sound levels were measured during the
muzzle blast of a 5 in. (12.7 cm) deck-mounted gun, the largest caliber gun currently used in proposed
Navy activities. The highest sound level in the water (on average 200 dB re 1 µPa measured 5 ft. below
the surface) was obtained when the gun was fired at the lowest angle, placing the blast closest to the
water surface (U.S. Department of the Navy 2000; Yagla and Stiegler 2003). The average impulse at that
4-27
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
location was 19.6 Pa-s. The corresponding average peak in-air pressure was 178 dB re 20 µPa, measured
at the water surface below the firing point.
Gunfire also sends energy through the ship structure, into the water, and away from the ship. This effect
was investigated in conjunction with the measurement of 5 in. (12.7 cm) gun blasts described above.
The energy transmitted through the ship to the water for a typical round was about 6 percent of that
from the air blast impinging on the water. Therefore, sound transmitted from the gun through the hull
into the water is a minimal component of overall weapons firing noise.
The projectile shock wave in air by a shell in flight at supersonic speeds propagates in a cone (generally
about 65 degrees) behind the projectile in the direction of fire (Pater 1981). Measurements of a 5 in.
(12.7 cm) projectile shock wave ranged from 140 to 147 dB re 20 µPa taken at the surface at 0.59 nm
distance from the firing location and 10 degrees off the line of fire for safety (approximately 623 ft.
[190 m] from the shell’s trajectory). Sound level intensity decreases with increased distance from the
firing location and increased angle from the line of fire (Pater 1981). Like sound from the gun firing blast,
sound waves from a projectile in flight would enter the water primarily in a narrow cone beneath the
sound source. The region of underwater sound influence from a single traveling shell would be relatively
narrow, the duration of sound influence would be brief at any point, and sound level would diminish as
the shell gains altitude and loses speed. Multiple, rapid gun firings would occur from a single firing point
toward a target area. Vessels participating in gunfire activities would maintain enough forward motion
to maintain steerage, normally at speeds of a few knots. Acoustic impacts from weapons firing would
often be concentrated in space and duration.
Launch Noise
Missiles can be rocket- or jet-propelled. Sound due to missile and target launches is typically at a
maximum at initiation of the booster rocket. It rapidly fades as the missile or target reaches optimal
thrust conditions and the missile or target reaches a downrange distance where the booster burns out
and the sustainer engine continues. Launch noise level for the Hellfire missile, which is launched from
aircraft, is about 149 dB re 20 µPa at 14.8 ft. (4.5 m) (U.S. Department of the Army 1999).
Non-Explosive Impact Noise
Mines, non-explosive bombs, and intact missiles and targets could impact the water with great force
and produce a large impulse and loud noise. Sounds of this type are produced by the kinetic energy
transfer of the object with the target surface, and are highly localized to the area of disturbance.
McLennan (1997) calculated the sound from large targets (over 4,400 lb. [2,000 kg]) hitting the water at
speeds of over 3,280 ft. per second (1,000 m per second) to have source levels in water of
approximately 291 dB re 1 μPa re 1 m, although with very short pulse durations. However, the author
does caution that the model may be an oversimplification for several stated reasons, and that
measurements of actual levels may yield values 10–20 dB less than theoretical predictions. Sound
associated with the impact event is typically of low frequency (less than 250 Hz) and of short duration.
4-28
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
The effect of weapons firing, launch, and impact noise on water column EFH should be temporary and
minimal, for the same reasons small airgun fire associated with swimmer defense testing is not expected
to cause direct trauma to fish and invertebrates, or affect water column EFH and HAPC. Fish lacking a
swim bladder are even less likely to experience direct trauma from weapons firing, launch, and impact
noise. Abiotic substrate and associated seagrass or sedentary invertebrate beds should be unaffected
because the pressure wave generated by weapons firing, launch, and impact noise would not be strong
enough to disrupt abiotic substrates, would not be used in locations near these habitats, and would only
last for a very limited duration.
4.1.2
ENERGY STRESSORS
This section analyzes the potential impacts of energy stressors that can occur during training and testing
activities within the Study Area.
4.1.2.1
Electromagnetic Devices
The training activities that involve the use of magnetic influence mine neutralization systems include:
•
•
Mine Countermeasure – Towed Mine Detection
Maritime Homeland Defense/Security Mine Countermeasures
The testing activities that involve the use of magnetic influence mine neutralization systems include:
•
Mine Countermeasure Mission Package
The majority of devices involved in the activities described above include towed or unmanned MIW
systems that simply mimic the electromagnetic signature of a vessel passing through the water. None of
the devices include any type of electromagnetic “pulse.” An example of a representative device is the
Organic Airborne and Surface Influence Sweep. The Organic Airborne and Surface Influence Sweep is
towed from a forward flying helicopter and works by emitting an electromagnetic field and mechanically
generated underwater sound to simulate the presence of a ship. The sound and electromagnetic
signature cause nearby mines to detonate.
Generally, voltage used to power these systems is around 30 volts relative to seawater. This amount of
voltage is comparable to two automobile batteries. Since saltwater is an excellent conductor, only very
moderate voltages of 35 volts (capped at 55 volts) are required to generate the current. These small
levels represent no danger of electrocution in the marine environment, because the difference in
electric charge is very low in saltwater.
The static magnetic field generated by the electromagnetic devices is of relatively minute strength.
Typically, the maximum magnetic field generated would be approximately 23 gauss (G). This level of
electromagnetic density is very low compared to magnetic fields generated by other everyday items.
The magnetic field generated is between the levels of a refrigerator magnet (150–200 G) and a standard
household can opener (up to 4 G at 4 in. [10.2 cm]). The strength of the electromagnetic field decreases
quickly away from the cable. The magnetic field generated at a distance of 13.12 ft. (4 m) from the
source is comparable to the earth’s magnetic field, which is approximately 0.5 G. The strength of the
field at just under 26 ft. (8 m) is only 40 percent of the earth’s field, and only 10 percent at 79 ft. (24 m).
At a radius of 656 ft. (200 m) the magnetic field would be approximately 0.002 G (U.S. Department of
the Navy 2005).
4-29
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Potential Impacts to the Water Column
An electromagnetic charge could affect the water column habitat by impacting fish and invertebrates
inhabiting this area. A comprehensive review of information regarding the sensitivity of marine
organisms to electric and magnetic impulses, including fishes comprising the subclass elasmobranchii
(sharks, skates, and rays; hereafter referred to as elasmobranchs), as well as other bony fishes, is
presented in Normandeau (2011). The synthesis of available data and information contained in this
report suggests that while many fish species (particularly elasmobranchs) are sensitive to
electromagnetic fields, further investigation is necessary to understand the physiological response and
magnitude of the potential effects. Most examinations of electromagnetic fields on marine fishes have
focused on buried undersea cables associated with offshore wind farms in European waters (Boehlert
and Gill 2010; Gill 2005; Ohman et al. 2007).
Many fish groups including elasmobranchs, salmon, and others, have an acute sensitivity to electrical
fields, known as electroreception (Bullock et al. 1983; Helfman et al. 2009). Elasmobranchs are more
sensitive than the others. In elasmobranchs, behavioral and physiological response to electromagnetic
stimulus varies by species and age, and appears to be related to foraging behavior (Rigg et al. 2009).
Many elasmobranchs respond physiologically to electric fields of 10 nanovolts (nV) per cm and
behaviorally at 5 nV per cm (Collin and Whitehead 2004). Electroreceptive marine fishes identified
above with ampullary (pouch) organs can detect considerably higher frequencies of 50 Hz to more than
2 kHz (Helfman et al. 2009). The distribution of electroreceptors on the head of these fishes, especially
around the mouth (e.g., along the rostrum of sawfishes), suggests that these sensory organs may be
used in foraging. Additionally, some researchers hypothesize that the electroreceptors aid in social
communication (Collin and Whitehead 2004).
While elasmobranchs and other fishes can sense the level of the earth’s electromagnetic field, the
potential effects on fish resulting from changes in the strength or orientation of the background field are
not well understood. Electroreceptors are thought to aid in navigation, orientation, and migration of
sharks and rays (Kalmijn 2000). The exact mechanism is unknown and no magnetic sensory organ has
been discovered, but magnetite (a magnetic mineral) is incorporated into the tissues of these fishes
(Helfman et al. 2009). Some species of salmon and tuna been shown to respond to magnetic fields and
may also contain magnetite in their tissues (Helfman et al. 2009). When the electromagnetic field is
enhanced or altered, sensitive fishes may experience an interruption or disturbance in normal sensory
perception. Research on the electrosensitivity of sharks indicates that some species respond to electrical
impulses with an apparent avoidance reaction (Helfman et al. 2009; Kalmijn 2000). This avoidance
response has been exploited as a shark deterrent, to repel sharks from areas of overlap with human
activity (Marcotte and Lowe 2008).
Both laboratory and field studies confirm that elasmobranchs (and some teleost [bony] fishes) are
sensitive to electromagnetic fields, but the long-term impacts are not well-known. Electromagnetic
sensitivity in some marine fishes (e.g., salmonids) is already well-developed at early life stages (Ohman
et al. 2007), with sensitivities reported as low as 0.6 millivolt per cm in Atlantic salmon (Formicki et al.
2004); however, most of the limited research that has occurred focuses on adults. Some species appear
to be attracted to undersea cables, while others show avoidance (Ohman et al. 2007). Under controlled
laboratory conditions, the scalloped hammerhead (Sphyrna lewini) and sandbar shark (Carcharhinus
plumbeus) exhibited altered swimming and feeding behaviors in response to very weak electric fields
(less than 1 nV per cm) (Kajiura and Holland 2002). A field trial in the Florida Keys demonstrated that
southern stingray (Dasyatis americana) and nurse shark (Ginglymostoma cirratum) detected and
avoided a fixed magnetic field producing a flux of 950 G (O'Connell et al. 2010). The maximum
4-30
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
electromagnetic fields typically generated during Navy training and testing activities is approximately
23 G.
Little information exists regarding invertebrate susceptibility to electromagnetic fields. Most corals are
thought to use water temperature, day length, and tidal fluctuations as cues for spawning. Magnetic
fields are not known to control coral spawning release or larval settlement. Some arthropods (e.g., spiny
lobster and American lobster) can sense magnetic fields, and this is thought to assist the animal with
navigation and orientation (Lohmann et al. 1995; Normandeau et al. 2011). These animals travel
relatively long distances during their lives, and it is possible that magnetic field sensation exists for other
invertebrates that travel long distances. Marine invertebrates, including several commercially important
species and federally managed species, have the potential to use magnetic cues (Normandeau et al.
2011). Susceptibility experiments have focused on arthropods, but several mollusks and echinoderms
are also susceptible. However, because susceptibility is variable within taxonomic groups it is not
possible to make generalized predictions for groups of marine invertebrates. Sensitivity thresholds vary
by species ranging from 0.3 to 30 millitesla, and responses included non-lethal physiological and
behavioral changes (Normandeau et al. 2011). The primary use of magnetic cues seems to be navigation
and orientation. Human-introduced electromagnetic fields have the potential to disrupt these cues and
interfere with navigation, orientation, and migration. Because electromagnetic fields weaken
exponentially with distance from the source, large and sustained magnetic fields present greater
exposure risks than small and transient fields, even if the small field is many times stronger than the
earth’s magnetic field (Normandeau et al. 2011). Transient or moving electromagnetic fields may cause
temporary disturbance to susceptible organisms’ navigation and orientation.
The temporary behavioral effect of electromagnetic stressors on susceptible fish and invertebrates is
not expected to result in a population-level response. Therefore, the effect on water column EFH would
be temporary and minimal.
Potential Impacts to Benthic Substrates and Biogenic Habitats
Substrate is unaffected by electromagnetic devices due to lack of a physical disturbance component.
Beds of submerged rooted vegetation are unaffected because they lack a central nervous system
susceptible to electromagnetic stressors. Sedentary invertebrate beds and reefs should not be impacted
because their navigation and orientation is not important, though mobile larvae may be affected.
Therefore, for substrate and biogenic habitat EFH, there is no adverse impact expected from
electromagnetic stressors. Likewise, there are no adverse impacts expected on these habitats within
HAPCs.
There is no adverse impact expected from electromagnetic stressors on substrates and biogenic habitat,
and there are no adverse impacts expected on these habitats within HAPCs.
4.1.3
PHYSICAL DISTURBANCE AND STRIKE STRESSORS
This section analyzes the potential impacts of the various types of physical disturbance and strike
stressors resulting from the military conducting its training and testing activities within the Study Area.
The water column, benthic substrates (e.g., soft and hard bottom), and biogenic habitats (e.g., coral, live
bottom) designated as EFH are potentially subject to physical disturbance by vessels, in-water devices,
military expended materials, and seafloor devices associated with military training and testing.
4-31
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
This section describes the potential characteristics of physical disturbance and strike stressors from
military training and testing activities. It also describes the relative magnitude and location of these
activities to provide the basis for analysis of potential physical disturbance to designated EFH.
4.1.3.1
Vessels
Vessels are a part of nearly all training and testing exercises that occur in the Study Area. As such, Navy
vessels are frequently transiting throughout the Study Area and in and out of ports. Table 4-11 provides
a list of vessel types, as well as examples of each type, their typical length, and speed. The potential
impacts of these movements to designated EFH and HAPCs are outlined below.
Table 4-11: Representative Vessel Types, Lengths, and Speeds
Type
Example(s)
Length
Typical
Operating
Speed
Max Speed
> 300 m
10–15 knots
30+ knots
Aircraft Carrier
Aircraft Carrier (CVN)
Surface Combatant
Cruisers (CG), Destroyers (DDG), Frigates
(FFG), Littoral Combat Ships (LCS)
100–200 m
10–15 knots
30+ knots
Amphibious Warfare
Ship
Amphibious Assault Ship (LHA, LHD),
Amphibious Transport Dock (LPD), Dock
Landing Ship (LSD)
100–300 m
10–15 knots
20+ knots
Support Craft/Other
Amphibious Assault Vehicle (AAV);
Combat Rubber Raiding Craft (CRRC);
Landing Craft, Mechanized (LCM); Landing
Craft, Utility (LCU); Submarine Tenders
(AS); Yard Patrol Craft (YP)
5–45 m
Variable
20 knots
Support Craft/Other
– Specialized High
Speed
High Speed Ferry/Catamaran; Patrol
Coastal Ships (PC); Rigid Hull Inflatable
Boat (RHIB)
20–40 m
Variable
50+ knots
Submarines
Fleet Ballistic Missile Submarines (SSBN),
Attack Submarines (SSN), Guided Missile
Submarines (SSGN)
100–200 m
8–13 knots
20+ knots
Note: m = meters
Surface ships, propelled either by water jet pump or by propeller, and small craft would be used in the
Study Area. Boats in the Study Area may approach the shore or beach below the mean high tide line to
transport personnel or equipment to and from shore. Some activities involve vessels towing in-water
devices used in MIW activities, but these are operated in a manner to ensure they avoid contacting the
sea floor.
Some vessels, such as amphibious vehicles, might contact the seafloor substrate in the surf zone while
transitioning over the beach onto land (although this is intentionally avoided to preserve equipment).
Over the beach landings are possible for various training activities, such as Amphibious Assault,
Amphibious Raid, Noncombatant Evacuation Operation, Humanitarian Assistance/Disaster Relief,
Civilian Port Defense, Personnel Insertion/Extraction, Underwater Survey, Joint Expeditionary Exercise,
Marine Air Ground Task Force (Amphibious) – Battalion Exercise, and Special Purpose Marine Air Ground
Task Force Exercise. These events each have their individual requirements and platforms, and may use
various beaches and landings on Guam, Tinian, and Rota. Guam beach landings may include Dadi Beach,
Reserve Craft Beach, Toyland Beach, Sumay Channel/Cove, Clipper Channel, San Luis Beach, Gab Gab
Beach, and Haputo Beach. Tinian beaches and landings may include Unai Chulu, Unai Dankulo, Unai
4-32
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Babui, and Tinian Harbor. Figures 4-3 and 4-4 show the beach landing sites on Guam in relation to
vegetation and coral coverage, and Figures 4-5 and 4-6 show the Tinian beach landing locations in
relation to vegetation and coral coverage.
4-33
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 4-5: Tinian Amphibious Landing Beaches in Relation to Marine Vegetation
4-34
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Figure 4-6: Tinian Amphibious Landing Beaches in Relation to Coral
4-35
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Prior to any amphibious over-the-beach training activity conducted with larger amphibious vehicles such
as Landing Craft Air Cushions (LCACs) or Amphibious Assault Vehicles (AAVs) (e.g., Amphibious Assaults),
a hydrographic survey and a beach survey would be required. The surveys would be conducted to
identify and designate boat lanes and beach landing areas that are clear of coral, hard bottom substrate,
and obstructions. LCAC landing and departure activities would be scheduled at high tide. In addition,
LCACs would stay fully on cushion or hover when over shallow reef to avoid corals and hard bottom
substrate. This is a standard operating procedure for safe operation of LCACs. Over-the-beach
amphibious activity would only occur within designated areas based on the hydrographic and beach
surveys. Similarly, AAV activities would only be scheduled within designated boat lanes and beach
landing areas and would conduct their beach landings and departures at high tide one vehicle at a time
within their designated boat lane (Commander, Naval Forces Marianas [COMNAVMAR] Instruction
3500.4A). Based on the surveys, if the beach landing area and boat lane is clear, the activity could be
conducted, and crews would follow procedures to avoid obstructions to navigation, including coral
reefs; however, if there is any potential for impacts on corals or hard bottom substrate, the Navy will
coordinate with applicable resource agencies before conducting the activity. Hydrographic and beach
surveys would not be necessary for beach landings with small boats, such as rigid hull inflatable boats
(RHIBs).
Potential Impacts to the Water Column
As vessels transit through an area, the water column would be temporarily disturbed by the vessels’
movement. However, as the water would not be altered in any measurable or lasting manner, there
would be no adverse impact to the water column itself.
Amphibious vessels would approach the shore and could beach, which would disturb sediments and
increase turbidity. The impact of large, power-driven vessels on the substrate in the surf zone would be
minor because of the dispersed nature of the amphibious landings and the dynamic nature of sediments
in areas of surf and tidal energy.
Potential Impacts to Benthic Substrate
Vessel movements could affect soft bottom habitats during amphibious landings by increasing turbidity.
Ocean approaches would be expected to have minimal effects on soft bottom marine habitats because
of the nature of high-energy surf and shifting sands. The movement of sediment by wave energy would
fill in disturbed soft-bottom habitat similar to sediment recovery from a severe storm. Therefore, vessel
movements in the Study Area would be expected to have a minimal effect to soft bottom marine
habitats.
Physical disturbances and strikes of hard bottom substrates by vessels would cause damage to the
vessel and are avoided. Therefore, there would be no adverse impact to hard bottom substrates or
artificial structures as a result of vessel movements.
Potential Impacts to Biogenic Habitats
As with hard bottom substrates, physical disturbances and strikes of benthic biogenic habitats by vessels
would cause damage to the vessel and are avoided. Therefore, there would be no adverse impact to
benthic biogenic habitats as a result of vessel movements.
For both training and testing activities, vessel movements would have minimal effects on the water
column, soft or hard bottom substrates, or benthic biogenic habitats designated as EFH or HAPC.
4-36
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
4.1.3.2
In-Water Devices
In-water devices include towed devices and unmanned vehicles such as remotely operated vehicles,
unmanned surface vehicles, and unmanned undersea vehicles. These devices are self-propelled and
unmanned or towed through the water from a variety of platforms including helicopters, unmanned
underwater vehicles, or surface ships. In-water devices are generally smaller than most Navy vessels,
ranging from several inches to about 15 m. See Table 4-12 for a range of in-water devices used.
These devices can operate anywhere from the water surface to the benthic zone. Certain devices do not
have a realistic potential to strike living marine resources because they either move slowly through the
water column (e.g., most unmanned undersurface vehicles) or are closely monitored by observers
manning the towing platform (e.g., most towed devices).
Table 4-12: Representative Types, Sizes, and Speeds of In-Water Devices
Type
Example(s)
Length
Typical
Operating Speed
Towed
Device
Airborne Laser Mine Detection System (ALMDS); Airborne Mine
Neutralization System (AMNS); AQS Systems; Improved Surface
Tow Target (ISTT); Towed SONAR System; MK-103, MK-104 and
MK-105 Minesweeping Systems; OASIS, Orion, Shallow Water
Intermediate Search System, Towed Pinger Locator 30
< 10 m
10–40 knots
Unmanned
Surface
Vehicle
MK-33 SEPTAR Drone Boat, QST-35A Seaborne Powered Target,
Ship Deployable Seaborne Target (SDST), Small Waterplane Area
Twin Hull (SWATH), Unmanned Influence Sweep System (UISS)
< 15 m
Variable, up to
50+ knots
Unmanned
Undersea
Vehicle
Acoustic Mine Targeting System, AMNS, AN-ASQ Systems,
Archerfish Common Neutralizer, Crawlers, CURV 21, Deep Drone
8000, Deep Submergence Rescue Vehicle, Gliders, Expendable
Mobile ASW Training Targets (EMATTs), Light and Heavy Weight
Torpedoes, Magnum Remotely Operated Vehicle (ROV), Manned
Portables, MINIROVs (MK 30 ASW Targets, RMMV (Remote MultiMission Vehicle), Remote Minehunting System (RMS), Unmanned
Influence Sweep
< 15 m
1–15 knots
Notes: m = meter(s), OASIS = Organic Airborne and Surface Influence Sweep
Potential Impacts to the Water Column
As in-water devices pass through an area, the water column would be temporarily disturbed. However,
as the water would not be altered in any measurable or lasting manner, there would be no adverse
impact to the water column itself.
Potential Impacts to Benthic Substrate
Physical disturbances and strikes of benthic substrates by in-water devices would cause damage to the
in-water devices and are avoided. Therefore, there would be no adverse impact to benthic substrates as
a result of the use of in-water devices.
Potential Impacts to Biogenic Habitats
As with benthic substrates, physical disturbances and strikes of benthic biogenic habitats by in-water
devices would cause damage to the device and are avoided. Therefore, there would be no adverse
impact to benthic biogenic habitats as a result of the use of in-water devices.
For both training and testing activities, the use of in-water devices would have no effect on the water
column, soft or hard bottom substrates, or benthic biogenic habitats designated as EFH or HAPC.
4-37
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
4.1.3.3
Military Expended Materials
Many different types of military expended materials remain at sea following military training and testing
activities that occur throughout the Study Area, as described in Section 2.4 (Description of the Action
Area). Military expended materials include: (1) NEPM; (2) fragments from high explosive munitions; and
(3) expended materials other than ordnance, such as sonobuoys, ship hulks, expendable targets and
aircraft stores (fuel tanks, carriages, dispensers, racks, carriages or similar types of support systems on
aircraft which could be expended or recovered).
The potential for physical disturbance to habitats designated as EFH by expended materials from
military training and testing activities exists throughout the Study Area, although the types of military
expended materials vary by activity and region with some locations having greater concentration of
activity than others. Section 2.2.3 (Military Expended Materials) provides a description of expended
materials that are used in military training and testing activities.
Potential Impacts to the Water Column
As the military expended materials would either drift in the water column or pass quickly through the
water column as they sink to the seafloor without altering the water in any measurable or lasting
manner, there would be no adverse impact to the water column itself. Impacts associated with the
degradation of military expended materials and their effect on water quality are discussed in
contaminant stressors.
Potential Impacts to Benthic Substrate
Military expended materials have the potential to physically disturb marine substrates to the extent that
they impair the substrate’s ability to function as a habitat. These disturbances can result from several
sources including the physical impact of the expended material contacting the substrate, the covering of
the substrate by the expended material, or the alteration of the substrate from one type to another
(e.g., converting soft bottom substrate into hard bottom resulting from solid expended materials
overlying soft substrates).
The likelihood of military expended materials adversely impacting substrates and biogenic habitats as
they come into contact with the seafloor depends on several factors including the size, type, mass, and
speed of the material; water depth; the amount of material expended; the frequency of training or
testing; and the type of substrate or biogenic community. Most of the kinetic energy of the expended
material, however, is dissipated within the first few yards of the object entering the water causing it to
slow considerably by the time it reaches the seafloor. Because the damage caused by a strike is
proportional to the force of the strike, slower speeds may result in lesser impacts.
Due to the depth of water in which most training and testing events take place, a direct strike on hard
bottom is unlikely to occur with sufficient force to damage the substrate. Any potential damage would
be to a small portion of the structural habitat. The value of many of these substrates as habitat,
however, is not entirely dependent on the precise shape of the structure. An alteration in shape or
structure caused by military expended materials would not necessarily reduce the habitat value of hard
bottom. In softer substrates (e.g., sand, mud, silt, clay, and composites), the impact of the expended
material coming into contact with the seafloor, if large enough and striking with sufficient momentum,
may result in a depression and a localized redistribution of sediments as they are temporarily
resuspended into the water column. During military training and testing, countermeasures such as flares
and chaff are introduced into the marine environment. These types of military expended materials are
4-38
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
not expected to impact substrates as strike stressors given their smaller size and low velocity when
deployed compared to projectiles, bombs, and missiles.
Another potential physical disturbance military expended materials could have on substrates would be
to cover them or to alter the type of substrate and, therefore, its function as habitat. The majority of
military expended materials that settle on hard bottoms, while covering the substrate, would still serve
the same habitat function as the substrate it is covering by providing a hard surface on which organisms
can settle and attach (Figure 4-7 and Figure 4-8). Full colonization or fouling of the expended material
would occur over an approximately 18-month timeframe, depending on the area, as depicted in a study
done on artificial reefs using limestone deposits (Carter and Prekel 2008). An exception would be
expended materials like the parachutes utilized to deploy sonobuoys, lightweight torpedoes,
expendable mobile ASW training targets, and other devices from aircraft, that would not provide a hard
surface for colonization or fouling. In these cases, the hard bottom that is covered by the expended
material would not be physically damaged; however, if that portion of the hard bottom is covered by the
expended material, it would have its ability to function as a habitat for colonizing or encrusting
organisms impaired.
Most military expended materials that settle on soft bottom habitats, while not damaging the actual
substrate, would inhibit the substrate’s ability to function as a habitat by covering it with a hard surface.
This would effectively alter the substrate from a soft surface to a hard structure and, therefore, would
alter the ability of the substrate from one capable of supporting a soft bottom community to one that
would be more appropriate as habitat for organisms more commonly found associated with hard
bottom environments (Figure 4-9). Expended materials that settle in the shallower, more dynamic
environments of the continental shelf would likely be eventually covered over by sediments due to
currents and other coastal processes or encrusted by organisms. In the deeper waters of the continental
slope and beyond where currents do not play as large of a role, expended materials may remain
exposed on the surface of the substrate with minimal change for extended periods (Figure 4-9). Softer
expended materials, such as parachutes, would also not damage the sediments but would likely impair
its ability to function as a habitat to some degree. Impacts associated with the degradation of military
expended materials and its effect on sediment quality is discussed in contaminant stressors.
Potential Impacts to Biogenic Habitats
As with substrates, military expended materials have the potential to adversely impact the benthic
invertebrates and vegetation that compose the biogenic habitats (e.g., coral, sponges, macroalgae,
hydroids, amphipod tubes, bryozoans) coinciding with areas where training and testing events occur.
Due to their size and minimal weight, smaller items such as small-caliber projectiles may result in little to
no damage to biogenic habitats while larger, heavier items such as large-caliber projectiles, bombs, or
missiles may break or crush the sessile invertebrates (e.g., coral, sponges, etc.) which may occur where
military materials would be expended. Damage to these habitats would be primarily confined to the
area of impact. As observed in recent benthic surveys in the Jacksonville OPAREA, expended munitions
and other hard objects that land in areas of live/hard bottom serve as colonizing structures in much the
same way as the surrounding substrates (Figure 4-7 and Figure 4-8), so recovery of the area would be
expected over time.
Other types of military expended materials such as parachutes, associated with certain air-dropped
munitions and devices, may not adversely impact a habitat through its initial contact, but may
potentially cover and potentially smother the habitat over time instead. Unlike munitions and many
other solid expended materials, it is unlikely that benthic invertebrates would colonize materials such as
4-39
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
parachutes, potentially resulting in a loss of biogenic habitat in areas where parachutes settle for as long
as they remain in place and intact.
Estuarine and nearshore biogenic habitats such as seagrass, mangroves, and wetlands are unlikely to be
impacted by military expended materials due to their close proximity to shore, well away from most
areas of training and testing where military materials would be expended.
Figure 4-7: A MK-58 Smoke Float Observed in an Area Dominated by Coral Rubble on the Continental Slope
Note: Observed at approximately 191 fathoms (350 meters) in depth and 60 nautical miles east of Jacksonville,
Florida. Of note is the use of the smoke float as a colonizing substrate for a cluster of sea anemones (U.S.
Department of the Navy 2010).
Figure 4-8: An Unidentified, Non-Military Structure Observed on the Ridge System Running Parallel to the
Continental Shelf Break
4-40
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Note: Observed at approximately 44 fathoms (80 meters) in depth and 55 nautical miles east of Jacksonville,
Florida. Of note is that encrusting organisms and benthic invertebrates readily colonize the artificial structure to a
similar degree as the surrounding rock outcrop (U.S. Department of the Navy 2010).
Figure 4-9: (Left) A 76-Millimeter Cartridge Casing on Soft Bottom. (Right) A Blackbelly Rosefish (Helicolenus
dactylopterus) Using the Casing for Shelter When Disturbed
Note: The casing was observed in a sandy area on the continental slope approximately 232 fathoms (425 meters) in
depth and 70 nautical miles east of Jacksonville, Florida. The casing has not become covered by sediments due to
the depth and the relatively calm, current-free environment. When disturbed, the rosefish retreated inside the
casing for protection.
4.1.3.3.1
Training Activities
Military expended materials used as part of training activities occurring in the Study Area, as well as
outside of these areas, have the potential to adversely affect benthic and biogenic habitats designated
as EFH. In addition, designated HAPCs coinciding with areas of training activity may also be adversely
affected. The portions of the water column designated as EFH would be minimally impacted by military
expended materials from training events.
High-explosive military expended material would typically fragment into small pieces. Ordnance that
fails to function as designed and inert munitions would result in larger pieces of military expended
material settling to the seafloor. Once on the seafloor, military expended material would be buried by
sediments, corroded from exposure to the marine environment, or colonized by benthic organisms.
Because training activities involving military expended materials have the potential to impact substrates
designated as EFH within the areas where training is occurring, the Study Area was evaluated to
determine what the level of impact could be under the Proposed Action. In an attempt to quantify the
potential level of disturbance of military expended materials on bottom substrates within the Study
Area, an analysis of two worst case scenarios were developed. As a conservative measure for the
analyses, within each category of expended items (e.g., bombs, missiles, rockets, large-caliber
projectiles, etc.), the size of the largest item which would be expended was used to represent the sizes
of all items in the category. For example, the footprint of missiles used during training exercises range
from 1.6 to 37.4 ft.2 (0.15 to 3.5 m2), respectively. For the analyses, all missiles were assumed to be
equivalent to the largest in size, or 37.4 ft.2 (3.5 m2). In addition, it was also assumed that the impact of
the expended material on the seafloor is twice the size of its footprint. This assumption accounts for any
4-41
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
displacement of sediments at the time of impact as well as any subsequent movement of the item on
the seafloor due to currents or other forces. This should more accurately reflect the potential
disturbance to soft bottom habitats, but should overestimate disturbance to hard bottom habitats since
no displacement of the substrate would occur. In addition, items with casings (e.g., small, medium, and
large-caliber munitions; flares; sonobuoys; etc.) have their impact footprints doubled to account for
both the item and its casing. To be conservative, items and their casings were assumed to be the same
size.
During sinking exercises (SINKEXs), large amounts of military expended material and a vessel hulk would
be expended. Sinking exercises in the Study Area, however, would occur over 50 nm from shore, where
the substrate would be primarily clays and silts. Impacts of military materials expended over deep-water
would be negligible because the military would typically avoid hard-bottom sub-surface features (e.g.,
seamounts). Vessel hulks used during SINKEXs would alter the bottom substrate, converting soft bottom
habitat into an artificial, hard-bottom structure. The amount of area affected by vessel hulks would be a
fraction of the available training area, and the vessel hulk would be an anchoring point in the open
ocean where the predominant habitat is soft bottom.
Potential impacts to soft bottom habitats from military expended materials would range from
temporary to permanent, depending upon the nature of the environment in which the expended
material settled. In areas subject to dynamic coastal processes such as tidal influx or currents, the
military expended materials may be covered by sediments over time. In such cases, the temporal impact
of the military expended material on the environment would be temporary (recovery in days to weeks)
to short term (recovery in less than 3 years). However, were the military expended materials to settle on
soft bottom in areas rarely disturbed by currents or other forces, such as on many areas of the
continental slope, the items may persist on the bottom indefinitely. In such cases, the items would cover
the soft bottom with a hard structure (the military expended material itself), thus inhibiting the soft
bottom’s ability to function as a habitat within the direct vicinity of the item. In such instances, the
military expended material would function more as an artificial structure rather than as soft bottom
habitat (see Figure 4-9). This would result in a long term (recovery in more than 3 years but less than 20
years were the item to decompose or break down over time), or permanent (recovery in more than 20
years) impact to the habitat. The spatial extent of the impact would be minimal, limited to the footprint
of the individual military expended material. In cases where multiple military materials are expended in
the same area, the same habitat may be impacted numerous times during a given training activity and
the overall impact to the habitat would be cumulative of the footprints of all of the military expended
materials to settle on the habitat.
Potential impacts to hard bottom substrates would primarily be temporary to short term. The military
expended materials that settled on hard bottom would initially impair the substrate’s ability to function
as a habitat, but would ultimately serve the same function as the habitat they cover leading to only a
temporary or short-term impact (see Figure 4-7 and Figure 4-8). The exception would be items made of
soft material, such as parachutes, that would impair the substrate or structure’s ability to function as a
habitat for as long as it was present. The spatial extent of the impact would be the same as noted for
soft bottom substrates.
A total of 261,470 military items would be expended annually in the Study Area during training activities,
which would result in a total impact area of approximately 1,702,924 ft.2 (158,208 m2). The majority of
the impact area would be ship hulks expended during SINKEXs. With an impact area of 632,000 ft.2
(58,740 m2) for each vessel and up to two SINKEXs per year, ship hulks would account for about 75
4-42
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
percent (1,264,544 ft.2 [117,480 m2]) of the annual impact area for training activities under the
Proposed Action.
Military expended materials resulting from training activities would adversely affect soft bottom
substrates designated as EFH in areas where these activities occur. Based on the results of a worst case
scenario where all military expended materials settled in areas of soft bottom substrates (Table 4-13),
the effect would be minimal based on the small amount of available habitat affected. The duration of
the effect to the areas that were affected, however, would range from long term to permanent.
Military expended materials resulting from training activities would adversely affect hard bottom
substrates designated as EFH in areas where these activities occur. Based on the results of a worst case
scenario where all military expended materials settled in areas of hard bottom substrates (Table 4-13),
the effect would be minimal based on the small amount of available habitat affected. The duration of
the effect to the areas that were affected would range from temporary to short term.
Biogenic habitats may also be potentially impacted by military expended materials. While the least
common of the benthic habitat types and, therefore, the least likely to be impacted, benthic biogenic
habitats have concentrated distributions throughout the Study Area, particularly occurring along the
coastal portions of the Study Area. The primary types of biogenic habitats that may potentially be
impacted by military expended materials include coral and coral reefs, live bottom (e.g., areas with
sponges, bryozoans, hydroids, amphipod tubes), and attached macroalgae. Impacts to benthic biogenic
habitats would range from short term to permanent depending on the type of organisms impacted.
Most benthic organisms and macroalgae would recover from an impact over a short time period (less
than 3 years). Military expended material in the coastal portions of the Study Area would be limited to
small-caliber projectiles, flares, parachutes, and target fragments. These materials would be small, and
would typically be covered by sediment or colonized by benthic organisms. The small size of military
expended materials would not change the habitat structure. Therefore, military expended material from
training activities in the Study Area would have a minimal effect on biogenic habitats.
4-43
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 4-13: Annual Numbers and Impacts of Military Expended Materials Proposed for Use under the Proposed
Action
Size
2
(m )
Impact
Footprint
2
(m )
Bombs
1.5022
3.0044
Bombs (NEPM)
1.5022
Small caliber
0.0028
Medium caliber
Military Expended
Material
Training Activities
Testing Activities
2
2
Number
Impact (m )
Number
Impact (m )
212
636.93
0
0
3.0044
848
2,547.73
0
0
0.0056
86,140
482.34
2,000
11.2
0.0052
0.0104
8,250
85.8
2,040
21.21
Medium caliber (NEPM)
0.0052
0.0104
85,500
889.2
2,040
21.21
Large Caliber
0.0938
0.1876
1,300
243.88
3,920
735.4
Large Caliber (NEPM)
0.0938
0.1876
5,238
982.65
8,680
1,628.37
Missiles
3.4715
6.9430
113
784.5
20
138.86
Missiles (NEPM)
3.4715
6.9430
0
0
20
138.86
Rockets
0.0742
0.1484
114
16.92
0
0
Rockets (NEPM)
Chaff (cartridges) –
aircraft
0.0742
0.1484
0
0
0
0
0.0001
0.0002
25,840
5.17
600
0.12
Flares
Acoustic countermeasures
0.1133
0.2266
25,600
5,800.96
300
67.98
0.0289
0.0578
0
0
0
0
Expendable Targets
9
18
414
7,452
360
6,481.66
Ship hulk (SINKEX)
29,370
58,740
2
117,480
0
0
Torpedo/accessories
0.7
1.4
63
88.2
116
162.40
Sonobuoys
0.1134
0.2268
10,980
2,490.26
1,213
137.55
Explosive sonobuoys
0.0906
0.1812
11
1.99
793
143.69
0.84
1.68
10,845
18,219.6
1,727
2,901.36
23,829
12,588.21
Decelerators/Parachutes
Total
261,470
158,208
Notes: NEPM = Non-explosive Practice Munitions, SINKEX = Sinking Exercise, m2 = square meter(s)
Coral would take the longest to recover from any injury sustained as a result of military expended
materials, as it is slow growing and it often takes decades for a damaged reef to recover. Impacts to
coral would range from long term (recovery in more than 3 years but less than 20 years) to permanent
(recovery in more than 20 years), depending on the severity of the damage and the type of coral
impacted. Coral reefs occur within the Study Area (see Figures 3-11 to 3-20). Deep-water corals also
occur in the Study Area; however, given the limited spatial extent of deep-water coral within the Study
Area and the general location where activities occur, it is highly unlikely that military expended materials
would land in the vicinity of deep-water coral found within the Study Area.
4-44
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Military expended materials resulting from training activities would adversely affect live bottom
organisms (e.g., areas with sponges, bryozoans, hydroids, amphipod tubes) and macroalgae
designated as EFH in areas where these activities occur. However, due to the small amount of area
expended materials would impact and the low likelihood that military material would be expended on
biogenic habitat (based on the limited distribution), the effect to these habitats from military
expended materials would be minimal. For areas that would potentially be affected, the duration of
the impact would be short term.
Military expended materials resulting from training activities would adversely affect coral and coral
reefs designated as EFH in areas where these activities occur. Due to the size of the area in which
activities would occur and the limited distribution of biogenic habitats, the effect to these habitats
from military expended materials would be minimal. However, should an effect occur, the duration of
the effect would be long term to permanent.
4.1.3.3.2
Testing Activities
Military expended materials from testing activities occurring in the Study Area, as well as outside of
these areas, have the potential to adversely impact benthic and biogenic habitats designated as EFH. In
addition, designated HAPCs coinciding with areas of testing activities may also be adversely affected.
The portions of the water column designated as EFH would not be impacted by military expended
materials from testing events.
Using the same methodology as for training activities, testing activities were also analyzed to determine
the potential impacts of military expended materials on benthic substrates under a worst case scenario
of all military expended materials used during testing exercises within a given testing range settling to
the bottom. Based on the results, military expended materials resulting from testing activities would
impact less than 1 percent of the available seafloor within the Study Area annually, even under a worst
case scenario (Table 4-13). Those impacts that do occur would be the same as characterized in the
discussion in the previous section (see Section 4.1.3.3.1, Training Activities).
The potential impacts to biogenic habitats from military expended materials resulting from testing
activities would be the same as described for the training activities in Section 4.1.3.3.1 (Training
Activities).
4-45
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Military expended materials resulting from testing activities would adversely affect live bottom
organisms (e.g., areas with sponges, bryozoans, hydroids, amphipod tubes) and macroalgae
designated as EFH in areas where these activities occur. However, due to the small amount of area
expended materials would impact and the low likelihood that military material would be expended on
biogenic habitat (based on the limited distribution), the effect to these habitats from military
expended materials would likely be minimal. For areas that could potentially be affected, the duration
of the impact would be short term.
Military expended materials resulting from testing activities would adversely affect coral reefs
designated as EFH in areas where these activities occur. Due to the sizes of the Study Area in which
activities would occur and the limited distribution of biogenic habitats, the effect to these habitats
from military expended materials would likely be minimal. However, were an effect to occur, the
duration of the impact would be long term to permanent.
4.1.3.4
4.1.3.4.1
Seafloor Devices
Seafloor Devices
Seafloor devices represent any item used during training or testing activities that intentionally comes
into contact with the seafloor, but are later recovered. These items include moored mine shapes, and
anchors. Seafloor devices are stationary and do not pose a threat to highly mobile organisms.
Mine shapes are typically deployed via surface vessels or fixed-wing aircraft. Under the Proposed Action,
480 mine shapes would be used during mine laying training activities. Mine shapes would be used in
Warning Area 517, which is located over predominately soft bottom habitat in the offshore area.
Additional seafloor devices are utilized during pierside integrated swimmer defense activities and
testing activities at the North Pacific Acoustic Lab’s Deep Water site. The deep water experimental site
consists of an acoustic tomography array, a distributed vertical line array, and moorings in the deepwater environment (depths greater than 3,280 ft. [1,000 m]) of the northwestern Philippine Sea. These
locations would include seafloors consisting of soft bottom habitat of unconsolidated sediments. Most
moored mines deployed from surface vessels are typically secured with up to a 2,700 lb. (1,225 kg)
concrete mooring block (approximately 30 in. [76.2 cm] to a side). Moored mines deployed from
fixed-wing aircraft enter the water and impact the bottom, becoming semi-submerged. Upon impact,
the mine casing separates and the semi-buoyant mine floats through the water column until it reaches
the end of the mooring line. Bottom mines are typically positioned manually and are allowed to free sink
to the bottom to rest. Mine shapes are normally deployed over soft sediments and are recovered within
7–30 days following the completion of the training or testing events.
Additionally there would be 18 precision anchoring activities which would occur within predetermined
shallow water anchorage locations near ports. These locations would include seafloors consisting with
soft bottom habitat of unconsolidated sediments. The intent of these training exercises is to practice
anchoring the vessel within 100 yards (yd.) of the planned anchorage location. These training activities
typically occur within predetermined shallow water anchorage locations near ports with seafloors
consisting of soft bottom substrate.
Potential Impacts to the Water Column
The use of seafloor devices would not alter the water in any measurable or lasting manner. Therefore,
there would be no adverse impact to the water column itself.
4-46
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Potential Impacts to Benthic Substrate
As a result of their temporary nature, mine shapes would not permanently impact the substrate on
which they are placed. However, their presence would temporarily impair the ability of the substrate to
function as a habitat for as long as the mine shape is in place. As mine shapes are primarily deployed
over soft bottom substrates, hard bottom would not be impacted.
The level of impact to substrates from precision anchoring training exercises would depend on the size
of the anchor used, which would vary according to vessel type. Since these activities only take place in
pre-designated areas consisting of soft bottom substrates, areas of hard bottom would not be affected.
As most of these activities occur in areas subject to constant wave action and cycles of erosion and
deposition, disturbed areas would likely be reworked by waves and tides shortly after the disturbance.
The use of seafloor devices during training and testing activities could potentially have an adverse
effect on soft bottom substrates. These effects would be minimal in size and temporary (recovery in
days to weeks) in duration. Hard bottom substrates would not be affected by the use of seafloor
devices as they are generally avoided.
Potential Impacts to Biogenic Habitats
As mine shape deployment and precision anchoring exercises are typically done only in areas of soft
bottom substrates, areas of live/hard bottom and coral would not be impacted. In addition, as a result
of the distance from shore that these activities are conducted, submerged aquatic vegetation,
mangroves, marshes, shellfish beds, and wetlands would also not be impacted.
The use of seafloor devices during training and testing activities would adversely affect live bottom
organisms (e.g., areas with sponges, bryozoans, hydroids, amphipod tubes) and macroalgae
designated as EFH in areas where these activities occur. These activities could potentially have an
adverse effect on soft bottom substrates. These effects would be minimal in size and temporary
(recovery in days to weeks) in duration. Hard bottom substrates would not be affected by the use of
seafloor devices as they are generally avoided.
4.1.4
CONTAMINANT STRESSORS
This section considers the impacts on marine sediment and water quality from explosives, explosion
byproducts, and chemicals or substances other than explosives associated with military expended
materials (e.g., metals, chemicals, and other materials). This analysis focuses on changes in the
chemistry of substrate and water column that may adversely affect the quality of EFH for managed
species. The impacts on managed species via sediment or water that do not require trophic transfer
(e.g., bioaccumulation, predation) to be observed are considered here.
4.1.4.1
Explosives and Explosive Byproducts
High-order explosions consume most of the explosive material, creating typical combustion products. In
the case of Royal Demolition Explosive (RDX), 98 percent of the products are common seawater
constituents and the remainder is rapidly diluted below threshold effect level (U.S. Department of the
Navy 2008) (Table 4-14). Explosion byproducts associated with high-order detonations present no
stressors to fish and invertebrates through sediment or water chemistry. Low-order detonations and
unexploded ordnance present elevated likelihood of effects on fish or invertebrates. Deposition of
undetonated explosive materials into the marine environment can be reasonably well estimated by the
4-47
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
known failure and low-order detonation rates of high explosives (Table 4-15). Undetonated explosives
associated with ordnance disposal and mine clearance are collected after training is complete;
therefore, potential impacts are assumed to be inconsequential and not detectable for these training
and testing activities. The fish and invertebrates inhabiting EFH may be exposed by contact with the
explosive, contact with contaminants in the EFH, and ingestion of contaminated sediments.
Table 4-14: Byproducts from the Underwater Detonation of a High Blast Explosive
Predicted
Concentration (mg/L)
Permissible
Concentration (mg/L)
Aluminum oxide
0.4340
n/a
Carbon
0.1430
n/a
Carbon monoxide
0.0293
0.552
Ethane
0.0047
120
Carbon dioxide
0.0026
1.0
Ammonia
0.0023
0.092
Propane
0.0014
120
Hydrogen cyanide
0.0003
0.001
Methane
0.0001
120
< 0.0001
─
Byproduct
Other compounds*
* Other compounds include methyl alcohol, formaldehyde, acetylene, and phosphine.
Predicted concentrations were well below permissible concentrations.
Notes: “<” means “less than,” mg/L= milligram(s) per liter, n/a = not applicable
Table 4-15: Failure Rates and Low-Order Detonation Rates of Military Ordnance
Failure Rate (%)
Low-Order Detonation
Rate (%)
Guns/artillery
4.68
0.16
Hand grenades
1.78
─
High explosive ordnance
3.37
0.09
Rockets
3.84
─
Submunitions*
8.23
─
Ordnance
* Submunitions are munitions contained within and distributed by another device such as a rocket.
Table 4-16 provides a list of ordnance constituents remaining after low-order detonations and with
unconsumed explosives. These constituents are in addition to the high explosives contained in the
ordnance. Lead azide, titanium compounds, perchlorates, barium chromate, and fulminate of mercury
are not natural constituents of seawater. Lead oxide is a rare, naturally occurring mineral. It is one of
several lead compounds that form films on lead objects in the marine environment (Agency for Toxic
Substances and Disease Registry 2007). Metals are discussed in Section 4.1.4.2.
4-48
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 4-16: Constituents Remaining After Low-Order Detonations and from Unconsumed Explosives
Ordnance Component
Constituent
Pyrotechnics
Tracers
Spotting Charges
Barium chromate (BaCrO4)
Potassium perchlorate
Chlorides
Phosphorus
Titanium compounds
Oxidizers
Lead (II) oxide (PbO)
Delay Elements
BaCrO4
Potassium perchlorate
Lead chromate
Fuses
Potassium perchlorate
Detonators
Fulminate of mercury [Hg(CNO)2]
Potassium perchlorate
Primers
Lead azide [Pb(N3)2]
Indirect impacts of explosives byproducts and unexploded ordnance on fish and invertebrates via
sediment is possible in the immediate vicinity of the ordnance. Degradation products of RDX are not
toxic to marine organisms at realistic exposure levels (Rosen and Lotufo 2010). TNT and its degradation
products impact developmental processes in fish and invertebrates and are acutely toxic to adults at
concentrations similar to real-world exposures (Rosen and Lotufo 2007a, b; 2010). Relatively low
solubility of most explosives and their degradation products means that concentrations of these
contaminants in the marine environment are relatively low and readily diluted. Furthermore, while
explosives and their degradation products were detectable in marine sediment approximately 6–12 in.
(15–30 cm) away from degrading ordnance, the concentrations of these compounds were not
statistically distinguishable from background beyond 3 and 6 ft. (1 and 2 m) from the degrading
ordnance. Most explosives and explosive degradation products have very low solubility in sea water.
This means that dissolution occurs extremely slowly, and harmful concentrations of explosives and
degradation are not likely to accumulate except within confined spaces. Additionally, a low
concentration of contaminants, slowly delivered into the water column, is readily diluted to non-harmful
concentrations. Filter feeders (such as sessile invertebrates) in the immediate vicinity of degrading
explosives may be more susceptible to bioaccumulation of contaminants. While invertebrates may be
adversely impacted by the indirect effects of degrading explosives via water (Rosen and Lotufo 2007a,
2010), this is extremely unlikely in realistic scenarios.
Taken together, fish or invertebrates may be affected by the degrading explosives within a very small
radius of the explosive 1–6 ft. (0.3–2 m). The area of substrate impacted is small and the explosive
byproduct is minimal (would easily dissipate into the water column); therefore, the effects of explosives
and explosive byproducts used are short-term and minimal on water column and substrate EFH.
4.1.4.2
Metals
Certain metals and metal-containing compounds are harmful to fish and invertebrates at concentrations
above background levels (e.g., cadmium, chromium, lead, mercury, zinc, copper, manganese, and many
others) (Negri et al. 2002; Wang and Rainbow 2008). Metals are introduced into seawater and
sediments as a result of training and testing activities involving vessel hulks, targets, ordnance,
munitions, and other military expended material including batteries. In most instances, because of the
4-49
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
physical and chemical reactions that occur with metals in marine systems (e.g., precipitation), metals
often concentrate in sediments. Thus, metal contaminants in sediments are more of an issue than
metals in the water column. Many metals bioaccumulate and some physiological impacts begin to occur
only after several trophic transfers concentrate the toxic metals. Impacts of metals on fish and
invertebrates via sediment and water involve concentrations several orders of magnitude lower than
concentrations achieved via bioaccumulation. Fish and invertebrates may be exposed by contact with
the metal, contact with contaminants in the sediment or water, and ingestion of contaminated material.
Despite the acute toxicity of some metals (e.g., hexavalent chromium or tributyltin) (Negri et al. 2002),
concentrations above safe limits are scarcely encountered even in live fire areas of the former Navy
training range off Vieques, Puerto Rico, where deposition of metals from Navy activities is very high (Pait
et al. 2010). Other studies find no harmful concentrations of metals associated with deposition of
military metals into the marine environment (Buchman 2008). It is conceivable that fish or invertebrate
eggs or larvae could be impacted by metals via sediment within a few inches of the object.
No adverse effect on EFH from metals is anticipated based on studies comparing metal contamination
levels and levels considered safe. It is unlikely that fish or invertebrates will be adversely affected by the
physiological effects of metals.
4.1.4.3
Chemicals
Several Navy training and testing activities introduce potentially harmful chemicals into the marine
environment, principally ship hulks (SINKEXs), flares and propellants for rockets, missiles, and torpedoes.
In the past, polychlorinated biphenyls (PCBs) have been raised as a chemical pollutant issue because
they have been found in certain solid materials on vessel hulks used as targets during vessel-SINKEXs
(e.g., insulation, wires, felts, and rubber gaskets). Currently, vessels used for SINKEXs are selected from a
list of Navy approved vessels that have been cleaned in accordance with U.S. Environmental Protection
Agency guidelines. By rule, a SINKEX must be conducted at least 50 nm offshore and in water at least
6,000 ft. (1,830 m) deep (40 C.F.R. 229.2). The U.S. Environmental Protection Agency estimates that as
much as 100 lb. (45 kg) of PCBs remain onboard sunken vessels. The agency considers the contaminant
levels released during the sinking of a target to be within the standards of the Marine Protection,
Research and Sanctuaries Act (16 U.S.C. 1341, et seq.) (U.S. Environmental Protection Agency 1999).
Based on the foregoing considerations, PCBs will not be considered further.
Properly functioning flares, missiles, rockets, and torpedoes combust most of their propellants, leaving
benign or readily diluted soluble combustion byproducts (e.g., hydrogen cyanide). Operational failures
allow release of propellants and their degradation products into the marine environment. The greatest
risk to fish and invertebrates from flares, missile, and rocket propellants is perchlorate, which is highly
soluble in water, persistent, and impacts metabolic processes in many plants and animals. Perchlorate
contamination rapidly disperses throughout the water column and water within sediments. While it
impacts terrestrial biological processes at low concentrations (e.g., less than 10 parts per billion), toxic
concentrations are unlikely to be encountered in seawater. The principal mode of perchlorate toxicity in
the environment is bioaccumulation.
In contrast to perchlorate, the principal toxic components of torpedo fuel—propylene glycol dinitrate
and nitrodiphenylamine—adsorb to sediments, have relatively low toxicity, and are readily degraded by
biological processes. The MK-48 torpedo weighs roughly 3,700 lb. (1,680 kg) and uses Otto Fuel II as a
liquid propellant. Otto Fuel II is composed of propylene glycol dinitrate and nitro-diphenylamine
4-50
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
(76 percent), dibutyl sebacate (23 percent) and 2-nitrodiphenylamine as a stabilizer (2 percent).
Combustion byproducts of Otto Fuel II include nitrous oxides, carbon monoxide, carbon dioxide,
hydrogen, nitrogen, methane, ammonia, and hydrogen cyanide. During normal venting of excess
pressure or upon failure of the torpedo's buoyancy bag, the following are discharged: carbon dioxide,
water, hydrogen, nitrogen, carbon monoxide, methane, ammonia, hydrochloric acid, hydrogen cyanide,
formaldehyde, potassium chloride, ferrous oxide, potassium hydroxide, and potassium carbonate (U.S.
Department of the Navy 1996a, b).
It is conceivable that marine fish and invertebrate eggs, or larvae could be impacted by propellants via
sediment in the immediate vicinity of the object (e.g., within a few inches), but these potential impacts
would diminish rapidly as the propellant degrades. Fish and invertebrates may be exposed by contact
with the chemicals, contact with chemical contaminants in the sediment or water, and ingestion of
contaminated material.
No adverse effect on EFH from the chemicals discussed above is anticipated based on the rapid dispersal
and degradation of the chemicals. It is unlikely that fish or invertebrates will be adversely affected by
the physiological effects of chemicals other than explosives and explosive byproducts.
4.1.4.4
Other Materials
All military expended material, including targets and vessel hulks involved in SINKEXs contains materials
other than metals, explosives, or chemicals. Principal components of these military expended materials
include aluminized fiberglass (chaff), carbon or Kevlar fiber (missiles), and plastics (canisters, targets,
sonobuoy components, parachutes). Chaff has been extensively studied, and no indirect toxic effects are
known at realistic concentrations in the marine environment (Arfsten et al. 2002). Glass, carbon, and
Kevlar fibers are not known to have potential toxic effects on marine invertebrates. Plastics contain
chemicals that have potential effects on fish and invertebrates (Derraik 2002; Mato et al. 2001; Teuten
et al. 2007).
Potentially harmful chemicals in plastics are not readily adsorbed to marine sediments; instead, fish and
invertebrates are most at risk via ingestion or bioaccumulation. Because plastics retain many of their
chemical properties as they physically degrade into plastic particles (Singh and Sharma 2008), the
exposure risks to marine fish and invertebrates are dispersed over time. Additionally, plastic waste in
the ocean chemically attracts hydrocarbon pollutants such as PCB and dichlorodiphenyltrichloroethane,
which accumulate up to 1 million times more in plastic than in ocean water (Takada et al. 2001). It is
conceivable that marine fish and invertebrates could be indirectly impacted by chemicals associated
with plastics, but, absent bioaccumulation, these effects would be limited to direct contact with the
material.
Marine invertebrates and fish may be exposed by contact with the plastic, contact with associated
plastic chemical contaminants in the sediment or water, and ingestion of contaminated material.
No adverse effect on EFH from these materials (e.g., plastics) is anticipated because they are not readily
adsorbed to marine sediments and direct contact is required for harmful effects to organisms. It is
unlikely that fish or invertebrates will be adversely impacted by the physiological effects of other
materials.
4-51
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
4.1.5
STUDY AREA COMBINED IMPACT OF STRESSORS
Of all the potential stressors, only explosives on or near the bottom and military expended materials
have the potential to adversely impact marine habitats as a substrate for biological communities. The
impact area for underwater explosions and military expended materials were all much less than
1 percent of the total area of documented soft bottom or hard bottom in the Study Area. The
percentages are even lower for substrate impacts in the Study Area as a whole (Table 4-17). Even
multiplying by 5 years, the impacts are all less than 1 percent of the benthic substrate with very unlikely
worst case scenarios.
Table 4-17: Combined Impact on Marine Substrates from the Proposed Action
2
Impact Footprint (m )
Underwater Explosions
Military Expended
Materials
Total
156,745
158,769
2,024
Note: m2 = square meters
Chapter 5 (Mitigation Measures) describes standard operating procedures (SOPs) and mitigation
measures proposed to help reduce the potential impacts of explosives on or near the bottom and
military expended materials on marine substrates and associated biogenic habitats.
4-52
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
5 MITIGATION MEASURES
This section describes the Navy’s SOPs and mitigation measures. Many of these measures also help
reduce or avoid potential impacts to EFH or HAPCs.
5.1
STANDARD OPERATING PROCEDURES
The Navy currently employs standard practices to provide for the safety of personnel and equipment,
including ships and aircraft, as well as the success of the training and testing activities. For the purpose
of this document, we will refer to standard practices as SOPs. Because of their importance for
maintaining safety and mission success, SOPs have been considered as part of the Proposed Action. The
only SOPs specifically designed to reduce or avoid EFH are for towed in-water devices and amphibious
landings. Prior to deploying a towed device, there is a standard operating procedure to search the
intended path of the device for any floating debris (e.g., driftwood) or other potential obstructions (e.g.,
animals), since they have the potential to cause damage to the device. Prior to any amphibious over-thebeach training activity conducted with larger amphibious vehicles such as LCACs or AAVs (e.g.,
Amphibious Assaults), a hydrographic survey and a beach survey would be required. The surveys would
be conducted to identify and designate boat lanes and beach landing areas that are clear of coral, hard
bottom substrate, and obstructions. LCAC landing and departure activities would be scheduled at high
tide. In addition, LCACs would stay fully on cushion or hover when over shallow reef to avoid corals and
hard bottom substrate. Over-the-beach amphibious activity would only occur within designated areas
based on the hydrographic and beach surveys. Similarly, AAV activities would only be scheduled within
designated boat lanes and beach landing areas and would conduct their beach landings and departures
at high tide one vehicle at a time within their designated boat lane (COMNAVMAR Instruction 3500.4A).
Based on the surveys, if the beach landing area and boat lane is clear, the activity could be conducted,
and crews would follow procedures to avoid obstructions to navigation, including coral reefs; however,
if there is any potential for impacts on corals or hard bottom substrate, the Navy will coordinate with
applicable resource agencies before conducting the activity. Hydrographic and beach surveys would not
be necessary for beach landings with small boats, such as RHIBs.
5.2
MITIGATION MEASURES
The Navy recognizes that the Proposed Action has the potential to impact EFH or HAPCs. Unlike SOPs,
which are established for reasons other than environmental benefit, mitigation measures are
modifications to the Proposed Action that are implemented for the sole purpose of reducing a specific
potential environmental impact on a particular resource. The procedures discussed in this chapter, most
of which are currently or were previously implemented as a result of formal or informal consultations
with regulatory agencies during the MIRC EIS/OEIS and MITT EIS/OEIS process.
The mitigation measures fall under two categories: Lookouts and mitigation zones. The Lookouts on
Navy vessels are trained to identify marine mammals, sea turtles, and floating macroalgae and to avoid
physical impacts where possible; target areas should be clear of marine species. Mitigation zones are
buffer areas between potential impacts and observed marine life on the surface or mapped on the
bottom. The mitigation measures presented below (Table 5-1) were developed for training and testing
activities in the MITT Study Area. While these mitigation measures were implemented as a result of
potential impacts to marine mammals and sea turtles, they may indirectly benefit EFH and HAPCs. Those
that have designated stand offs from benthic habitats will have a direct positive impact on EFH and
HAPCs.
5-1
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 5-1: Summary of Recommended Mitigation Measures
Activity Category or
Mitigation Area
Recommended
Lookout Procedural
Measure
Recommended Mitigation
Zone and Protection Focus
Indirect or Direct
Beneficial Effects on EFH
Acoustic (Non-Impulsive Stressors)
Low-Frequency and HullMounted Mid-Frequency
Active Sonar during AntiSubmarine Warfare and
Mine Warfare
Activity Category or
Mitigation Area
2 Lookouts (general)
1 Lookout (minimally
manned, moored, or
anchored)
Recommended
Lookout Procedural
Measure
Low-Frequency: 200 yd.
(183 m) shutdown for marine
mammals and sea turtles
Hull-Mounted Mid-Frequency:
1,000 yd. (914 m) and 500
yd. (457 m) power downs
and 200 yd. (183 m)
shutdown for marine
mammals and sea turtles.
Indirect
Recommended Mitigation
Zone and Protection Focus
Indirect or Direct
Beneficial Effects on EFH
Acoustic (Explosive/Impulsive Stressors)
Improved Extended Echo
Ranging Sonobuoys
1 Lookout
600 yd. (549 m) for marine
mammals and sea turtles.
Indirect
Explosive Sonobuoys
using 0.6–2.5 lb. NEW
1 Lookout
350 yd. (320 m) for marine
mammals and sea turtles.
Indirect
Anti-Swimmer Grenades
1 Lookout
200 yd. (183 m) for marine
mammals and sea turtles.
Indirect
Mine Countermeasures
and Mine Neutralization
using Positive Control
Firing Devices
General: 1 or 2
Lookouts (NEW
dependent)
NEW dependent for marine
mammals and sea turtles and
flocks of seabirds.
Indirect
Up to 10 min. time-delay
using up to 29 lb. NEW:
1,000 yd. (915 m) for marine
mammals and sea turtles.
Indirect
200 yd. (183 m) for marine
mammals and sea turtles.
Indirect
Mine Neutralization
Activities Using DiverPlaced Time-Delay Firing
Devices
Gunnery Exercises –
Small- and MediumCaliber using a Surface
Target
Diver-placed: 2
Lookouts
Lookouts will survey
the mitigation zone for
seabirds prior to and
after the detonation
event.
4 Lookouts
Lookouts will survey
the mitigation zone for
seabirds prior to and
after the detonation
event.
1 Lookout
Gunnery Exercises –
Large-Caliber using a
Surface Target
600 yd. (549 m) for marine
mammals and sea turtles.
1 Lookout
70 yd. (64 m) within 30
degrees on either side of the
gun target line on the firing
side for marine mammals and
sea turtles.
5-2
Indirect
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 5-1: Summary of Recommended Mitigation Measures (continued)
Activity Category or
Mitigation Area
Recommended
Lookout Procedural
Measure
Missile Exercises
(Including Rockets) up to
250 lb. NEW using a
Surface Target
Recommended Mitigation
Zone and Protection Focus
Indirect or Direct
Beneficial Effects on EFH
900 yd. (823 m) for marine
mammals and sea turtles.
1 Lookout
Direct
350 yd. (320 m) for surveyed
shallow coral reefs.
Missile Exercises
(Including Rockets) from
251 to 500 lb. NEW using
a Surface Target
2,000 yd. (1.8 km) for marine
mammals and sea turtles.
1 Lookout
Direct
350 yd. (320 m) for surveyed
shallow coral reefs.
Explosive: 2,500 yd. (2.3 km)
for marine mammals and sea
turtles.
Bombing Exercises,
Explosive and NonExplosive
1 Lookout
Non-Explosive: 1,000 yd.
(914 m) for marine mammals
and sea turtles.
Direct
Both: 350 yd. (320 m) for
surveyed shallow coral reefs.
Torpedo (Explosive)
Testing
1 Lookout
2,100 yd. (1.9 km) for marine
mammals and sea turtles
and jellyfish aggregations.
Indirect
2 Lookouts
2.5 nm for marine mammals
and sea turtles and jellyfish
aggregations.
Indirect
1 Lookout
1,600 yd. (1.4 km) for marine
mammals and sea turtles.
Indirect
Sinking Exercises
At-Sea Explosive Testing
Physical Strike and Disturbance
500 yd. (457 m) for whales.
Vessel Movements
1 Lookout
Towed In-Water Device
Use
200 yd. (183 m) for all other
marine mammals (except
bow riding dolphins).
Indirect
1 Lookout
250 yd. (229 m) for marine
mammals
Indirect
No Lookouts in
addition to standard
personnel standing
watch
Avoidance of precision
anchoring within the anchor
swing diameter of shallow
coral reefs, live hardbottom,
artificial reefs, and
shipwrecks.
Direct
Precision Anchoring
5-3
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 5-1: Summary of Recommended Mitigation Measures (continued)
Activity Category or
Mitigation Area
Recommended
Lookout Procedural
Measure
Recommended Mitigation
Zone and Protection
Focus
Indirect or Direct
Beneficial Effects on
EFH
No Lookouts in addition
to standard personnel
standing watch
The Navy will not conduct
precision anchoring within
the anchor swing diameter,
or explosive mine
countermeasure and
neutralization activities
(except in existing
anchorages and near-shore
training areas around Guam
and within Apra Harbor)
within 350 yd. (320 m) of
surveyed shallow coral
reefs, live hardbottom,
artificial reefs, and
shipwrecks.
No explosive or nonexplosive small-, medium-,
and large-caliber gunnery
exercises using a surface
target, explosive or nonexplosive missile exercises
using a surface target,
explosive and non-explosive
bombing exercises, or at-sea
explosive testing within
350 yd. (320 m) of surveyed
shallow coral reefs
Direct
Shallow Coral Reefs,
Hardbottom Habitat,
Artificial Reefs, and
Shipwrecks
Notes: EFH = Essential Fish Habitat, NEW = Net Explosive Weight, lb. = pounds, yd. = yards, m = meters, km = kilometers
The mitigation zones for seafloor habitats and shipwrecks address precision anchoring, explosive mine
countermeasures and neutralization activities, and other activities involving explosive or non-explosive
munitions. The Navy will not conduct the following activities within 350 yd. (320 m) of known surveyed
shallow coral reefs, live hard bottom, artificial reefs, and shipwrecks: explosive or non-explosive small,
medium, and large caliber GUNEXs using a surface target; explosive missile exercises using a surface
target; explosive and non-explosive bombing exercises; or at-sea explosives testing. The Navy will not
conduct precision anchoring within the anchor watch circle diameter, or explosive mine countermeasure
and neutralization activities near known surveyed shallow coral reefs, live hardbottom, artificial reefs,
and shipwrecks. To facilitate these protective measures, the Navy will include maps of known shallow
coral reefs, artificial reefs, shipwrecks, and live hard bottom during planning of training and testing
events.
The Navy’s currently implemented seafloor habitats and shipwreck mitigation zones are based off the
range to effects for marine mammals or sea turtles, which are driven by hearing thresholds. The
recommended measures are modified to focus on reducing potential physical impacts to seafloor
habitats and shipwrecks from explosives, and physical strike from military expended materials. The
recommended 350 yd. (320 m) mitigation zone is based off the estimated maximum crater impact for
explosions discussed in Section 4.1.1.2.1 (Explosives). The use of non-explosive military expended
materials would result in a smaller footprint of potential impact; however, the Navy recommends
applying the explosive mitigation zone to all explosive and non-explosive activities as listed above for
5-4
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
ease of implementation. This standard mitigation zone will consequently result in an additional
protection buffer during the non-explosive activities listed above. Avoiding or minimizing physical
disturbance and strike of these resources will likely reduce the impact on these resources.
The Navy proposes implementing the recommended measures described above because: (1) they are
likely to result in avoidance or reduction of physical disturbance and strike to sensitive habitats and
shipwrecks; and (2) they have acceptable operational impacts to the proposed activity with regard to
safety, practicability, impact to readiness, and Navy policy.
5-5
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
This Page Intentionally Left Blank
5-6
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
6 CONCLUSIONS
The potential impacts from the Proposed Action on EFH and HAPC among the Fishery Management
Council region did not exceed a determination of minimal. Refer to relevant sections for supporting
details for each conclusion. The individual stressor impacts were all either no effect or minimal and
ranged in duration from temporary to permanent, depending on the habitat impacted (Table 6-1).
Table 6-1: Potential Impacts on Essential Fish Habitat from Each Stressor
Stressors
Water Column
Acoustic stressors (Section 4.1.1)
Non-impulsive
Minimal and temporary
• Sonar
• Vessel noise
Explosive and other
Minimal and temporary
impulsive
• Underwater
explosions
• Swimmer
defense airguns
• Weapons firing,
launch, and
impact noise
Substrate
Biogenic
No effect
No effect
Minimal and short term (soft
bottom) to permanent (hard
bottom); mitigation avoids
mapped hard bottom.
• Attached macroalgae:
minimal and long term based
on hard substrate impacts
• Submerged rooted
vegetation: minimal and longterm
• Sedentary invertebrate beds:
minimal and short term to
permanent (based on
substrate impacts); mitigation
avoids mapped hard bottom
• Reefs: minimal and long term;
mitigation avoids coral reefs
Energy stressors (Section 4.1.2)
Electromagnetic
Less than minimal and
No effect
devices
temporary
Physical disturbance and strike stressors (Section 4.1.3)
Vessel movement
No effect
No effect
In-water devices
No effect
No effect
6-1
No effect
No effect
Biogenic habitats: no effect;
mitigation avoids sensitive
nearshore habitats
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table 6-1: Potential Impacts on Essential Fish Habitat and Habitat Areas of Particular Concern from Each Stressor
(continued)
Stressors
Water Column
Substrate
Physical disturbance and strike stressors (Section 4.1.3) (continued)
Military expended
Minimal and temporary
Minimal and long term to
materials
permanent
Seafloor devices
No effect
Minimal and temporary (soft
bottom)
No effect (hard bottom)
Contaminant stressors (Section 4.1.4)
Explosives and
Minimal and short term
explosive byproducts
Minimal and short term
Metals
Chemicals
Other materials
No effect
No effect
No effect
No effect
No effect
No effect
Biogenic
• Attached macroalgae: Minimal
and short term;
• Submerged rooted vegetation;
minimal and short term;
• Sedentary invertebrate beds:
minimal and long term to
permanent (based on
substrate impacts)
• Reefs: minimal and long term;
mitigation avoids shallow
coral reefs
Minimal and temporary
• Sedentary invertebrate beds
and reefs: Minimal and short
term; mitigation avoids
shallow coral reefs
• Other biogenic habitats: no
effect
No effect
No effect
No effect
Note: HAPC = Habitat Area of Particular Concern
Pursuant to the EFH requirements of the MSA and implementing regulations, explosives on or near the
bottom and military expended materials may adversely affect EFH or HAPC at a minimal level, for
variable (habitat dependent) duration (refer to Section 4.1.5, Study Area Combined Impact of Stressors,
for analysis). The mitigation measures (see Table 5-1) should reduce the minimal potential impact from
permanent (e.g., hard bottom, submerged rooted vegetation, reefs) to short term (e.g., soft bottom,) for
explosives on or near the bottom and military expended materials deposited in nearshore and shallow
offshore habitats.
6-2
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
7 REFERENCES
Chapter 1
U.S. Department of the Navy. (2013). Mariana Islands Training and Testing Draft Environmental Impact
Statement/Overseas Environmental Impact Statement.
Chapter 2
Rosen, G. and G. R. Lotufo. (2010). "Fate and effects of Composition B in multispecies marine
exposures." Environ Toxicol Chem 29(6): 1330-1337.
U.S. Department of the Navy (2013). Mariana Islands Range Complex Airspace Environmental
Assessment/Overseas Environmental Assessment.
Chapter 3
Abraham, T., M. Beger, D. Burdick, E. Cochrane, P. Craig, G. Didonato, D. Fenner, A. Green, Y. Golbuu, J.
Gutierrez, M. Hasurmai, C. Hawkins, P. Houk, D. Idip, D. Jacobson, E. Joseph, T. Keju, J. Kuartei, S.
Palik, L. Penland, S. Pinca, K. Rikim, J. Starmer, M. S. Trianni, S. Victor and L. Whaylen. (2004). Status
of the coral Reefs in Micronesia and American Samoa. Status of Coral Reefs of the World: 2004. R.
Kelty and J. Kuartei: 381–409.
Aburto-Oropeza, O., E. Ezcurra, G. Danemann, V. Valdez, J. Murray and E. Sala. (2008). "Mangroves in
the Gulf of California increase fishery yields." Proceedings of the National Academy of Sciences
105(30): 10456–10459.
Bhattarai, B. and C. Giri. (2011). Assessment of mangrove forests in the Pacific region using Landsat
imagery. Journal of Applied Remote Sensing 5.
Biber, P. D., C. L. Gallegos and W. J. Kenworthy. (2007). "Calibration of a Bio-optical Model in the North
River, North Carolina (Albemarle–Pamlico Sound): A Tool to Evaluate Water Quality Impacts on
Seagrasses." Estuaries and Coasts 31(1): 177–191.
Birkeland, C. (1997). Status of coral reefs in the Marianas. Pages 91-100 in R.W. Grigg, and C. Birkeland,
eds. Status of Coral Reefs in the Pacific. Honolulu, Hawaii: Sea Grant College Program, School of
Ocean and Earth Science and Technology, University of Hawaii.
Bouillon, S. (2009). The management of natural coastal carbon sinks. D. D. A. Laffoley and G. Grimsditch.
Brown, B. E. (1997). "Coral bleaching: causes and consequences." Coral Reefs 16 Supplement: S129–
S138.
Carilli, J. E., R. D. Norris, B. Black, S. M. Walsh and M. McField. (2010). "Century-scale records of coral
growth rates indicate that local stressors reduce coral thermal tolerance threshold." Global Change
Biology 16(4): 1247–1257.
Carrell, T., D. Boyer, R. Davi, M. G. Driver, K. Foster, D. J. Lenihan, D. T. Lotz, T. B. McGrath, J. E. Miculka
and T. Rock. (1991). Micronesia Submerged Cultural Resources Assessment, Submerged Cultural
Resources Unit, Southwest Cultural Resources Center, Southwest Region, National Park Service, U.S.
Department of the Interior. Southwest Cultural Resources Center Professional Papers No. 36. Santa
Fe, New Mexico.
Castro, C. and M. E. Huber. (2007). Chemical and physical features of seawater and the world Ocean. In.
Marine Biology. New York, NY, McGraw-Hill: 45–68.
7-1
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Castro, P. and M. E. Huber. (2000). Marine prokaryotes, protists, fungi, and plants. In. Marine Biology,
McGraw-Hill: 83–103.
Chapman, L. (2004). Nearshore Domestic Fisheries Development in Pacific Island Countries and
Territories. S. o. t. P. Community. New Caledonia: 254.
Coale, K. H., K. S. Johnson, S. E. Fitzwater, S. P. G. Blain, T. P. Stanton and T. L. Coley. (1998). "IronEx-I, an
in situ iron-enrichment experiment: Experimental design, implementation and results." Deep-Sea
Research II 45: 919-945.
Coale, K. H., K. S. Johnson, S. E. Fitzwater, R. M. Gordon, S. Tanner, F. P. Chavez, L. Feroli, C. Sakamoto, P.
Rogers, F. J. Millero, P. Steinberg, P. Nightingale, D. Cooper, W. P. Cochlan, M. R. Landry, J.
Constantinou, G. Rollwagen, A. Trasvina and R. Kudela. (1996). "A massive phytoplankton bloom
induced by an ecosystem-scale iron fertilization experiment in the Equatorial Pacific." Nature 383:
495-501.
Cohen, A. L., D. C. McCorkle, S. de Putron, G. A. Gaetani and K. A. Rose. (2009). "Morphological and
compositional changes in the skeletons of new coral recruits reared in acidified seawater: Insights
into the biomineralization response to ocean acidification." Geochemistry Geophysics Geosystems
10(7): 1-12.
Commonwealth of the Northern Mariana Islands. (2001, July 17, 2001). "Shipwrecks, Groundings,
Marine Debris and Dredging." Retrieved June 13, 2005, from www.crm.gov.mp.
Cowardin, L. M., F. C. Carter, F. C. Golet and E. T. LaRoe. (1979). Classification of wetlands and
deepwater habitats of the United States. U.S. Department of the Interior Fish and Wildlife Service.
Washington, D.C., Northern Prairie Wildlife Research Center Home Page.
Daniel, R. and D. Minton. (2004). Inventory and monitoring program Pacific Island Network Monitoring
Plan - Appendix A: Marine Report, National Parks Service.
Davis, A. R. (2009). The role of mineral, living and artificial substrata in the development of subtidal
assemblages. In. Marine Hardbottom Communities: Patterns, Dynamics, Diversity and Change. M.
Wahl. New York, NY, Springer-Verlag. 206: 19-37.
Dawes, C. J. (1998). Marine Botany. New York, NY, John Wiley and Sons, Inc.
Department of the Navy (DoN). (2004). Year 2003 assessment of marine and fisheries resources-Farallon
de Medinilla, Commonwealth of the Northern Mariana Islands. Final report. Submitted to Pacific
Division, Naval Facilities Engineering Command, Pearl Harbor, Hawaii, under contract no. N6274202-D-1802, DO 002, by the Environmental Company, Honolulu, Hawaii.
Department of the Navy (DoN). (2005). Marine Resources Assessment for the Marianas Operations Area.
Pacific Division, Naval Facilities Engineering Command, Pearl Harbor, Hawaii. Contract #N62470-02D-9997, CTO 0027. Prepared by Geo-Marine, Inc., Plano, Texas.
Department of the Navy (DoN). (2007). Overseas Environmental Assessment. Surface Routine Training
Exercises in East and Gulf Coast Operation Areas and Seaward. January 2007.
Downs, C. A., E. Kramarsky-Winter, C. M. Woodley, A. Downs, G. Winters, Y. Loya and G. K. Ostrander.
(2009). "Cellular pathology and histopathology of hypo-salinity exposure on the coral Stylophora
pistillata." Science of the Total Environment 407(17): 4838-4851.
Eldredge, L. G. (1979). Marine Biological Resources within the Guam Seashore Study Area and the War in
the Pacific National Historical Park, University of Guam Marine Laboratory.
7-2
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Eldredge, L. G. (1983). Summary of Environmental and Fishing Information on Guam and
theCommonwealth of the Northern Mariana Islands: Historical Backgroun, Description of the
Islands, and Review of the Climate, Oceanography, and Submarine Topography, U.S. Department of
Commerce, National Oceanic and Atmospheric Administration, National Marine Fisheries Service,
Southwest Fisheries Center: 191.
Eldredge, L. G., R. Dickinson and S. Moras. (1977). Marine survey of Agat Bay. Mangilao, Guam,
University of Guam Marine Laboratory.
Embley, R. W., E. T. Baker, W. W. Chadwick Jr., J. E. Lupton, J. A. Resing, G. J. Massoth and K. Nakamura.
(2004). "Explorations of Mariana Arc Volcanoes Review New Hydrothermal Systems." EOS,
Transactions, American Geophysical Union 85(4): 37-44.
The Environmental Company. (2004). Year 2003 Assessment of Marine and Fisheries Resources, Farallon
de Medinilla, Commonwealth of the Northern Mariana Islands [Final report]. (pp. 46). Honolulu, HI.
Prepared for Pacific Division Naval Facilities Engineering Command.
Environmental Services Duenas & Associates, I. (1997). Saipan Lagoon Use Management Plan, Survey of
Sea Cucumbers and Fish in the Saipan Lagoon, Northern Mariana Islands: 57.
Federal Geographic Data Committee. (2012). Coastal and Marine Ecological Classification Standard.
Marine and Coastal Spatial Data Subcommittee. FGDC-STD-018-2012.
Fonseca, M. S., W. J. Kenworthy and G. W. Thayer (1998). Guidelines for the Conservation and
Restoration of Seagrasses in the United States and Adjacent Waters. Silver Spring, Maryland, NOAA
Coastal Ocean Office: 222.
Fox, H. E. and R. L. Caldwell. (2006). "Recovery From Blast Fishing On Coral Reefs: A Tale of Two Scales."
Ecological Applications 16(5): 1631–1635.
Garrison, T. (1998). Seawater chemistry. In. Oceanography: An Invitation to Marine Science. Belmont,
CA, Wadsworth Publishing Company: 138-153.
Garrison, T. (2004). Essentials of Oceanography. Pacific Grove, CA, Brooks/Cole-Thomas Learning.
Gaspin, J. B., Wiley, M. L. & Peters, G. B. (1976). Experimental investigations of the effects of underwater
explosions on swimbladder fish, II: 1975 Chesapeake Bay tests. White Oak Laboratory. Silver Spring,
Maryland. (document number) NSWC/WOL/TR 76-61.
Glynn, P. W. (1993). "Coral reef bleaching: ecological perspectives." Coral Reefs 12(1): 1-17.
Guam Department of Agriculture Division of Aquatic and Wildlife. (2004). Sport Fish Restoration FAD
Locations.
Guam Department of Agriculture, Division of Aquatic and Wildlife Resources. (2005). Guam
Comprehensive Wildlife Conservation Strategy (GCWCS). Page 61. Mangilao, Guam.
Heck, K. L., Jr., G. Hays and R. J. Orth. (2003). "Critical evaluation of the nursery role hypothesis for
seagrass meadows." Marine Ecology Progress Series 253: 123-136.
Hessler, R. R. and P. F. Lonsdale. (1991). "Biogeography of Mariana Trough hydrothermal vent
communities." Deep Sea Research 38(2): 185-199.
Hogarth, P. J. (1999). The Biology of Mangroves. New York, New York, Oxford University Press: 228.
Holland, K. T. and P. A. Elmore. (2008). "A review of heterogeneous sediments in coastal environments."
Earth-Science Reviews 89(3-4): 116-134.
7-3
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Humphris, S. E. (1995). Hydrothermal processes at mid-ocean ridges. Reviews of Geophysics
Supplement, Vol. 33. 33.
Hunter, E., Chant, R., Bowers, L., Glenn, S., Kohut, J. (2007). "Spatial and temporal variability of diurnal
wind forcing in the coastal ocean." Geophysical Research Letters 34(3): L03607.
Karleskint, G., R. Turner and J. W. Small, Jr. (2006). Introduction to Marine Biology. Belmont, CA,
Thomson Brooks/Cole: 460.
Kemp, W. M., R. Batiuk, R. Bartleson, P. Bergstrom, V. Carter, C. Gallegos, W. Hunley, L. Karrh, E. W.
Koch, J. Landwehr, K. Moore, L. Murray, M. Naylor, N. Rybicki, J. C. Stevenson and D. Wilcox. (2004).
"Habitat requirements for submerged aquatic vegetation in Chesapeake Bay: water quality, light
regime, and physical-chemical factors." Estuaries 27: 263-377.
Kennedy, J. (2012). "Red Algae (Rhodophyta)." About.com Mainre Life from
http://marinelife.about.com/od/plants/p/redalgae.htm.
Kennett, J. P. (1982). Marine Geology. New Jersey, Prentice Hall.
Kojima, S. (2002). "Deep-Sea Chemoautosynthesis-Based Communities in the Northwestern Pacific."
Journal of Oceanography 58: 343-363.
Kolinski, S. P., D. M. Parker, L. I. Ilo and J. K. Ruak. (2001). "An Assessment of the Sea Turtles and Their
Marine and Terrestrial Habitats at Saipan, Commonwealth of the Northern Mariana Islands."
Micronesica 34(1): 55-72.
Laegdsgaard, P. and C. R. Johnson. (1995). "Mangrove habitats as nurseries: unique assemblages of
juvenile fish in subtropical mangroves in eastern Australia." Marine Ecology Progress Series 126: 6781.
Lalli, C. M. and T. R. Parsons. (1993). Biological Oceanography: An Introduction. Vancouver, Canada,
University of British Columbia.
Lalli, C. M. and T. R. Parsons. (1997). Biological Oceanography: An Introduction. Vancouver, Canada,
University of British Columbia.
Langmann, B., K. Zaksek, M. Hort and S. Duggen. (2010). "Volcanic ash as fertiliser for the surface
ocean." Atmospheric Chemistry and Physics 10: 3891-3899.
Libes, S. M. (1992). An Introduction to Marine Biogeochemistry. New York, NY, John Wiley and Sons, Inc.:
734.
Lobban, C. S. and R. T. Tsuda. (2003). "Revised checklist of benthic marine macroalgae and seagrasses of
Guam and Micronesia." Micronesica 35-36: 54-99.
Lotz, D. (1998). World War II Remnants: Guam, Northern Mariana Islands, A Guide and History. Guam,
Making Tracks.
Mach, K. J., B. B. Hale, M. W. Denny and D. V. Nelson. (2007). "Death by small forces: a fracture and
fatigue analysis of wave-swept macroalgae." Journal of Experimental Biology 210(13).
Madden, C. J., K. Goodin, R. J. Allee, G. Cicchetti, C. Moses, M. Finkbeiner and D. Bamford. (2009).
Coastal and Marine Ecological Classification Standard - Version III, National Oceanic and
Atmospheric Administration and NatureServe.
The Mariana Trench - Biology - Part 1. (2003). The Mariana Trench - Biology - Part 1. 2005.
7-4
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Martin, J. H. and M. R. Gordon. (1988). "Northeast Pacific iron distributions in relation to phytoplankton
productivity." Deep-Sea Research 35(2): 177-196.
Minton, D., Brown, V., Dugger, K., Flores, T., Foster, K., Houk, P., Iguel, J., Kessler, C., Kolinski, S., Schiles,
T., Starmer, J., Suhraj, N., Tenorio, M., Tranni, M. (2009). Marine Resource Surveys of Tinian,
Commonwealth of the Northern Mariana Islands.
Mitsch, W. J., J. G. Gosselink, C. J. Anderson and L. Zhang. (2009). Wetland Ecosystems. Hoboken, NJ,
John Wiley & Sons, Inc.: 295.
Moffitt, R.B., and J. J. Polovina. (1987). Distribution and Yield of the Deepwater Shrimp Heterocarpus
Resource in the Marianas. Fishery Bulletin. Vol. 85 No. 2.
Moffitt, R.B. (1993). Deepwater demersal fish. Pages 73-95 in A. Wright and L. Hill, eds. Nearshore
marine resources of the South Pacific. Honiara, Solomon Islands: Forum Fisheries Agency; Suva, Fiji:
Institute of Pacific Studies; and Canada: International Centre for Ocean Development.
Moffitt, R.B., and F.A. Parrish. (1996). Habitat and life history of juvenile Hawaiian pink snapper,
Pristipomoides filamentosus. Pacific Science 50(4):371-381.
National Centers for Coastal Ocean Science and National Oceanic and Atmospheric Administration.
(2005). Shallow-water bethic habitats of American Samoa, Guam, and the Commonwealth of the
Northern Mariana Islands.
National Marine Fisheries Service. (2002). Magnuson-Stevens Act provisions; Essential Fish Habitat
(EFH)-Final rule. Federal Register. 67: 2343-2383.
National Marine Fisheries Service. (2010). "Deep Sea Corals." Retrieved September 2010, from
http://www.oar.noaa.gov/oceans/t_deepseacorals.html.
National Oceanic and Atmospheric Administration. (2007). National Artificial Reef Plan (as Amended):
Guidelines for siting, construction, development, and assessment of artifical reefs: 61.
National Oceanic and Atmospheric Administration. (2009). National Oceanographic Data Center.
Nybakken, J. W. (1993). Marine Biology, an Ecological Approach. New York, NY, Harper Collins College
Publishers.
Pacific Basin Environmental Consultants, I. (1985). CNMI Marine Parks Management Plan.
Pacific Regional Integrated Sciences and Assessment Program. (2012). Places: Commonwealth of the
Northern Mariana Islands (CNMI). Pacific RISA Managing Climate Risk in the Pacific. 2012.
Paulay, G. (2003). "Marine biodiversity of Guam and the Marianas: overview." Micronesica 35-36: 3-25.
Paulay, G., L. Kirkendale, G. Lambert, and J. Starmer. 1997. The biodiversity of Apra Harbor: Significant
areas and introduced species, with focus on sponges, echinoderms and ascidians. Prepared for U.S.
Department of Defense, COMNAVMARIANAS, University of Guam Marine Laboratory, Mangilao,
Guam.
Paulay, G., L. Kirkendale, G. Lambert and C. Meyer. (2002). "Anthropogenic Biotic Interchange in a Coral
Reef Ecosystem: A Case Study from Guam." Pacific Science 56(4): 403-422.
Paulay, G., M.P. Puglisi, and J.A. Starmer. (2003). " The non-scleractinian Anthozoa (Cnidaria) of the
Mariana Islands." Micronesica 35-36: 138-155.
Phillips, R. C. and E. G. Meñez. (1988). "Seagrasses." Smithsonian Contributions to the Marine Sciences
34: 104.
7-5
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Pickard, G. L. and W. J. Emery (1990). Descriptive Physical Oceanography: An Introduction. Oxford,
Pergamon Press.
Quinn, N.J., and B.L. Kojis. 2003. The dynamics of coral reef community structure and recruitment
patterns around Rota, Saipan, and Tinian, Western Pacific. Bulletin of Marine Science 72(3):979-996.
Randall, R. H. (1985). Habitat Geomorphology and Community Structure of Corals in the Mariana Islands.
Fifth International Coral Reef Congress, Tahiti.
Randall, R. H. (1995). "Biogeography of Reef-Building Corals in the Mariana and Palau Islands in Relation
to Back-Arc Rifting and the Formation of the Eastern Philippine Sea." Nat. Hist. Res. 3(2): 193-210.
Randall, R. H. (2003). "An annotated checklist of hydrozoan and scleractinian corals collected from Guam
and other Mariana Islands." Micronesica 35-36: 121-137.
Randall, R. H., H. G. Siegrist Jr. and A. W. Siegrist. (1984). "Community Structure of Reef-Building Corals
on a Recently Raised Holocene Reef on Guam, Mariana Islands." Palaeontographica Americana 54:
394-398.
Richmond, R.H. 1994. Effects of coastal runoff on coral reproduction. Pages 360-364 in R.N. Ginzburg,
ed. Proceedings on the Colloquium on Global Aspects of Coral Reefs: Health, Hazards, and History.
Miami, Florida: University of Miami, Rosenstiel School of Marine and Atmospheric Science.
Richmond, R.H., and G.W. Davis. 2002. Status of the coral reefs of Guam. Pages 189-194 in D.D. Turgeon,
R.G. Asch, B.D. Causey, R.E. Dodge, W. Jaap, K. Banks, J. Delaney, B.D. Keller, R. Speiler, C.A. Matos,
J.R. Garcia, E. Diaz, D. Catanzaro, C.S. Rogers, Z. Hillis-Starr, R. Nemeth, M. Taylor, G.P. Schmahl,
M.W. Miller, D.A. Gulko, J.E. Maragos, A.M. Friedlander, C.L. Hunter, R.S. Brainard, P. Craig, R.H.
Richmond, G. Davis, J. Starmer, M. Trianni, P. Houk, C.E. Birkeland, A. Edward, Y. Golbuu, J.
Gutierrez, N. Idechong, G. Paulay, A. Tafileichig, and N. Vander Velde. 2002. The state of coral reef
ecosystems of the United States and Pacific Freely Associated States: 2002. Silver Spring, Maryland:
National Oceanic and Atmospheric Administration/National Ocean Service/National Centers for
Coastal Ocean Science.
Riegl, B., R.P. Moyer, B.K. Walker, K. Kohler, D. Gilliam, and R.E. Dodge. 2008. A Tale of Germs, Storms,
and Bombs: Geomorphology and Coral Assemblage Structure at Vieques (Puerto Rico) Compared to
St. Croix (U.S. Virgin Islands). Journal of Coastal Research 24 (4): 1008-1021.
Roach, J. (2004). Mangroves are nursuries for reef rish, study finds. National Geographic News.
Rogers, A. D. (1994). "The biology of seamounts." Advances in Marine Biology 30: 305-350.
Rosen, G. and G. R. Lotufo. (2010). "Fate and effects of Composition B in multispecies marine
exposures." Environ Toxicol Chem 29(6): 1330-1337.
Ruwa, R. K. (1996). Intertidal wetlands. In East African Ecosystems and Their Conservation. T. R.
McClanahan and T. P. Young. New York, New York, Oxford University Press: 101-130.
Scott, D. A. (1993). A Directory of Wetlands in Oceania, Wetlands International. 2005.
Smith, S.H. (2004). March 2004 ecological assessment of the marine community in the vicinity of Kilo
Wharf, Apra Harbor, Guam. Pearl Harbor, Hawaii: Naval Facilities Engineering Command Pacific.
Smith, S.H. (2007). Ecological assessment of stony corals and associated organisms in the eastern
portion of Apra Harbor, Guam. Pearl Harbor, Hawaii: Naval Facilities Engineering Command Pacific.
7-6
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Smith, S.H., Marx, D.E., and Shannon, L.H. (2013). Calendar Year 2012 Assessment of Near Shore Marine
Resources at Farallon de Medinilla, Commonwealth of the Northern Mariana Islands. Prepared for
Pacific Fleet Environmental.
Smithsonian National Museum of Natural History (1995). Volcanic activity report: Ruby. In Bulletin of the
Global Volcanism Network. [Web page]. Retrieved from
http://www.geo.mtu.edu/volcanoes/central_pacific/marianas/gvn/marianas.v20n10.html, 6
January 2005.
South Atlantic Fishery Management Council. (1998). Final habitat plan for the South Atlantic region:
Essential fish habitat requirements for fishery management plans of the South Atlantic Fishery
Management Council. Charleston, SC, South Atlantic Fishery Management Council.
Spalding, M., M. Taylor, C. Ravilious, F. Short and E. Green. (2003). Global overview: The distribution and
status of seagrasses. In. World Atlas of Seagrasses. E. P. Green and F. T. Short. Berkeley, CA,
University of California Press: 5-26.
Speybroeck, J., D. Bonte, W. Courtens, T. Gheskiere, P. Grootaert, J. P. Maelfait, S. Provoost, K. Sabbe, E.
W. M. Stienen, V. Van Lancker, W. Van Landuyt, M. Vincx and S. Degraer. (2008). "The Belgian sandy
beach ecosystem: a review." Marine Ecology-an Evolutionary Perspective 29(Supplement 1): 171185.
Starmer, J., M.S. Trianni, and P. Houk. 2002. Status of coral reefs in the Commonwealth of the Northern
Mariana Islands. Pages 195-204 in D.D. Turgeon, R.G. Asch, B.D. Causey, R.E. Dodge, W. Jaap, K.
Banks, J. Delaney, B.D. Keller, R. Speiler, C.A. Matos, J.R. Garcia, E. Diaz, D. Catanzaro, C.S. Rogers, Z.
Hillis-Starr, R. Nemeth, M. Taylor, G.P. Schmahl, M.W. Miller, D.A. Gulko, J.E. Maragos, A.M.
Friedlander, C.L. Hunter, R.S. Brainard, P. Craig, R.H. Richmond, G. Davis, J. Starmer, M. Trianni, P.
Houk, C.E. Birkeland, A. Edward, Y. Golbuu, J. Gutierrez, N. Idechong, G. Paulay, A. Tafileichig, and N.
Vander Velde. The State of Coral Reef Ecosystems of the United States and Pacific Freely Associated
States: 2002. Silver Spring, Maryland: National Oceanic and Atmospheric Administration, National
Ocean Service, National Centers for Coastal Ocean Science.
Starmer, J., J. Asher, F. Castro, D. Gochfeld, J. Gove, A. Hall, P. Houk, E. Keenan, J. Miller, R. Moffit, M.
Nadon, R. Schroeder, E. Smith, M. Trianni, P. Vroom, K. Wong and K. Yuknavage. (2005). "The State
of Coral Reef Ecosystems of the Commonwealth of the Northern Mariana Islands." The State of
Coral Reef Ecosystems of the Commonwealth of the Northern Mariana Islands.
Starmer, J., J. Asher, F. Castro, D. Gochfeld, J. Gove, A. Hall, P. Houk, E. Keenan, J. Miller, R. Moffit, M.
Nadon, R. Schroeder, E. Smith, M. Trianni, P. Vroom, K. Wong and K. Yuknavage. (2008). The State of
Coral Reef Ecosystems of the Commonwealth of the Northern Mariana Islands. NOAA. In: J. Waddell
(ed.), The State of the Coral Reef Ecosystems of the United States and Pacific Freely Associated
States: 2008. Center for Coastal Monitoring and Assessment Biogeography Team.
Stuben, D., S. H. Bloomer, N. E. Taibi, T. Neumann, V. Bendel, U. Puschel, A. Barone, A. Lange, W.
Shiying, L. Cuizhong and Z. Deyu. (1992). "First results of study of sulphur-rich hydrothermal activity
from an island-arc environment: Esmeralda Bank in the Mariana Arc." Marine Geology 103: 521-528.
Takada, H., Mato, Y., Endo, S., Yamashita, R., and Zakaria, M. (2001). Pellet Watch: Global Monitoring of
Persistent Organic Pollutants (POPs) using Beached Plastic Resin Pellets. Accessed on March 18,
2014 from http://www.pelletwatch.org/documents/takadaproceeding.pdf.
Thompson, P. L. (2002). 76th SEABEES of World War II. 76th Bees - An Untold Story.
7-7
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Thurman, H. V. (1997). Primary productivity. In. Introductory Oceanography. Upper Saddle River, NJ,
Prentice Hall: 377-378.
Tomczak, M. and J. S. Godfrey. (2005). The Pacific Ocean. In. Regional Oceanography: An Introduction,
Pergamon.
Tsuda, R. T. (2009). Seaweeds Overview. 2011.
Tsuda, R. T., F. R. Fosberg and M. H. Sachet. (1977). "Distribution of Seagrases in Micronesia."
Micronesica 13(2): 191-193.
United Nations Educational Scientific and Cultural Organization. (2009). Global Open Oceans and Deep
Seabeds (GOODS) - Biogeographic Classification. Paris, France, UNESCO - IOC: 82.
U.S. Department of the Navy. (2003). Integrated Natural Resources Management Plan Farallon De
Medinilla and Tinian Military Lease Areas Commonwealth of the Northern Mariana Islands Plan
Duration: FY03-12: 359.
U.S. Department of the Navy. (2005). Year 2004 assessment, marine and fisheries resources--Farallon de
Medinilla, Commonwealth of the northern Mariana Islands. Page 46 pp. in Second working copy.
U.S. Department of the Navy. (2010). Mariana Islands Range Complex Final Environmental Impact
Statement/Overseas Environmental Impact Statement: 952.
U.S. Environmental Protection Agency. (2010). Water Quality Criteria - Suspended and Bedded
Sediments. Appendix 3.
U.S. Naval Base Guam. 2013. Marine ecological assessment of X-Ray Wharf, ApraHarbor, Guam. U.S.
Navy Naval Facilities Engineering Command. 36 pp.
Valiela, I. (1995). Marine Ecological Processes. New York, NY, Springer-Verlag.
van Oppen, M. J. H. and J. M. Lough. (2009). Coral Bleaching Patterns, Processes, Causes and
Consequences, Springer.
Vermeij, M. J. A., K. L. Marhaver, C. M. Huijbers, I. Nagelkerken and S. D. Simpson. (2010). "Coral larvae
move toward reef sounds." PLoS ONE 5(5): e10660.
Western Pacific Regional Fishery Management Council. (1979). Fishery Management Plan for the
Precious Coral Fisheries (and Associated Non-precious Corals) of the Western Pacific Region.
September.
Western Pacific Regional Fishery Management Council. (1981). Final Combined Fishery Management
Plan, Environmental Impact Statement, Regulatory Analysis and Draft Regulations for the Spiny
Lobster Fisheries of the Western Pacific Region. May.
Western Pacific Regional Fishery Management Council. (1986a). Combined Fishery Management Plan,
Environmental Assessment and Regulatory Impact Review for the Bottomfish and Seamount
Groundfish Fisheries of the Western Pacific Region; Final. Honolulu, HI.
Western Pacific Regional Fishery Management Council. (1986b). Fishery Management Plan for the
Pelagic Fisheries of the Western Pacific Region. July.
Western Pacific Regional Fishery Management Council. (2001). Final Fishery Management Plan for Coral
Reef Ecosystems of the Western Pacific Region. National Oceanic and Atmospheric Administration.
1.
7-8
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Western Pacific Regional Fishery Management Council. (2009). Fishery ecosystem Plan for the Mariana
Archipelago. NMFS. Honolulu, HI: 251.
Western Pacific Regional Fishery Management Council. (2012). Scientists weigh in on 2013 Fish Quotas,
Western-Central Pacific Tuna Management. 26 October 2012.
Wetzel, R. G. (2001). Limnology of Lake and River Ecosystems. San Diego, CA, Academic Press.
Wilkinson, C. (2002). Executive Summary. In. Status of Coral Reefs of the World: 2002. C. Wilkinson,
Global Coral Reef Monitoring Network: 7-31.
Wilkinson, C. (2004). Status of Coral Reefs of the World: 2004, Australian Institute of Marine Science.
Witman, J. D. and P. K. Dayton. (2001). Rocky subtidal communities. In. Marine community ecology.
M.D. Bertness et al: 339-366.
Wolanski, E., R. H. Richmond, G. Davis, E. Deleersnijder and R. R. Leben. (2003). "Eddies around Guam,
an island in the Mariana Islands group." Continental Shelf Research 23: 991-1003.
Chapter 4
Agency for Toxic Substances and Disease Registry. (2007). Toxicological profile for lead. Atlanta, GA, U.S.
Department of Health and Human Services, Public Health Services.
Amoser, S. and F. Ladich. (2005). "Are hearing sensitivities of freshwater fish adapted to the ambient
noise in their habitats?" Journal of Experimental Biology 208: 3533-3542.
Aplin, J. A. (1947). "The effect of explosives on marine life." California Fish and Game 33: 23-30.
Arfsten, D., C. Wilson and B. Spargo. (2002). "Radio Frequency Chaff: The Effects of Its Use in Training on
the Environment." Ecotoxicology and Environmental Safety 53: 1-11.
Astrup, J. (1999). "Ultrasound detection in fish - a parallel to the sonar-mediated detection of bats by
ultrasound-sensitive insects?" Comparative Biochemistry and Physiology, Part A 124: 19-27.
Astrup, J. and B. Mohl. (1993). "Detection of Intense Ultrasound by the Cod Gadus Morhua." Journal of
Experimental Biology 182: 71-80.
Au, W. W. L. and K. Banks. (1998). "The acoustics of the snapping shrimp Synalpheus parneomeris in
Kaneohe Bay." Journal of Acoustical Society of America, 103(1): 7.
Berglind, R., D. Menning, R. Tryman, A. Helte, P. Leffler and R. M. Karlsson. (2009). Environmental effects
of underwater explosions: a literature study, Totalforsvarets Forskningsinstitut, FOI.
Boehlert, G. W. and A. B. Gill. (2010). "Environmental and ecological effects of ocean renewable energy
development: A current synthesis." Oceanography 23(2): 68-81.
Bottger, S. A., J. B. McClintock and T. S. Klinger. (2001). "Effects of inorganic and organic phosphates on
feeding, feeding absorption, nutrient allocation, growth and righting responses of the sea urchin
Lytechinus variegatus." Marine Biology 138: 741–751.
Buchman, M. F. (2008). NOAA screening quick reference tables (NOAA OR&R Report 08-1). Seattle, WA,
Office of Response and Restoration Division. National Oceanic and Atmospheric Administration.
2011.
Budelmann, B. U. (1992a). Hearing by Crustacea. Evolutionary Biology of Hearing. R. R. F. a. A. N. P. D. B.
Webster. New York, Springer Verlag: 131-139.
7-9
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Budelmann, B. U. (1992b). Hearing in nonarthropod invertebrates. Evolutionary Biology of Hearing. R. R.
F. a. A. N. P. D. B. Webster. New York, Springer Verlag: 141-155.
Bullock, T. H., D. A. Bodznick and R. G. Northcutt. (1983). "The Phylogenetic Distribution of
Electroreception - Evidence for Convergent Evolution of a Primitive Vertebrate Sense Modality."
Brian Research Reviews 6(1): 25-46.
Buran, B. N., X. Deng and A. N. Popper. (2005). "Structural variation in the inner ears of four deep-sea
elopomorph fishes." Journal of Morphology 265: 215-225.
Carter, A. and S. Prekel. (2008). Benthic colonization and ecological successional patterns on a planned
nearshore artificial reef (AR) system in Broward County, SE Florida. In: 11th International Coral Reef
Symposium, Fort Lauderdale, FL, 7-11 July 2008.
Casper, B., P. Lobel and H. Yan. (2003). "The hearing sensitivity of the little skate, Raja erinacea: A
comparison of two methods." Environmental Biology of Fishes 68: 371-379.
Casper, B. and D. Mann. (2006). "Evoked potential audiograms of the nurse shark (Ginglymostoma
cirratum) and the yellow stingray (Urabatis jamaicensis)." Environmental Biology of Fishes 76: 101108.
Casper, B. M. and D. A. Mann. (2009). "Field hearing measurements of the Atlantic sharpnose shark
Rhizoprionodon terraenovae." J Fish Biol 75(10): 2768-2776.
Cato, D. H. and M. J. Bell. (1992). "Ultrasonic Ambient Noise in Australian Shallow Waters at Frequencies
up to 200 kHz." Materials Research Labs Ascot from http://handle.dtic.mil/100.2/ADA251679.
Chesapeake Biological Laboratory. (1948). Effects of underwater explosions on oysters, crabs and fish: a
preliminary report.
Collin, S. P. and D. L. Whitehead. (2004). "The functional roles of passive electroreception in non-electric
fishes." Animal Biology 54(1): 1-25.
Continental Shelf Associates. (2004). Explosive removal of offshore structures - information synthesis
report.
Coombs, S. and A. Popper. (1979). "Hearing Differences Among Hawaiian Squirrelfish (Family
Holocentridae) Related to Differences in the Peripheral Auditory System." Journal of Comparative
Physiology 132: 203-307.
Deng, X., H. J. Wagner and A. N. Popper. (2011). "The Inner Ear and its Coupling to the Swim Bladder in
the Deep-Sea Fish Antimora rostrata (Teleostei: Moridae)." Deep Sea Res Part 1 Oceanogr Res Pap
58(1): 27-37.
Derraik, J. G. B. (2002). "The pollution of the marine environment by plastic debris: A review." Marine
Pollution Bulletin 44: 842-852.
Doksæter, L., O. R. Godo, N. O. Handegard, P. H. Kvadsheim, F. P. A. Lam, C. Donovan and P. J. O. Miller.
(2009). "Behavioral responses of herring (Clupea harengus) to 1-2 and 6-7 kHz sonar signals and
killer whale feeding sounds." The Journal of the Acoustical Society of America 125(1): 554-564.
Egner, S. and D. Mann. (2005). "Auditory sensitivity of sergeant major damselfish Abudefduf saxatilis
from post-settlement juvenile to adult." Marine Ecology Progess Series 285: 213-222.
Formicki, K., A. Tanski, M. Sadowski and A. Winnicki. (2004). "Effects of magnetic fields on fyke net
performance." Journal of Applied Ichthyology 20(5): 402-406.
7-10
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Gaspin, J. B., G. B. Peters and M. L. Wisely. (1976). Experimental investigations of the effects of
underwater explosions on swimbladder fish. C. B. tests. White Oak, MD, Naval Ordnance Lab. II.
Gill, A. B. (2005). "Offshore renewable energy: ecological implications of generating electricity in the
coastal zone." Journal of Applied Ecology 42(4): 605-615.
Gochfeld, D. J. (2004). "Predation-induced morphological and behavioral defenses in a hard coral:
implications for foraging behavior of coral-feeding butterflyfishes." Marine Ecology-Progress Series
267: 145-158.
Goertner, J. F. (1982). Prediction of Underwater Explosion Safe Ranges for Sea Mammals. Dahlgren, VA,
Naval Surface Weapons Center: 25.
Goertner, J. F., M. L. Wiley, G. A. Young and W. W. McDonald. (1994). Effects of underwater explosions
on fish without swimbladders. Silver Spring, MD, Naval Surface Warfare Center.
Goodall, C., C. Chapman and D. Neil. (1990). The acoustic response threshold of Norway lobster
Nephrops norvegicus (L.) in a free found field. Frontiers in Crustacean Neurobiology W. D. K. K.
Weise, J. Tautz, H. Reichert and B. Mulloney. Basel, Birkhauser: 106 - 113.
Gorodilov, L. V. and A. P. Sukhotin. (1996). "Experimental investiagion of craters generated by explosions
of underwater surface charges on sand." Combustion, Explosion, and Shock Waves 32(3): 344-346.
Gregory, J. and P. Clabburn. (2003). "Avoidance behaviour of Alosa fallax fallax to pulsed ultrasound and
its potential as a technique for monitoring clupeid spawning migration in a shallow river." Aquatic
Living Resources 16: 313-316.
Hamernik, R. P. and K. D. Hsueh. (1991). "Impulse noise: some definitions, physical acoustics and other
considerations." Journal of the Acoustical Society of America 90(1): 189-196.
Hastings, M. and A. Popper. (2005). Effects of Sound on Fish: 82.
Hastings, M., A. Popper, J. Finneran and P. Lanford. (1996). "Effects of low-frequency underwater sound
on hair cells of the inner ear and lateral line of the teleost fish Astronotus ocellatus." Journal of the
Acoustical Society of America 99(3): 1759-1766.
Hastings, M. C. (1990). "Effects of underwater sound on fish."
Hastings, M. C. (1995). "Physical effects of noise on fishes." Proceedings of INTER0NOISE 95, The 1995
International Congress on Noise Control Engineering.
Hawkins, A. D. and A. D. F. Johnstone. (1978). "The hearing of the Atlantic Salmon, Salmo salar." Journal
of Fish Biology 13: 655-673.
Heberholz, J. and B. A. Schmitz. (2001). "Signaling via water currents in behavioral interactions of
snapping shrimp (Alpheus heterochaelis)." Biological Bulletin 201: 6-16.
Helfman, G. S., B. B. Collette, D. E. Facey and B. W. Bowen. (2009). The Diversity of Fishes.
Hu, Y. H., H. Y. Yan, W. S. Chung, J. C. Shiao and P. P. Hwang. (2009). "Acoustically evoked potentials in
two cephalopods inferred using the auditory brainstem response (ABR) approach." Comparative
Biochemistry and Physiology, Part A 153: 278–283.
Jeffs, A., N. Tolimieri and J. C. Montgomery. (2003). "Crabs on cue for the coast: the use of underwater
sound for orientation by pelagic crab stages." Marine Freshwater Resources 54: 841-845.
7-11
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Jorgensen, R., K. Olsen, I. Petersen and P. Kanapthipplai. (2005). Investigations of potential effects of low
frequency sonar signals on survival, development and behaviour of fish larvae and juveniles, The
Norwegian College of Fishery Science, University of Tromso, Norway: 51.
Kaifu, K., T. Akamatsu and S. Segawa. (2008). "Underwater sound detection by cephalopod statocyst."
Fisheries Science 74: 781–786.
Kajiura, S. M. and K. N. Holland. (2002). "Electroreception in Juvenile Scalloped Hammerhead and
Sandbar Sharks." The Journal of Experimental Biology 205: 3609-3621.
Kalmijn, A. J. (2000). "Detection and processing of electromagnetic and near-field acoustic signals in
elasmobranch fishes." Philosophical Transactions of the Royal Society of London Series B-Biological
Sciences 355(1401): 1135-1141.
Kane, A. S., J. Song, M. B. Halvorsen, D. L. Miller, J. D. Salierno, L. E. Wysocki, D. Zeddies and A. N.
Popper. (2010). "Exposure of fish to high intensity sonar does not induce acute pathology." Journal
of Fish Biology 76(7): 1825-1840.
Keevin, T. M. and G. L. Hempen. (1997). The environmental effects of underwater explosions with
methods to mitigate impacts. St. Louis, MO.
Kenyon, T. (1996). "Ontogenetic changes in the auditory sensitivity of damselfishes (pomacentridae)."
Journal of Comparative Physiology 179: 553-561.
Ketten, D. R. (1998). Marine Mammal Auditory Systems: A Summary of Audiometric and Anatomical
Data and its Implications for Underwater Acoustic Impacts. La Jolla, CA, U.S. Department of
Commerce, National Oceanic and Atmospheric Administration, National Marine Fisheries Service,
Southwest Fisheries Science Center: 74.
Kvadsheim, P. H. and E. M. Sevaldsen. (2005). "The potential impact of 1-8 kHz active sonar on stocks of
juvenile fish during sonar exercises." Forsvarets Forskningsinstitutt.
Ladich, F. (2008). "Sound communication in fishes and the influence of ambient and anthropogenic
noise." Bioacoustics 17: 35-37.
Ladich, F. and A. N. Popper. (2004). Parallel Evolution in Fish Hearing Organs. Evolution of the Vertebrate
Auditory System, Springer Handbook of Auditory Research. A. N. P. a. R. R. F. G. A. Manley. New
York, Springer-Verlag.
Latha, G., S. Senthilvadivu, R. Venkatesan and V. RajendranLindholm. (2005). "Sound of shallow and
deep water lobsters: Measurements, analysis, and characterization." Journal of the Acoustical
Society of America(117): 2720-2723.
Lohmann, K. J., N. D. Pentcheff, G. A. Nevitt, G. D. Stetten, R. K. Zimmer-Faust, H. E. Jarrard and L. C.
Boles. (1995). "Magnetic orientation of spiny lobsters in the ocean: Experiments with undersea coil
systems." Journal of Experimental Biology 198(10): 2041-1048.
Lombarte, A. and A. N. Popper. (1994). "Quantitative analyses of postembryonic hair cell addition in the
otolithic endorgans of the inner ear of the European hake, Merluccius merluccius (Gadiformes,
Teleostei)." Journal of Comparative Neurology 345(419-428).
Lovell, J. M., M. M. Findlay, R. M. Moate, J. R. Nedwell and M. A. Pegg. (2005). "The inner ear
morphology and hearing abilities of the Paddlefish (Polyodon spathula) and the Lake Sturgeon
(Acipenser fulvescens)." Comparative Biochemistry and Physiology Part A 142: 286-296.
7-12
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Lovell, J. M., R. M. Moate, L. Christiansen and M. M. Findlay. (2006). "The relationship between body
size and evoked potentials from the statocysts of the prawn Palaemon serratus." The Journal of
Experimental Biology 209: 2480-2485.
Mackie, G. O. and C. L. Singla. (2003). "The Capsular Organ of Chelyosoma productum (Ascidiacea:
Corellidae): A New Tunicate Hydrodynamic Sense Organ." Brain, Behavior and Evolution 61: 45-58.
Mann, D., D. Higgs, W. Tavolga, M. Souza and A. Popper. (2001). "Ultrasound detection by clupeiform
fishes." Journal of the Acoustical Society of America: 3048-3054.
Mann, D., A. Popper and B. Wilson. (2005). "Pacific herring hearing does not include ultrasound." Biology
Letters 1: 158-161.
Mann, D. A., Z. Lu, M. C. Hastings and A. N. Popper. (1998). "Detection of ultrasonic tones and simulated
dolphin echolocation clicks by a teleost fish, the American shad (Alosa sapidissima)." Journal of the
Acoustical Society of America 104(1): 562-568.
Mann, D. A., Z. Lu and A. N. Popper. (1997). "A clupeid fish can detect ultrasound." Nature: 389-341.
Marcotte, M. M. and C. G. Lowe. (2008). "Behavioral responses of two species of sharks to pulsed, direct
current electrical fields: Testing a potential shark deterrent." Marine Technology Society Journal
42(2): 53-61.
Mato, Y., T. Isobe, H. Takada, H. Kanehiro, C. Ohtake and T. Kaminuma. (2001). "Plastic Resin Pellets as a
Tranport Medium for Toxic Chemicals in the Marine Environment." Environmental Science
Technology 35: 318-324.
McCauley, R. D., J. Fewtrell, A. J. Duncan, C. Jenner, M.-N. Jenner, J. D. Penrose, R. I. T. Prince, A.
Adhitya, J. Murdoch and K. McCabe. (2000). "Marine seismic surveys: A study of environmental
implications." Appea Journal 2000: 692-708.
McLennan, M. W. (1997). "A simple model for water impact peak pressure and width: a technical
memorandum."
Miller, J. D. (1974). "Effects of noise on people." Journal of the Acoustical Society of America 56(3): 729764.
Mintz, J. and R. Filadelfo. (2011). Exposure of Marine Mammals to Broadband Radiated Noise. Specific
Authority N0001-4-05-D-0500. CNA Analysis & Solutions: 42.
Mintz, J. D. and C. L. Parker. (2006). Vessel Traffic and Speed Around the U.S. Coasts and Around Hawaii.
Alexandria, VA, CNA Corporation: 48.
Montgomery, J. C., A. Jeffs, S. D. Simpson, M. Meekan and C. Tindle. (2006). "Sound as an orientation
cue for the pelagic larvae of reef fishes and decapod crustaceans." Adv Mar Biol 51: 143-196.
Mooney, T. A., R. T. Hanlon, J. Christensen-Dalsgaard, P. T. Madsen, D. Ketten and P. E. Nachtigall.
(2010). "Sound detection by the longfin squid (Loligo pealeii) studied with auditory evoked
potentials: sensitivity to low-frequency particle motion and not pressure." J Exp Biol 213: 3748-3759.
Myrberg, A. A. (2001). "The acoustical biology of elasmobranchs." Environmental Biology of Fishes
60(31-45).
Negri, A. P., L. D. Smith, N. S. Webster and A. J. Heyward. (2002). "Understanding ship-grounding
impacts on a coral reef: potential effects of anti-foulant paint contamination on coral recruitment."
Marine Pollution Bulletin 44(2): 111-117.
7-13
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Normandeau, Exponent, T. T. and A. Gill. (2011). Effects of EMFs from Undersea Power Cables on
Elasmobranchs and Other Marine Species. Camarillo, CA, U.S. Dept. of the Interior, Bureau of Ocean
Energy Management, Regulation, and Enforcement, Pacific OCS Region.
O'Connell, C. P., D. C. Abel, P. H. Rice, E. M. Stroud and N. C. Simuro. (2010). "Responses of the southern
stingray (Dasyatis americana) and the nurse shark (Ginglymostoma cirratum) to permanent
magnets." Marine and Freshwater Behaviour and Physiology 43(1): 63-73.
O'Keeffe, D. J. and G. A. Young. (1984). Handbook on the Environmental Effects of Underwater
Explosions, Naval Surface Weapons Center.
Ohman, M. C., P. Sigray and H. Westerberg. (2007). "Offshore windmills and the effects electromagnetic
fields an fish." Ambio 36(8): 630-633.
Packard, A., H. E. Karlsen and O. Sand. (1990). "Low frequency hearing in cephalopods." Journal of
Comparative Physiology A 166: 501-505.
Pait, A. S., A. L. Mason, D. R. Whitall, J. D. Christensen and S. I. Hartwell. (2010). Chapter 5: Assessment
of chemical contaminants in sediments and corals in Vieques. In. An ecological characterization of
the marine resources of Vieques, Puerto Rico. L. J. Bauer and M. S. Kendall. Silver Spring, MD, NOAA
MCCOS 110: 101–150.
Patek, S. N. and R. L. Caldwell. (2006). "The stomatopod rumble: Low frequency sound production in
Hemisquilla californiensis." Marine and Freshwater Behaviour and Physiology 39(2): 99-111.
Patek, S. N., L. E. Shipp and E. R. Staaterman. (2009). "The acoustics and acoustic behavior of the
California spiny lobster (Panulirus interruptus)." Journal of the Acoustical Society of America 125(5):
3434-3443.
Pater, L. L. (1981). Gun blast far field peak overpressure contours, Naval Surface Weapons Center.
Popper, A. (1977). "A Scanning Electron Microscopic Study of the Sacculus and Lagena in the Ears of
Fifteen Species of Teleost Fishes." J. Morph 153: 397-417.
Popper, A. (1980). "Scanning Electron Microscopic Study of the Sacculus and Lagena in Several Deep-Sea
Fishes." The American Journal of Anatomy 157: 115-136.
Popper, A. N., M. Salmon and K. W. Horch. (2001). "Acoustic detection and communication by decapod
crustaceans." Journal of Comparative Physiology A 187: 83-89.
Popper, A. (2003). "Effects of Anthropogenic Sounds on Fishes." Fisheries 28(10): 24-31.
Popper, A. N. (2008). Effects of mid- and High-Frequency Sonars on Fish. Newport, Rhode Island, Naval
Undersea Warfare Center Division: 52.
Popper, A. N. and R. R. Fay. (2010). "Rethinking sound detection by fishes." Hearing Research.
Popper, A. N., R. R. Fay, C. Platt and O. Sand. (2003). Sound detection mechanisms and capabilities of
teleost fishes. Sensory Processing in Aquatic Environment. S. P. Collin and N. J. Marshall. New York,
Springer-Verlag.
Popper, A. N. and M. C. Hastings. (2009). "The effects of anthropogenic sources of sound on fishes."
Journal of Fish Biology 75(3): 455-489.
Popper, A. N. and B. Hoxter. (1987). "Sensory and nonsensory ciliated cells in the ear of the sea lamprey,
Petromyzon marinus." Brain, Behavior and Evolution 30: 43-61.
7-14
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Popper, A. N. and C. R. Schilt. (2008). Hearing and acoustic behavior (basic and applied). In:. Fish
Bioacoustics. J. F. Webb, R. R. Fay and A. N. Popper. New York, Springer Science + Business Media,
LLC.
Radford, C., A. Jeffs and J. C. Montgomery. (2007). "Directional swimming behavior by five species of
crab postlarvae in response to reef sound." Bulletin of Marine Science 2(80): 369-378.
Radford, C., J. Stanley, C. Tindle, J. C. Montgomery and A. Jeffs. (2010). "Localised coastal habitats have
distinct underwater sound signatures." Marine Ecology Progress Series 401: 21-29.
Ramcharitar, J., D. Higgs and A. Popper. (2006). "Audition in sciaenid fishes with different swim bladderinner ear configurations." Journal of the Acoustical Society of America 119(1): 439-443.
Ramcharitar, J. U., X. Deng, D. Ketten and A. N. Popper. (2004). "Form and function in the unique inner
ear of a teleost: The silver perch (Bairdiella chrysoura)." Journal of Comparative Neurology 475(4):
531-539.
Rigg, D. P., S. C. Peverell, M. Hearndon and J. E. Seymour. (2009). "Do elasmobranch reactions to
magnetic fields in water show promise for bycatch mitigation?" Marine and Freshwater Research
60(9): 942-948.
Rosen, G. and G. Lotufo. (2007a). "Toxicity of explosive compounds to the marine mussel, Mytilus
galloprovincialis, in aqueous exposures." Ecotoxicology and Environmental Safety 68(2).
Rosen, G. and G. R. Lotufo. (2007b). "Bioaccumulation of explosive compounds in the marine mussel,
Mytilus galloprovincialis." Ecotoxicol Environ Safety 68(2): 237-245.
Rosen, G. and G. R. Lotufo. (2010). "Fate and effects of Composition B in multispecies marine
exposures." Environ Toxicol Chem 29(6): 1330-1337.
Simpson, S. D., A. N. Radford, E. J. Tickle, M. G. Meekan and A. Jeffs. (2011). "Adaptive Avoidance of Reef
Noise." PLoS ONE 6(2).
Singh, B. and N. Sharma. (2008). "Mechanistic implications of plastic degradation." Polymer Degradation
and Stability 93(3): 561-584.
Sisneros, J. A. and A. H. Bass. (2003). "Seasonal Plasticity of Peripheral Auditory Frequency Sensitivity."
The Journal of Neuroscience 23(3): 1049-1058.
Slabbekoorn, H., N. Bouton, I. van Opzeeland, A. Coers, C. ten Cate and A. N. Popper. (2010). "A noisy
spring: The impact of globally rising underwater sound levels on fish." Trends in Ecology and
Evolution 25(7): 419-427.
Smith, M. E., A. B. Coffin, D. L. Miller and A. N. Popper. (2006). "Anatomical and functional recovery of
the goldfish (Carassius auratus) ear following noise exposure." Journal of Experimental Biology
209(Pt 21): 4193-4202.
Sprague, M. W. and J. J. Luczkovich. (2004). "Measurement of an individual silver perch Bairdiella
chrysoura sound pressure level in a field recording." Journal of the Acoustical Society of America
116(5): 3186-3191.
Stanley, J., C. Radford and A. Jeffs. (2010). "Induction of settlement in crab megalopae by ambient
underwater reef sound." Behavioral Ecology 21(3): 113-120.
Teuten, E., S. Rowland, T. Galloway and R. Thompson. (2007). "Potential for Plastics to Transport
Hydrophobic Contaminants." Environmental Science Technology 41: 7759-7764.
7-15
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
U.S. Department of the Army. (1999). Finding of No Significant Impact (FONSI) for the Life Cycle
Environmental Assessment (LCEA) for the HELLFIRE Modular Missile System.
U.S. Department of the Navy. (1996a). Draft environmental assessment of the use of selected Navy test
sites for development tests and fleet training exercises of the MK- 46 and MK- 50 torpedoes,
Program Executive Office, Antisubmarine Warfare Assault and Special Mission Programs.
(CONFIDENTIAL).
U.S. Department of the Navy. (1996b). Environmental assessment of the use of selected Navy test sites
for development tests and fleet training exercises of the MK-48 torpedoes, Program Executive Office
Undersea Warfare, Program Manager for Undersea Weapons. (CONFIDENTIAL).
U.S. Department of the Navy. (2000). Noise Blast Test Results Aboard the USS Cole. Gun Blast
Transmission into Water Test with a 5-Inch/54 Caliber Naval Gun (Standard Ordnance).
U.S. Department of the Navy. (2005). Biological Assessment for Sinking Exercises (SINKEXs) in the
Western North Atlantic Ocean. C. F. F. C. U.S. Department of the Navy. Newport, VA, Naval
Undersea Warfare Division, Newport.
U.S. Department of the Navy. (2008). Final Environmental Impact Statement/Overseas Environmental
Impact Statement (EIS/OEIS) Shock Trial of the MESA VERDE (LPD 19). 1, 2: 348, 404.
U.S. Department of the Navy. (2010). Jacksonville (JAX) Operating Area (OPAREA) Undersea Warfare
Trainng Range (USWTR) Bottom Mapping and Habitat Characterization, Florida. Final Cruise Report.
Norfolk, Virginia, Naval Facilities Engineering Command Atlantic.
U.S. Department of the Navy. (2012). Determination of Acoustic Effects on Marine Mammals and Sea
Turtles for the Hawaii-Southern California Training and Testing Environmental Impact
Statement/Overseas Environmental Impact Statement DRAFT - Version 0.5. Marine Species
Modeling Team. Newport, Rhode Island, Naval Undersea Warfare Center Division: 34.
U.S. Environmental Protection Agency. (1999). "Ocean Regulatory Programs August 1999 SINKEX Letter
Agreement between EPA and the Navy." Retrieved March 8, 2010.
Vermeij, M. J. A., K. L. Marhaver, C. M. Huijbers, I. Nagelkerken and S. D. Simpson. (2010). "Coral larvae
move toward reef sounds." PLoS ONE 5(5): e10660.
Wang, W. X. and P. S. Rainbow. (2008). "Comparative approaches to understand metal bioaccumulation
in aquatic animals." Comp Biochem Physiol C Toxicol Pharmacol 148(4): 315-323.
Wilson, M., R. T. Hanlon, P. L. Tyack and P. T. Madsen. (2007). "Intense ultrasonic clicks from
echolocating toothed whales do not elicit anti-predator responses or debilitate the squid Loligo
pealeii." Biology Letters 3: 225-227.
Wright, A., N. Soto, A. Baldwin, M. Bateson, C. Beale, C. Clark, T. Deak, E. Edwards, A. Fernandez, A.
Godinho, L. Hatch, A. Kakuschke, D. Lusseau, D. Martineau, M. Romero, L. Weilgart, B. Wintle, G.
Notarbartolo-di-Sciara and V. Martin. (2007). "Anthropogenic Noise as a Stressor in Animals: A
Multidisciplinary Perspective." International Journal of Comparative Psychology.
Wright, D. G. (1982). A Discussion Paper on the Effects of Explosives on Fish and Marine Mammals in the
Waters of the Northwest Territories. Canadian Technical Report of Fisheries and Aquatic Sciences.
Winnipeg, Manitoba, Western Region Department of Fisheries and Oceans: 1-16.
Wright, K. J., D. M. Higgs, A. J. Belanger and J. M. Leis. (2005). "Auditory and olfactory abilities of presettlement larvae and post-settlement juveniles of a coral reef damselfish (Pisces: Pomacentridae)."
Marine Biology 147(6): 1425-1434.
7-16
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Yagla, J. and R. Stiegler. (2003). Gun Blast Noise Transmission Across the Air-Sea Interface. euronoise.
Naples: 9.
Yelverton, J. T., D. R. Richmond, W. Hicks, K. Saunders and E. R. Fletcher. (1975). The Relationship
Between Fish Size and Their Response to Underwater Blast. Defense Nuclear Agency. Washington,
D.C., Lovelace Foundation for Medical Education and Research: 40.
Young, G. A. (1991). Concise methods for predicting the effects of underwater explosions on marine life.
Silver Spring, Naval Surface Warfare Center.
Zelick, R., D. Mann and A. N. Popper. (1999). Acoustic communication in fishes and frogs. Comparative
Hearing: Fish and Amphibians a. A. N. P. R. R. Fay. New York, Springer-Verlag: 363-411.
7-17
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
This Page Intentionally Left Blank
7-18
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
APPENDIX A LIST OF FEDERALLY MANAGED SPECIES
WESTERN PACIFIC REGIONAL FISHERY MANAGEMENT COUNCIL
BOTTOMFISH MANAGEMENT UNIT SPECIES
Blue-lined surgeon (Acanthurus nigroris)
Bluespine unicornfish (Naso unicornus)
Brick soldierfish (Myripristis amaena)
Bridled triggerfish (Sufflamen fraenatum)
Brown surgeonfish (Acanthurus nigrofuscus)
Butterflyfish (Chaetodon auriga)
Cigar wrasse (Cheilio inermis)
Convict tang (Acanthurus triostegus)
Crown squirrelfish (Sargocentron diadema)
Doublebar goatfish (Parupeneus bifasciatus)
Dragon eel (Enchelycore pardalis)
Eye-striped surgeonfish (Acanthurus dussumieri)
False mullet (Neomyxus leuciscus)
Featherduster worm (Sabellidae)
File-lined squirrelfish (Sargocentron microstoma)
Galapagos shark (Carcharhinus galapagensis)
Giant moray eel (Gymnothorax javanicus)
Glasseye (Heteropriacanthus cruentatus)
Gray unicornfish (Naso caesius)
Great barracuda (Sphyraena barracuda)
Green snails turban shells (Turbo spp.)
Grey reef shark (Carcharhinus amblyrhynchos)
Hawaiian flag-tail (Kuhlia sandvicensis)
Hawaiian squirrelfish (Sargocentron xantherythrum)
Heller’s barracuda (Sphyraena helleri)
Mackerel scad (Decapterus macarellus)
Moorish idol (Zanclus cornutus)
Multi-barred goatfish (Parupeneus multifaciatus)
Octopus (Octopus cyanea)
Octopus (Octopus ornatus)
Orange goatfish (Mulloidichthys pfleugeri)
Orangespine unicornfish (Naso lituratus)
Orange-spot surgeonfish (Acanthurus olivaceus)
Parrotfish (Scarus spp.)
Pearly soldierfish (Myripristis kuntee)
Peppered squirrelfish (Sargocentron
punctatissimum)
Picassofish (Rhinecanthus aculeatus)
Pinktail triggerfish (Melichthys vidua)
Raccoon butterflyfish (Chaetodon lunula)
Razor wrasse (Xyrichtys pavo)
Red ribbon wrasse (Thalassoma quinquevittatum)
Ringtail surgeonfish (Acanthurus blochii)
Ring-tailed wrasse (Oxycheilinus unifasciatus)
Rockmover wrasse (Novaculichthys taeniourus)
Rudderfish (Kyphosus biggibus)
Rudderfish (Kyphosus cinerascens)
Rudderfish (Kyphosus vaigiensis)
Alfonsin (Beryx splendens)
Amberjack (Seriola dumerili)
Armorhead (Pseudopentaceros wheeleri)
Black jack (Caranx lugubris)
Blue stripe snapper (Lutjanus kasmira)
Giant trevally (Caranx ignoblis)
Gray jobfish (Aprion virescens)
Hawaiian grouper (Epinephelus quernus)
Longtail snapper (Etelis coruscans)
Pink snapper (Pristipomoides filamentosus)
Pink snapper (Pristipomoides sieboldii)
Raftfish (Hyperoglyphe japonica)
Silver jaw jobfish (Aphareus rutilansi)
Squirrelfish snapper (Etelis carbunculus)
Thick lipped trevally (Pseudocaranx dentex)
Yellow-barred snapper (Pristipomoides zonatus)
Yellowtail snapper (Pristipomoides auricilla)
CRUSTACEANS MANAGEMENT UNIT SPECIES
Deepwater shrimp (Heterocarpus spp.)
Kona crab (Ranina ranina)
Slipper lobster (Family Scyllaridae)
Spiny lobster (Panulirus penicillatus)
Spiny lobster (Panulirus marginatus)
PRECIOUS CORALS MANAGEMENT UNIT SPECIES
Black coral (Antipathes dichotoma)
Black coral (Antipathes grandis)
Black coral (Antipathes ulex)
Pink coral (Corallium laauense)
Pink coral (Corallium regale)
Pink coral (Corallium secundum)
Gold coral (Gerardia spp.)
Bamboo coral (Lepidisis olapa)
Gold coral (Narella spp.)
CORAL REEF ECOSYSTEM MANAGEMENT UNITS SPECIES,
CURRENTLY HARVESTED CORAL REEF TAXA
Banded goatfish (Parupeneus spp.)
Bandtail goatfish (Upeneus arge)
Bigeye (Priacanthus hamrur)
Bigeye scad (Selar crumenophthalmus)
Bigscale soldierfish (Myripristis berndti)
Black tongue unicornfish (Naso hexacanthus)
Black triggerfish (Melichthys niger)
Blacktip reef shark (Carcharhinus melanopterus)
Blue-lined squirrelfish (Sargocentron tiere)
A-1
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Saber or long jaw squirrelfish (Sargocentron
spiniferum)
Saddleback butterflyfish (Chaetodon ephippium)
Saddleback hogfish (Bodianus bilunulatus)
Side-spot goatfish (Parupeneus pleurostigma)
Spotfin squirrelfish (Neoniphon spp.)
Spotted unicornfish (Naso brevirostris)
Stareye parrotfish (Calotomus carolinus)
Striped bristletooth (Ctenochaetus striatus)
Stripped mullet (Mugil cephalus)
Sunset wrasse (Thalassoma lutescens)
Surge wrasse (Thalassoma purpureum)
Threadfin (Polydactylus sexfilis)
Undulated moray eel (Gymnothorax undulates)
Whitebar surgeonfish (Acanthurus leucopareius)
Whitecheek surgeonfish (Acanthurus nigricans)
Whitemargin unicornfish (Naso annulatus)
White-spotted surgeonfish (Acanthurus guttatus)
Whitetip reef shark (Triaenodon obesus)
Yellow goatfish (Mulloidichthys spp.)
Yellow tang (Zebrasoma flavescens)
Yellow-eyed surgeonfish (Ctenochaetus strigosus)
Yellowfin goatfish (Mulloidichthys vanicolensis)
Yellowfin soldierfish (Myripristis chryseres)
Yellowfin surgeonfish (Acanthurus xanthopterus)
Yellowmargin moray eel (Gymnothorax
flavimarginatus)
Yellowsaddle goatfish (Parupeneus cyclostomas)
Yellowstripe goatfish (Mulloidichthys flavolineatus)
Frogfishes (Antennariidae)
Goatfishes (Those species not listed as CHCRT)
Mullidae
Gobies (Gobiidae)
Groupers, seabass (Those species not listed as
CHCRT or in BMUS)
Serrandiae
Hawkfishes (Those species not listed as CHCRT)
Cirrhitidae
Herrings (Clupeidae)
Hydroid corals (Solanderidae)
Hydrozoans and Bryzoans
Jacks and scads (Those species not listed as CHCRT or
in BMUS)
Carangidae
Lace corals (Stylasteridae)
Live rock
Lobsters, shrimps, mantis shrimps, true crabs, and
hermit crabs (Those species not listed as Crustacean
Management Unit Species [CMUS])
Crustaceans
Mollusca (Those species not listed as CHCRT)
Moorish Idols (Zanclidae)
Mushroom corals (Fungiidae)
Octopi (Cephalopods)
Other clams (Other Bivalves)
Pipefishes and seahorses (Syngnathidae)
Puffer fishes and porcupine fishes (Tetradontidae)
Rays and skates (Dasyatididae)
Rays and skates (Myliobatidae)
Remoras (Echeneidae)
Rudderfishes (Those species not listed as CHCRT)
Kyphosidae
Sandperches (Pinguipedidae)
Scorpionfishes, lionfishes (Scorpaenidae)
Sea cucumbers and sea urchins (Echinoderms)
Sea slugs (Opistobranchs)
Sea snails (Gastropoda)
Sea squirts (Tunicates)
Seaweed (Algae)
Segmented worms (Those species not listed as
CHCRT) Annelids
Sharks (Sphyrnidae)
Sharks (Those species not listed as CHCRT)
Carcharhinidae
Small and large coral polyps (Fungiidae)
Snappers (Those species not listed as CHCRT or in
BMUS) Lutjanidae
Soft corals and gorgonians (Fungiidae)
Soft zoanthid corals (Zoanthinaria)
Solderfishes and squirrelfishes (Those species not
listed as CHCRT) Holocentridae
Sponges (Porifera)
Surgeonfishes (Those species not listed as CHCRT)
Acanthuridae
CORAL REEF ECOSYSTEM MANAGEMENT UNITS SPECIES,
POTENTIALLY HARVESTED CORAL REEF TAXA
Ahermatypic corals (Azooxanthellates)
Anchovies (Engraulidae)
Anemones (Actinaria)
Angelfishes (Pomacanthidae)
Barracudas (Those species not listed as CHCRT)
Black lipped pearl oyster (Pinctada margaritifera)
Blennies (Blenniidae)
Butterflyfishes (Chaetodontidae)
Cardinalfishes (Apogonidae)
Coral crouchers (Caracanthidae)
Cornetfish (Fistularia commersoni)
Damselfishes (Pomacentridae)
Eels (Those species not listed as CHCRT)
Muraenidae
Eels (Those species not listed as CHCRT)
Congridae
Eels (Those species not listed as CHCRT)
Ophichthidae
Flounders and soles (Bothidae)
Flounders and soles (Soleidae)
Flounders and soles (Pleurnectidae)
A-2
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Tilefishes (Malacanthidae)
Trigger fishes (Balistidae)
Trumpetfish (Aulostomus chinensis)
Trunkfishes (Ostraciidae)
All other coral reef ecosystem management unit
species that are marine plants, invertebrates, and
fishes that are not listed in the preceding tables or
are not bottomfish management unit species, CMUS,
Pacific pelagic management unit species, precious
coral or seamount groundfish.
PELAGIC MANAGEMENT UNITS SPECIES
Albacore (Thunnus alalunga)
Bigeye thresher shark (Alopias superciliosus)
BigeyeTtuna (Thunnus obesus)
Black marlin (Makaira indica)
Blue marlin (Makaira nigricans)
Blue shark (Prionace glauca)
Bluefin Tuna (Thunnus thynnus)
Thresher shark (Alopias vulpinus)
Diamondback squid (Thysanoteuthis rhombus)
Dogtooth tuna (Gymnosarda unicolor)
Frigate and bullet tunas (Auxis thazard, A. rochei)
Kawakawa (Euthynnus affinis)
Longfin mako shark (Isurus paucus)
Mackerel (Scomber spp.)
Mahimahi (Coryphaena hippurus, C. equiselas)
Neon flying squid (Ommastrephes bartamii)
Oceanic whitetip shark (Carcharhinus longimanus)
Ono (Acanthocybium solandri)
Opah (Lampris spp.)
Pelagic thresher shark (Alopias pelagicus)
Pomfret (family Bramidae)
Purple flying squid (Sthenoteuthis oualaniensis)
Sailfish (Istiophorus platypterus)
Salmon shark (Lamna ditropis)
Shortfin mako shark (Isurus oxyrinchus)
Silky shark (Carcharhinus falciformis)
Skipjack (Katsuwonus pelamis)
Slender tunas (Allothunnus fallai)
Spearfish (Tetrapturus spp.)
Striped Marlin (Tetrapurus audax)
Swordfish (Xiphias gladius)
Yellowfin Tuna (Thunnus albacares)
A-3
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
This Page Intentionally Left Blank
A-4
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
APPENDIX B PRIMARY HABITAT TYPES DESIGNATED AS ESSENTIAL FISH HABITAT
B PRIMARY HABITAT TYPES DESIGNATED AS ESSENTIAL FISH HABITAT
B.1 ESSENTIAL FISH HABITAT DESIGNATIONS BY PRIMARY HABITAT TYPE FOR EACH SPECIES/MANAGEMENT UNIT AND LIFE
STAGE
Table B-1: Western Pacific Regional Fishery Management Council Bottomfish Management Unit
WPRFMC Bottomfish Management Unit
Ma
La
Es
SB
Ss
Cr/Hs
Pr
J
A, J
Sz
DST
Pe
Comments
A
A, J, L, E
Adult depth of 0–250 m
A
A, J, L, E
Adult depth of 12–354 m
A
E, L
Adult depth of 0–265 m
E, L
Adult depth of 80 m
A
E, L
Adult depth of 3–180 m
A
E, L
Adult depth of 18–183 m
E, L
Adult depth of 20–380 m
A
E, L
Adult depth of 164–293 m
A
E, L
Juvenile depth of 65–100 m;
Adult depth of 100–200 m
A
E, L
Adult depth of 180–360 m
Shallow-water Species Complex (0–100 m)
Amberjack (Seriola dumerili)
Black jack (Caranx lugubris)
Blue stripe snapper (Lutjanus kasmira)
A
Giant trevally (Caranx ignoblis)
J
J
Gray jobfish (Aprion virescens)
A
Thicklip trevally (Pseudocaranx dentex)
A
A, J
J
J
A
J
A, J
J
A, J
A
A
A, J
Deep-water Species Complex (100–400 m)
Hawaiian grouper (Epinephelus quernus)
J
Longtail snapper (Etelis coruscans)
Pink snapper (Pristipomoides filamentosus)
A
J
Pink snapper (Pristipomoides sieboldii)
Silver jaw jobfish (Aphareus rutilans)
A
A
E, L
Adult depth of 6–100 m
Squirrelfish snapper (Etelis carbunculus)
A
A
E, L
Adult depth of 90–350 m
Yellow-barred snapper (Pristipomoides zonatus)
A
E, L
Adult depth of 100–200 m
Yellowtail snapper (Pristipomoides auricilla)
A
E, L
Adult depth of 180–270 m
Notes: Habitat: Mangrove (Ma), Lagoon (La), Estuarine (Es), Seagrass Beds (SB), Soft Substrate (Ss), Coral Reef/Hard Substrate (Cr/Hs), Patch Reefs (Pr), Surge Zone (Sz),
Deep-slope Terraces (DST), Pelagic/Open Ocean (Pe). Life History Stage: Egg (E), Larvae (L), Juvenile (J), Adult (A); m = meter(s), WPRFMC = Western Pacific Regional Fishery
Management Council
Source: Western Pacific Regional Fishery Management Council 2001, 2009
B-1
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table B-2: Western Pacific Regional Fishery Management Council Crustacean Management Unit
WPRFMC Crustacean Management Unit
Ma
La
Es
SB
Ss
Cr/Hs
Pr
Sz
DST
Pe
Comments
Spiny and Slipper Lobster Complex
Chinese slipper lobster (Parribacus antarticus)
A
Depth Distribution: 0–20 m
Ridgeback slipper lobster (Scyllarides haani)
A
Depth Distribution: 10–135 m
Spiny lobster (Panulirus penicillatus, Panulirus sp.)
All
A, J
All
All
All
L
Depth Distribution: 9–183 m
Kona Crab
Kona crab (Ranina ranina)
A
Adult depth of 24–115 m
Notes: Habitat: Mangrove (Ma), Lagoon (La), Estuarine (Es), Seagrass Beds (SB), Soft Substrate (Ss), Coral Reef/Hard Substrate (Cr/Hs), Patch Reefs (Pr), Surge Zone (Sz),
Deep-slope Terraces (DST), Pelagic/Open Ocean (Pe). Life History Stage: Egg (E), Larvae (L), Juvenile (J), Adult (A), Spawners (S); m = meter(s), WPRFMC = Western Pacific
Regional Fishery Management Council
Source: Western Pacific Regional Fishery Management Council 2001, 2009
Table B-3: Western Pacific Regional Fishery Management Council Crustacean Management Unit
WPRFMC Crustacean Management Unit
Ma
La
Es
SB
Ss
Cr/Hs
Pr
Sz
DST
Pe
Comments
Spiny and Slipper Lobster Complex
Chinese slipper lobster (Parribacus antarticus)
A
Depth Distribution: 0–20 m
Ridgeback slipper lobster (Scyllarides haani)
A
Depth Distribution: 10–135 m
Spiny lobster (Panulirus penicillatus, Panulirus sp.)
All
A, J
All
All
All
L
Depth Distribution: 9–183 m
Kona Crab
Kona crab (Ranina ranina)
A
Adult depth of 24–115 m
Notes: Habitat: Mangrove (Ma), Lagoon (La), Estuarine (Es), Seagrass Beds (SB), Soft Substrate (Ss), Coral Reef/Hard Substrate (Cr/Hs), Patch Reefs (Pr), Surge Zone (Sz),
Deep-slope Terraces (DST), Pelagic/Open Ocean (Pe). Life History Stage: Egg (E), Larvae (L), Juvenile (J), Adult (A), Spawners (S); m = meter(s), WPRFMC = Western Pacific
Regional Fishery Management Council
Source: Western Pacific Regional Fishery Management Council 2001, 2009
B-2
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table B-4: Western Pacific Regional Fishery Management Council Precious Corals Management Unit
WPRFMC Precious Corals Management Unit
Ma
La
Es
SB
Ss
Hs
Black coral (Antipathes dichomata)
A, J, S
Fern black coral (Antipathes ulex)
Pine black coral (Antipathes grandis)
Pr
Sz
DST
Pe
Comments
A, J, S
A, J, S
E, L
Depth Distribution: 30–110 m
A, J, S
A, J, S
A, J, S
E, L
Depth Distribution: 40–100 m
A, J, S
A, J, S
A, J, S
E, L
Depth Distribution: 45–110 m
Angel skin coral (Corallium secundum)
A, J, S
A, J, S
A, J, S
E, L
Depth Distribution: 350–475 m
Bamboo coral (Lepidisis olapa)
A, J, S
A, J, S
A, J, S
E, L
Depth Distribution: 300–400 m
Gold coral (Callogoria gilberti)
A, J, S
A, J, S
A, J, S
E, L
Depth Distribution: 300–1,500 m
Gold coral (Narella sp.)
A, J, S
A, J, S
A, J, S
E, L
Depth Distribution: 300–1,500 m
Gold coral (Calyprophora spp.)
A, J, S
A, J, S
A, J, S
E, L
Depth Distribution: 300–1,500 m
Gold coral (Acanella sp.)
A, J, S
A, J, S
A, J, S
E, L
Depth Distribution: 300–1,500 m
Hawaiian gold coral (Geraddia sp.)
A, J, S
A, J, S
A, J, S
E, L
Depth Distribution: 300–400 m
Midway deepsea coral (Corallium sp. nov)
A, J, S
A, J, S
A, J, S
E, L
Depth Distribution: 300–1,500 m
Pink coral (Corallium laauense)
A, J, S
A, J, S
A, J, S
E, L
Depth Distribution: 350–1,500 m
Red coral (Corallium regale)
A, J, S
A, J, S
A, J, S
E, L
Depth Distribution: 380–410 m
Shallow-water Species Assemblage (18–91 m)
Deep-water Species Assemblage (274–1,372 m)
Notes: Habitat: Mangrove (Ma), Lagoon (La), Estuarine (Es), Seagrass Beds (SB), Soft Substrate (Ss), Hard Substrate (Hs), Patch Reefs (Pr), Surge Zone (Sz), Deep-slope
Terraces (DST), Pelagic/Open Ocean (Pe); Life History Stage: Egg (E), Larvae (L), Juvenile (J), Adult (A), Spawners (S); m = meter(s), WPRFMC = Western Pacific Regional
Fishery Management Council
Source: Western Pacific Regional Fishery Management Council 2009
B-3
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table B-5: Western Pacific Regional Fishery Management Council Coral Reef Ecosystem Management Unit
WPRFMC Coral Reef Ecosystem Management Unit
Ma
La
Es
SB
Ss
Cr/Hs
Pr
Sz
DST
Pe
Acanthuridae (surgeonfishes)
Subfamily Acanthurinae (surgeonfishes)
Orange-spot surgeonfish (Acanthurus olivaceus)
Yellowfin surgeonfish (Acanthurus xanthopterus)
Convict tang (Acanthurus triostegus)
Eye-striped surgeonfish (Acanthurus dussumieri)
Blue-lined surgeon (Acanthurus nigroris)
Whitebar surgeonfish (Acanthurus leucopareius)
Whitecheek surgeonfish (Acanthurus nigricans)
White-spotted surgeonfish (Acanthurus guttatus)
Ringtail surgeonfish (Acanthurus blochii)
Brown surgeonfish (Acanthurus nigrofuscus)
Yellow-eyed surgeonfish (Ctenochaetus strigosus)
J
A, J, S
A, J, S
J
A, J, S
A, J, S
A, J, S
A, J
A, J
E, L
Subfamily Nasianae (unicornfishes)
Bluespine unicornfish (Naso unicornus)
Orangespine unicornfish (Naso lituratus)
Blacktongue unicornfish (Naso hexacanthus)
Whitemargin unicornfish (Naso annulatus)
Spotted unicornfish (Naso brevirostris)
Gray unicornfish (Naso caesius)
J
A, J, S
J
A, S
A, J, S
A, J, S
A, S
All
Balistidae (trigger fish)
Pinktail triggerfish (Melichthys vidua)
Black triggerfish (M. niger)
Picassofish (Rhinecanthus aculeatus)
Wedged Picassofish (R. rectangulus)
Bridled triggerfish (Sufflamen fraenatus)
J
A, J, S
J
A, J, S
A, J, S
A, S
L
Currently Harvested Coral Reef Taxa
B-4
A
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table B–5: Western Pacific Regional Fishery Management Council Coral Reef Ecosystem Management Unit (continued)
WPRFMC Coral Reef Ecosystem Management Unit
Carangidae (jacks)
Bigeye scad (Selar crumenophthalmus)
Mackerel scad (Decapterus macarellus)
Carcharhinidae
Grey reef shark (Carcharhinus amblyrhynchos)
Galapagos shark (Carcharhinus galapagenis)
Blacktip reef shark (Carcharhinus melanopterus)
Whitetip reef shark (Triaenodon obesus)
Ma
La
Es
SB
Ss
Cr/Hs
Pr
Sz
DST
Pe
A, J, S
A, J, S
A, J, S
J
A, J, S
A, J, S
A, J, S
A, J, S
All
E, L
A, J
A, J
A, J
J
A, J
A, J
A, J
A, J
A, J
A, J, S
A, J, S
J
A, J, S
A, J, S
A, S
E, L
A, J
A, J
A, J
Holocentridae (soldierfish/squirrelfish)
Bigscale soldierfish (Myripristis berndti)
Blotcheye soldierfish (Myripristis murdjan)
Bricksoldierfish (Myripristis amaena)
Yellowfin soldierfish (Myripristis chryseres)
Pearly soldierfish (Myripristis kuntee)
(Myripristis hexagona)
File-lined squirrelfish (Sargocentron microstoma)
Peppered squirrelfish (Sargocentron
punctatissimum)
Blue-lined squirrelfish (Sargocentron tiere)
Hawaiian squirrelfish (Sargocentron xantherythrum)
Saber squirrelfish (Sargocentron spiniferum)
Spotfin squirrelfish (Neoniphon spp.)
Kuhliidae (flagtails)
Hawaiian flagtail (Kuhlia sandvicensis)
A, J
B-5
A
E, L
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table B–5: Western Pacific Regional Fishery Management Council Coral Reef Ecosystem Management Unit (continued)
WPRFMC Coral Reef Ecosystem Management Unit
Ma
Kyphosidae (rudderfishes)
Highfin chub (K. cinerascens)
Lowfin chub (K. vaigiensis)
Labridae (wrasses)
Saddleback hogfish (Bodianus bilunulatus)
Razor wrasse (Xyrichtys pavo)
Ring-tailed wrasse (Oxycheilinus unifasciatus)
La
Es
A, J, S
A, J, S
J
J
SB
A, J
A, J
Cigar wrasse (Cheilio inermis)
Ss
Cr/Hs
Pr
Sz
A, J
A, J, S
A, J, S
A, J
A, J, S
A, J, S
A, J, S
A, J, S
E, L
A, J, S
A, J, S
A, J, S
A, J, S
E, L
A, J
Surge wrasse (Thalassoma purpureum)
Redribbon wrasse (Thalassoma quinquevittatum)
A, J
Sunset wrasse (Thalassoma lutescens)
A, J
Rockmover wrasse (Novaculichthys taeniourus)
A, J
Mullidae (goatfish)
Yellow goatfish (Mulloidichthys spp.)
(Mulloidichthys pfleugeri)
(Mulloidichthys vanicolensis)
(Mulloidichthys flavolineatus)
Banded goatfish (Parupeneus spp.)
(Parupeneus bifasciatus)
(Parupeneus cyclostomas)
(Parupeneus pleurostigma)
(Parupeneus multifaciatus)
Bandtail goatfish (Upeneus arge)
A, J
J
J
A
B-6
A, J
DST
Pe
All
E, L
A, J, S
A, J, S
A, J, S
A, J, S
A, J, S
A, J, S
A, J
A, J
A, J
A, J, S
E, L
E, L
A, J
A, J
E, L
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table B–5: Western Pacific Regional Fishery Management Council Coral Reef Ecosystem Management Unit (continued)
WPRFMC Coral Reef Ecosystem Management Unit
Octopodidae (octopuses)
Day octopus (Octopus cyanea)
Night octopus (Octupus ornatus)
Mugilidae (mullets)
Stripped mullet (Mugil cephalus)
Engel’s mullet (Moolgarda engeli)
False mullet (Neomyxus leuciscus)
Muraenidae (moray eels)
Yellowmargin moray (Gymnothorax flavimarginatus)
Giant moray (Gymnothorax javanicus)
Undulated moray (Gymnothorax undulatus)
Polynemidae (threadfins)
Threadfin/Moi (Polydactylus sexfilis)
Ma
La
Es
SB
Ss
Cr/Hs
Pr
A, J, S
All
A, J, S
All
All
All
All
J
A, J, S
A, J, S
J
A, J, S
A, J, S
A, J, S
A, J
A, J
A, J, S
A, J, S
J
A, J, S
Sphyraenidae (barracudas)
Heller’s barracuda (Sphyraena helleri)
Great barracuda (Sphyraena barracuda)
A, J
A, J, S
A, J, S
A, J, S
A, J, S
DST
Pe
All
L
A
A, J, S
A, J, S
Priacanthidae (bigeyes)
Glasseye (Heteropriacanthus cruentatus)
Bigeye (Priacanthus hamrur)
Scaridae (parrotfishes)
Parrotfishes (Scarus and Chlorurus spp.)
Stareye parrotfish (Calotomus carolinus)
A, J
Sz
A, J, S
E, L
E, L
A, J
A, J
A, J
A, J
A, J, S
A, J, S
J
A, J, S
A, J, S
E, L
A, J
E, L
E, L
A, S
All
Notes: Egg (E), Larvae (L), Juvenile (J), Adult (A), Spawners (S), Mangrove (Ma), Lagoon (La), Estuarine (Es), Seagrass Beds (SB), Soft substrate (Ss), Coral Reef/Hard
Substrate (Cr/Hr), Patch Reefs (Pr), Surge Zone (Sz), Deep-Slope Terraces (DST), Pelagic/Open Ocean (Pe); Life History Stage: Egg (E), Larvae (L), Juvenile (J), Adult (A),
Spawners (S); WPRFMC = Western Pacific Regional Fishery Management Council
Source: Western Pacific Regional Fishery Management Council 2009
B-7
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table B-6: Western Pacific Regional Fishery Management Council Pelagic Management Unit
WPRFMC Pelagic Management Unit
Ma
La
Es
SB
Ss
Cr/Hs
Pr
Sz
DST
Pe
Comments
Temperate Species
Striped marlin (Tetrapurus audax)
A, J, L, E
Depth Distribution: governed by temperature
stratification
Broadbill swordfish (Xiphias gladius)
A, J, L, E
Depth Distribution: surface to 1,000 m
Northern bluefin tuna (Thunnus thynnus)
A, J, L, E
No data
Albacore tuna (Thunnus alalunga)
A, J, L
Depth Distribution: surface to 380 m
Bigeye tuna (Thunnus obesus)
A, J, L, E
Depth Distribution: surface to 600 m
Mackerel (Scomber spp.)
A, J, L, E
No data
Sickle pomfret (Tatactichthys steindachneri)
A, J, L, E
Depth Distribution: surface to 300 m
Lustrous pomfret (Eumegistus illustris)
A, J, L, E
Depth Distribution: surface to 549 m
Yellowfin tuna (Thunnus albacares)
A, J, L, E
Depth Distribution: upper 100 m with
marked oxyclines
Kawakawa (Euthynnus affinis)
A, J, L, E
Depth Distribution: 36–200 m
Skipjack tuna (Katsuwonus pelamis)
A, J, L, E
Depth Distribution: surface to 263 m
Frigate tuna (Auxis thazard)
A, J, L, E
No data
Bullet tuna (Auxis rochei)
A, J, L, E
No data
Indo-Pacific blue marlin (Makaira nigricans)
A, J, L, E
Depth Distribution: 80–100 m
Black marlin (Makaira indica)
A, J, L, E
Depth Distribution: 457–914 m
Shortbill spearfish (Tetrapturus angustirostris)
A, J, L, E
Depth Distribution: 40–1,830 m
Sailfish (Istiophorus platypterus)
A, J, L, E
Depth Distribution: 10–20 m to 200–250 m
A, J, L, E
No data
A, J, L, E
No data
Tropical Species
Dolphinfish (Coryphaena hippurus)
A, J
Pompano dolphinfish (Coryphaena equiselas)
B-8
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
Table B–6: Western Pacific Regional Fishery Management Council Pelagic Management Unit (continued)
WPRFMC Pelagic Management Unit
Ma
La
Es
SB
Ss
Cr/Hs
Pr
Sz
DST
Pe
Comments
Tropical Species (continued)
Wahoo (Acanthocybium solandri)
A, J, L, E
Moonfish (Lampris guttatus)
Adult depth < 200 m
A, J
Depth Distribution: surface to 500 m
Escolar (Lepidocybium flavobrunneum)
A, J, L, E
Depth Distribution: surface to 200 m
Oilfish (Ruvettus pretiosus)
A, J, L, E
Depth Distribution: surface to 700 m
A, J
Depth Distribution: surface to 300 m
A, J
Depth Distribution: surface to 366 m
A, J
Depth Distribution: surface to 152 m
Bigeye thresher shark (Alopias superciliosus)
A, J
Depth Distribution: surface to 500 m
Shortfin mako shark (Isurus oxyrinchus)
A, J
Depth Distribution: surface to 500 m
Longfin mako shark (Isurus paucus)
A, J
No data
Salmon shark (Lamna ditropis)
A, J
Depth Distribution: surface to 152 m
A, J
Adult depth of 18–500 m
A, J
Adult depth of 37–152 m
Non-marketable Species Complex
Shark Species Complex
Crocodile shark (Pseudocarcharias kamoharai)
Thresher shark (Alopias vulpinus)
J
Pelagic thresher shark (Alopias pelagicus)
A
A
Silky shark (Carcharhinus falcirormis)
A
Oceanic whitetip shark (Carcharhinus
longimanus)
Blue shark (Prionace glauca)
A, J, L, E
Depth Distribution: surface to 152 m
Notes: Life History Stage: Egg (E), Larvae (L), Juvenile (J), Adult (A), Spawners (S). Habitat: Mangrove (Ma), Lagoon (La), Estuarine (Es), Seagrass Beds (SB), Soft Substrate (Ss), Coral
Reef/Hard Substrate (Cr/Hs), Patch Reefs (Pr), Surge Zone (Sz), Deep-slope Terraces (DST), Pelagic/Open Ocean (Pe); m = meter(s), WPRFMC = Western Pacific Regional Fishery
Management Council
Source: Western Pacific Regional Fishery Management Council 2001, 2009
B-9
Mariana Islands Training and Testing
Essential Fish Habitat Assessment
Final Report
This Page Intentionally Left Blank
B-10