* Your assessment is very important for improving the workof artificial intelligence, which forms the content of this project
Download a patient with haemorrhagic bullae
West Nile fever wikipedia , lookup
Hepatitis C wikipedia , lookup
Dirofilaria immitis wikipedia , lookup
Henipavirus wikipedia , lookup
Carbapenem-resistant enterobacteriaceae wikipedia , lookup
Anaerobic infection wikipedia , lookup
Herpes simplex wikipedia , lookup
Oesophagostomum wikipedia , lookup
Antiviral drug wikipedia , lookup
Marburg virus disease wikipedia , lookup
Hepatitis B wikipedia , lookup
Human cytomegalovirus wikipedia , lookup
Lymphocytic choriomeningitis wikipedia , lookup
Neonatal infection wikipedia , lookup
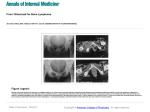
11. Heibl C, Trommet V, Burgstaller S, et al. Complications associated with the use of Port-a-Caths in patients with malignant or haematological disease: a single-centre prospective analysis. Eur J Cancer Care (Engl). 2010 Sep;19(5):676-81. 14. Wickham R, Purl S, Welker D. Long-term central venous catheters: issues for care. Semin Oncol Nurs. 1992 May;8(2):133-47. 15. Greene JN. Catheter-related complications of cancer therapy. Infect Dis Clin North Am. 1996 Jun;10(2):255-95. 12. Mermel LA, Allon M, Bouza E, et al. Clinical practice guidelines for the diagnosis and management of intravascular catheter-related infection: 2009 Update by the Infectious Diseases Society of America. Clin Infect Dis. 2009 Jul 1;49(1):1-45. 16. Freytes CO. Indications and complications of intravenous devices for chemotherapy. Curr Opin Oncol. 2000 Jul;12(4):303-7. 13. Kurul S, Saip P, Aydin T. Totally implantable venous-access ports: local problems and extravasation injury. Lancet Oncol. 2002 Nov;3(11):684-92. P H OTO Q U I Z A patient with haemorrhagic bullae H. Westdorp*, H.A.M. Sinnige Department of Internal Medicine, Jeroen Bosch Ziekenhuis, ’s-Hertogenbosch, the Netherlands, *corresponding author: tel.: +31 (0)73 553 3791, fax: +31 (0)73 553 3236, e-mail: [email protected] Figure 1. Haemorrhagic bullae in the left groin region Case report A 92-year-old man presented to our outpatient clinic with complaints of fatigue, muscle weakness and upper abdominal discomfort. There was no history of haematemesis, melaena, haematuria or epistaxis. In 2010, he was diagnosed with immune thrombocytopenic purpura (ITP) and was treated with prednisolone 1 mg/kg/ day (80 mg/day) with good response. The dose was tapered down slowly without disease relapse. Three weeks prior to presentation, the patient had discontinued the prednisone. Physical examination revealed purpura and petechiae, especially localised on the extremities. There was no lymphadenopathy or hepatosplenomegaly present. Laboratory findings showed a platelet count of less than 3 x 109/l. No other abnormal findings were noted. A bone marrow aspirate smear showed increased production of megakaryocytes without signs of dysplasia in all cell lines. The relapse of ITP was again treated with oral prednisolone 1 mg/kg/day, however, without effect. Also addition of intravenous immunoglobulin did not increase the platelet count. Because of the refractory character of the ITP we decided to give rituximab off-label. Five days after administration of the first dose of rituximab the patient developed haemorrhagic bullous lesions on the left hip ( figure 1). W H AT IS YO U R DIA G NOSIS ? See page 195 for the answer to the photo quiz. Bos, et al. Port-a-caths in oncology patients. m ay 2 012 , vo l . 7 0 , n o 4 189 A n s w e r t o ph o t o qu i z ( p a g e 1 8 9 ) A pa t i e n t w i t h h a e m o r r h ag i c b u l l a e Diagnosis The affected dermatomal pattern on one side of the body, in conjunction with the immunosuppressive treatment, made us think of an opportunistic infection with varicella zoster virus (VZV). The haemorrhagic aspect was suggested to be caused by the underlying thrombocytopenia, which initially persisted despite therapy with rituximab. A polymerase chain reaction on the bullous fluid was positive for VZV DNA, indicating an active VZV infection. ITP treatment with corticosteroids results in a complete response in 20% of adult patients. Most of the patients, however, will require a second-line treatment. For the clinical management of ITP we refer to a recent paper in this journal.1 Rituximab, a chimeric anti-CD20 monoclonal antibody, depletes CD20+ B-cells which results in low or even undetectable levels for two to six months, returning to pre-treatment levels within a year. Rituximab also induces complement-mediated cytotoxicity and dysfunction of CD4 T cells leading to abnormal cytotoxic T-cell-mediated responses.2 The above predisposes the patient to opportunistic infections. Data from 356 patients receiving rituximab monotherapy showed a 30% incidence rate of infectious events; 19% of patients had a bacterial infection, 10% viral infections, 1% fungal infections and 6% infections of unknown aetiology.3 One randomised controlled trial showed opportunistic viral infections which included three dermatomal herpes zoster infections and four localised herpes simplex infections. Isolated cases of other viral infections have been associated with rituximab, such as cytomegalovirus, West Nile virus, JC virus, BK virus and parvovirus. 4 Our patient was treated with valaciclovir 1000 mg twice a day. He responded very well to drug therapy and there was a major reduction of the bullae and herpes zoster rash. Additionally, after the third rituximab infusion, a significant increase in the platelet count to 70 x 109 cells/l was seen. Figure 1. Haemorrhagic bullae in the left groin region References 1. Schipperus M, Fijnheer R. New therapeutic options for immune thrombocytopenia. Neth J Med. 2011;69:480-5. 2. Kelesidis T, Daikos G, Boumpas D, Tsiodras S. Does rituximab increase the incidence of infectious complications? Int J Infect Dis. 2011;15:e2-16. 3. Kimby E. Tolerability and safety of Rituximab (MabThera). Cancer Treat Rev. 2005;31:456-73. 4. Aksoy S, Harputluoglu H, Kilickap S, et al. Rituximab-related viral infections in lymfoma patients. Leuk Lymphoma. 2007;48:1307-12. © Van Zuiden Communications B.V. All rights reserved. m ay 2 012 , vo l . 7 0 , n o 4 195