* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Modulation of visceral function by selective stimulation of the left
Sensory substitution wikipedia , lookup
Optogenetics wikipedia , lookup
Syncope (medicine) wikipedia , lookup
Psychoneuroimmunology wikipedia , lookup
Neural engineering wikipedia , lookup
Evoked potential wikipedia , lookup
Neuroregeneration wikipedia , lookup
Transcranial direct-current stimulation wikipedia , lookup
Microneurography wikipedia , lookup
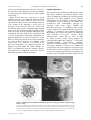
717 Exp Physiol 89.6 pp 717–725 Experimental Physiology Modulation of visceral function by selective stimulation of the left vagus nerve in dogs J. Rozman1 and M. Bunc2 1 2 ITIS d. o. o. Ljubljana, Center for Implantable Technology and Sensors, Lepi pot 11, Republic of Slovenia University of Ljubljana, School of Medicine, Institute of Pathophysiology, Zaloška 4, 1000 Ljubljana, Republic of Slovenia The superficial regions of the left vagus nerves of a dog were selectively stimulated with 39-electrode spiral cuffs having 13 circumferential groups of three electrodes (GTE) to modulate the function of the innervated internal organs and glands. Under general anaesthesia, the cuffs were chronically implanted around the nerve in the neck in two adult Beagle dogs and remained viable for 16 months. The regions were stimulated with biphasic, rectangular current pulses (2 mA, 200 µs, 20 Hz) delivered to the group of GTE lying close to the region innervating the specific internal organs or glands. The results showed that specific electrode configurations had actions on the heart (GTE 9), lungs (GTE 4) and pressure in the urinary bladder (GTE 1). It was also shown that GTE no. 10 significantly modified the endocrine function of the pancreas. The results of this study clearly demonstrate that internal organs and glands can be selectively stimulated via the selective stimulation of innervating superficial regions of the autonomous peripheral nerve. (Received 18 May 2004; accepted after revision 27 August 2004; first published online 13 September 2004) Corresponding author J. Rozman: ITIS d.o.o. Ljubljana, Center for Implantable Technology and Sensors, Lepi pot 11, 1000 Ljubljana, Republic of Slovenia. Email: [email protected] The left vagus nerve is an important route of information into the CNS (Berthoud & Neuhuber, 2000). One of its main functions is to monitor and control the activity of the internal organs and glands such as the heart, lungs, stomach, bladder and pancreas. Accordingly, there is a revival of interest in the influence of the vagal nerve fibres on these organs (Dixon et al. 1980; Roy et al. 1984; Woods & Porte, 1987; Berthoud et al. 1990; Schemann & Grundy, 1992; Levy, 1997). The principal functions of the heart are regulated by the sympathetic and parasympathetic divisions of the autonomic nervous system (Levy et al. 1993). The sympathetic nerves to the heart are facilitatory, whereas the parasympathetic (vagus) nerves are inhibitory (Mace & Levy, 1983; Levy, 1984). The vagus nerve can exert beat-by-beat control of cardiac function, while the onset and decay of the sympathetic effects are much more gradual (Levy et al. 1993). Yang & Levy (1992) have determined the influence of differences in the time of initiation of sympathetic and vagal stimulation on the cardiac autonomic interactions in anaesthetized dogs. Sympathetic stimulation alone increased the heart rate, vagal stimulation alone decreased C The Physiological Society 2004 the heart rate, and combined stimulation also decreased the heart rate (the vagal effects predominated). The actual pace set by the sino-atrial (SA) node, however, depends on the net effect of these antagonistic influences. It was shown by Dexter et al. (1992) that stimulation at precise times in successive cardiac cycles can elicit sinus arrhythmias. Moreover, Matheny & Shaar (1997) have suggested the technique of vagus nerve stimulation as a means of temporarily slowing down or arresting the heart during surgery. To increase the trial cycle length, Carlson et al. (1992) selectively stimulated parasympathetic nerve fibres that innervate the SA node. The activity of the autonomic innervation of the heart is coordinated by the cardiac control centre in the medulla oblongata which is, in turn, affected by higher brain areas and by sensory feedback from baroreceptors in the aorta and carotid arteries. Sensory nerve activity from the baroreceptors ascends via the vagus and glassopharyngeal nerves to the medulla. The left vagus nerve innervates the atrioventricular (AV) node. There can, however, be significant overlap in the anatomical distribution. Recently the central nervous system mediation of the baroreflex and the chronotropic responsiveness of the heart to vagal DOI: 10.1113/expphysiol.2004.027953 718 J. Rozman and M. Bunc efferent activity were independently assessed by recording the responses to electrical stimulation of the left vagus nerve (Ma et al. 2002). Reduced heart rate variability and baroreflex sensitivity are markers of cardiac vagal control and are used to assess activity in the autonomic nervous system (Levy & Martin, 1981; Huikuri et al. 1999). Both are powerful and independent indicators of adverse prognosis in patients with cardiac failure or myocardial infarction (Xenopoulos et al. 1996). Therefore, by using the method of selective stimulation of the left vagus nerve, heart rate variability can be altered towards normal and it is possible to elicit a variety of chronotropic and inotropic cardiac responses with or without alterations in systemic arterial pressure (Armour & Randall, 1985; Seidel et al. 1997; Setty et al. 1998; Sevre & Rostrup, 2001). The nerve fibres of the respiratory muscles arise from the vagus nerve and sympathetic chains. Dixon et al. (1980) have studied the distribution of the afferent and efferent nerves to the respiratory muscles in dogs anaesthetized with chloralose. The motor neurones that stimulate the respiratory muscles are controlled by two descending pathways controlling voluntary and involuntary breathing. The unconscious rhythmic control of breathing is influenced by input from the central chemoreceptors in the medulla and the peripheral chemoreceptors that respond to changes in the arterial partial pressure of CO2 (PaCO2 ), pH and the arterial partial pressure of O2 (PaO2 ). The peripheral chemoreceptors include the aortic bodies, located around the aortic arch, and the carotid bodies, located in each common carotid artery at the point where it branches into the internal and external carotid arteries. The aortic bodies send sensory information to the medulla in the vagus nerve (X); the carotid bodies stimulate sensory fibres in the glossopharingeal nerve (IX). Inspiration and expiration are produced by contraction and relaxation of the skeletal muscles in response to activity in the somatic motor neurones in the spinal cord. Their activity is controlled, in turn, by descending tracts from neurones in the respiratory control centres in the medulla and from neurones in the cerebral cortex. The rhythmicity centre, which controls automatic breathing and is located in the medulla, consists of interacting pools of neurones that fire either during inspiration (I neurones) or expiration (E neurones). The activity of the I and E neurones varies in a reciprocal way to produce a rhythmic pattern of breathing. Therefore, by using the method of selective stimulation of the corresponding superficial regions of the left vagus nerve, the information sent by the aortic bodies could be modified, and thus involuntary breathing could be externally controlled (Matran et al. 1991). The extrinsic motor innervation of the pancreas is provided by both the sympathetic and the parasympathetic Exp Physiol 89.6 pp 717–725 nervous system (Ahrén, 2000). A significant proportion of pancreatic neurones receives excitatory synaptic input from the vagal preganglionic axons (Miller, 1981). Stimulation of the vagal preganglionic fibres can directly affect pancreatic endocrine and exocrine secretion (Bergman & Miller, 1973; Berthoud & Powley, 1987). Ahrén & Taborsky (1986) studied the mechanisms of vagal nerve stimulation of glucagon and insulin secretion in halothane-anaesthetized dogs and concluded that vagal nerve stimulation produces a moderate increase of glucagon secretion and a marked increase of insulin secretion. The present study addresses the hypothesis that a certain superficial region of the peripheral autonomic nerve is composed mainly of fibres innervating a single internal organ or gland. Our study was aimed at demonstrating that stimulation of the autonomic nerve at one single site with nearby stimulating electrodes within the installed multielectrode spiral nerve cuff can potentially be used as a method for external modulation of function of the internal organs and glands innervated by the corresponding selectively stimulated superficial region. Methods Multi-electrode nerve cuff A cuff was made by bonding two 0.1 mm thick silicone sheets together. One sheet, stretched and fixed in that position, was covered with a layer of adhesive (MED-1511, NuSil, Carpinteria, CA, USA). A second unstretched sheet was placed on the adhesive and the composite was compressed to a thickness of 0.3 mm until the whole curing process was completed. When released, the composite curled into a spiral tube as the stretched sheet contracted to its natural length. The diameter of the cuff was related to the amount of stretch; the greater the stretch, the smaller the diameter. Thirty-nine rectangular electrodes with a width of 0.6 mm and length of 1.5 mm were made of 50 µm thick platinum ribbon (99.99% purity) and connected to lead wires (AS 631, Cooner Wire, Chatsworth, CA, USA) that were mounted on a third silicone sheet with a thickness of 0.1 mm. They were arranged in three parallel groups each containing 13 electrodes at a distance of 0.5 mm. The distance between the spiral groups was 6 mm. Each electrode marked with the same number within each of the three parallel spiral groups had the same position. Accordingly, 13 groups of three electrodes (GTEs) in the same line in a longitudinal direction were formed. All electrodes of the central and two outer groups were then connected to the corresponding lead wires. The silicone sheet with the arranged electrodes was then bonded on the inner side of the mechanically opened cuff. The length of the cuff was optimized so that the surface of the nerve covered by the spiral cuff would be as small as possible to prevent damage associated with a reduced blood supply C The Physiological Society 2004 Exp Physiol 89.6 pp 717–725 Left vagus nerve modulates visceral function and excessive mechanical trauma of the nerve. Therefore, the cuff, with an inner diameter of 2.5 mm, was trimmed to a length of 18 mm as shown in the inset in the top right corner of Fig. 1. Finally, all lead wires were connected to a special common connector to be implanted within the lateral subcutaneous tissue of the neck for the time between the experimental sessions. To connect the common connector to the outputs of the stimulator a special cable was developed. At one end of the cable to be connected to the common connector was a switching module designed to fit its pins. The switching module permitted a certain GTE to be connected to the stimulator individually or in combination with other GTEs. In the switching module a selective ‘quasi-bipolar’ stimulating configuration was produced. Namely, the two outer electrodes of a certain GTE to be connected as anodes to one end of a stimulator were short-circuited, while the corresponding central electrode was to be connected as a cathode to the other end. Furthermore, the common connector was designed to permit simple and reliable multiple use. This is very important because the common connector was designed to be reimplanted several times between individual experiments without any damage. Surgical implantation The experiment was performed on two Beagle dogs. Under fully aseptic conditions gas-sterilized (ethylene oxide) cuffs were implanted according to the following protocol approved by the ethics committee at the Veterinary Administration of the Republic of Slovenia, Ministry of Agriculture, Forestry and Food. The animals were premedicated with medetomidine, 40 µg kg−1 i.m. (Domitor, Orion Corp., Espoo, Finland) and methadone, 0.2 mg kg−1 s.c. (Heptanon, Pliva, Zagreb, Croatia). Induction was performed with propofol, 1.0– 2.0 mg kg−1 i.v. (Diprivan, Zeneca Pharmaceuticals Ltd, Macclesfield, UK). General anaesthesia was maintained with isoflurane, 0.8–1.5 vol.% (Forane, Abbott Laboratories Inc., Abbott Park, IL, USA) in 100% O2 . When necessary, during surgery analgesia was sustained with ketamine, 0.5–2.0 mg kg−1 i.v. (Ketamine, Veyx-Pharma GmbH, Schwarzenborn, Germany). Antibiotics (cefazolin, 20 mg kg−1 i.v.; Cefamezin, Krka, Novo Mesto, Republic of Slovenia) were administered perioperatively. The room temperature was kept between 23.4 and 24.4◦ C and the temperature of the skin of the neck was also continuously monitored. According to our model, the cuff was installed around the left vagus nerve in Figure 1. Radiograph of the implanted 39-electrode spiral cuff, showing its position on the left vagus nerve of a dog Inset in the top right corner shows the 39-electrode spiral cuff (1) with the subcutaneous common connector (2) and the switch module (3). Inset in the lower left corner shows a model of the reconstructed cross-sectional geometry of the left vagus nerve, within the spiral cuff, with the indicated GTEs no. 9, no. 4, no. 1 and no. 10, close to the superficial regions innervating the heart (6), lung (5), bladder (4) and pancreas (7). C The Physiological Society 2004 719 720 J. Rozman and M. Bunc the neck as shown in Fig. 1. The leads of the implanted cuff were routed and fixated to the corresponding common connector under the skin at the lateral side of the neck. Finally, the incision was closed and the animal allowed to recover. Analgesia during the early recovery period was provided by methadone, 0.3–0.5 mg kg−1 s.c. three times daily. Tramadol, 8.0 mg kg−1 s.c. three times daily (Tramal, Grünenthal GmbH, Stolberg, Germany) was administered for a further 2 days. Selective stimulation of the left vagus nerve Two months after implantation, the first stimulation session was performed. However, to obtain initial data about the level of glucagon, insulin and C-peptide in the blood, samples from the femoral artery were drawn before any experimental activities. During the stimulation sessions, the dogs were anaesthetized according to the aforementioned procedure. After taking the subcutaneously implanted common connector belonging to the cuff out of the body, it was thoroughly cleaned and dried. The common connector was then placed on the cleaned skin close to the surgical site. Then the common connector and the surgical site were covered with self-adhesive sterile surgical foil during the entire experiment. The connection of the common connector to the outputs of the stimulator was made simply by perforating the self-adhesive sterile surgical foil with the pins of the switch module and inserting them in the common connector. It was crucial for the stimulations that the connection permitted reliable mechanical and galvanical connections. Moreover, the common connector and the surgical site were insulated from the ambient atmosphere. The switches of the switching module were alternately turned on so as to connect the electrodes in a certain GTE to the stimulator. Each of the 13 GTEs was then denoted by a consecutive number, as shown in the inset in the bottom left corner of Fig. 1, which is a geometric model of the cuff fitted on the left vagus nerve. However, considering the dimensions of the nerve and the cuff it was expected that certain GTEs remained out of contact with the superficial regions of the nerve. The relative positions of GTEs closest to the superficial regions of the nerve innervating the heart, the lungs and the bladder were determined experimentally. This was done by delivering stimulating pulses quasi-bipolarly to all 13 GTEs within the cuff. The superficial regions on the circumference of the nerve innervating the aforementioned organs that were in contact with the corresponding GTE were selectively stimulated using rectangular, biphasic, charge-balanced current pulses with an intensity of 1.3–2.0 mA and a frequency of 20 Hz. The GTEs that elicited the largest measurable response in one Exp Physiol 89.6 pp 717–725 of the aforementioned organs were indicated as relevant to the investigation. Accordingly, when stimuli were delivered to the GTE that was in contact with the superficial region of the vagus nerve innervating the heart, the heart rate began to fall. Thus the defined superficial region was denoted as region 6, shown in the inset in the bottom left corner of Fig. 1. The current amplitude was then adjusted so that a continuous cessation of heart rate was obtained. This GTE was then indicated as relevant for selective stimulation of the heart. To quantify the changes in heart rate produced by selective stimulation of the corresponding superficial region of the left vagus nerve the first, second and third derivative of ECG were recorded. In our study, this chronotropic response to left vagus nerve stimulation was evaluated simply by recording the ECG using stainless-steel hypodermic needles inserted percutaneously in the animal’s limbs. The ECG signals were delivered to a custom-designed differential amplifier and to a DigiPack 1200 high-performance data acquisition system connected to a PC, featuring a Digidata 1200A high-performance data acquisition system designed and manufactured by Axon Instruments and high-speed data acquisition software. Furthermore, to quantify the changes in baroreflex sensitivity, which is another marker of cardiac vagal control, arterial and vein (CVP) blood pressures were measured using a disposable pressure transducer system for invasive blood pressure monitoring (model DPT6000; Smiths Medical Deutschland GmbH, Kirchseeon, Germany; company section: pvb-Critical Care). Furthermore, when stimuli were delivered to the GTE that was in contact with the superficial region of the nerve innervating the respiratory muscles, the breathing rate was reduced and its character was altered. Thus defined, the superficial region was denoted as region 5, shown in the inset in the bottom left of Fig. 1. The current amplitude was then adjusted so that a continuous cessation of breathing was obtained. This GTE was then indicated as relevant for selective stimulation of the lungs. To validate the changes in the rhythm and character of breathing produced by selective stimulation of the corresponding superficial region of the left vagal nerve, variations of the circumference of the thorax were measured. This was done using a metal belt instrumented with a customdesigned force transducer, which was mounted around the chest. The signals obtained from the force transducer were delivered to a custom-designed bridge amplifier and to the data acquisition system connected to the PC. When stimuli were delivered to the GTE that was in contact with the superficial region of the vagus nerve innervating the bladder, the pressure in the bladder began to fall. Thus defined, the superficial region was denoted as region 4, shown in the inset in the bottom left of Fig. 1. The current amplitude was then adjusted so that maximum reduction in pressure was obtained. This GTE C The Physiological Society 2004 Exp Physiol 89.6 pp 717–725 Left vagus nerve modulates visceral function was then indicated as relevant to selective stimulation of the bladder. To measure the changes of pressure in the bladder a special catheter inserted through the urethra and connected to a disposable pressure transducer system for invasive blood pressure monitoring (model DPT6000; Smiths Medical Deutschland GmbH) was used. The signal obtained from the pressure transducer was delivered to a custom-designed bridge amplifier and to the aforementioned data acquisition system connected to the PC. Finally, the relative position of the GTE closest to the superficial region of the nerve innervating the pancreas was determined indirectly. This was done by delivering a train of the aforementioned stimuli for periods of 10 s to each of the 13 GTEs within the cuff so that all the superficial regions were selectively stimulated. Each stimulation session was proceeded by a pause of 5 s during which the existing GTE was disconnected from the stimulator and the next one was connected. Thus defined, the superficial region was denoted as region 7, shown in the inset in the bottom left of Fig. 1. By the end of each pause, blood samples from the femoral artery were drawn. They were placed on ice, centrifuged at 4◦ C immediately after the end of the experiments, and the plasma was separated and frozen at −20◦ C until radioimmunoassay (RIA). Plasma glucose was regularly estimated by the glucose oxidase method. Hematocrit was determined at regular intervals throughout the experiments. In this way the GTE that elicited the highest rate of insulin secretion was determined. Kits from Linco Research, Inc. (St Charles, MI, USA) were used for RIA. The first kit was the Canine C-Peptide radioimmunoassay kit for quantitative determination of canine C-peptide in serum, plasma and other biological media. The second kit was the Human Insulin Specific kit for quantitative determination of insulin in serum, plasma and other biological media. The third kit was the Glucagon Radioimmunoassay kit, which uses an antibody specific for pancreatic glucagon. All of the aforementioned kits are for research purposes only. After the last experiment, both animals were killed using the veterinary drug T61 (Hoechst, Frankfurt, Germany). The whole study was performed within the time frame of 3 years. The first year was spent on the development of the model, development and fabrication of the cuffs, and for organization of the materials, apparatus and facilities. Modelling of the electric field generated in the superficial region of the nerve by a certain GTE was based on the geometrical model, dissected through the longitudinal axis of the nerve and selected GTE. To determine the potential distribution in the superficial regions the Finite Element Method was used. After the cuffs were implanted, the next 2 months were spent allowing the animals to fully recover from anaesthesia and tissue healing. In both animals the cuff remained implanted for a time period of 2 years, but C The Physiological Society 2004 721 during the last 6 months the nerves were not stimulated. Within the remaining time period of 16 months, four sessions in each animal were performed. They were repeated about every 5 months. In this very complex study we mainly tested selectivity and reproducibility of stimulation and measured only basic parameters of modified physiological function in stimulated organs and glands. Owing to the aforementioned complexity of the experimental work and the extent of the expected outcomes of the study a reliable statistical model could not be developed. Besides, in each animal unique conditions were established, so only selectivity and intra-individual reproducibility could be tested. Accordingly, owing to this lack of a reliable statistical model, we present the results obtained in the last of four sessions conducted in the second implanted animal. Results The four records of responses elicited in the heart, the lungs and the bladder by the stimuli delivered for 10 s on GTE no. 9, which were closest to region 6, innervating the heart, are presented in Fig. 2. Trace 1, representing breathing frequency, shows only a minor disturbance in breathing during the stimulation period. Trace 2, representing the changes in pressure within the bladder, shows that vagal stimulation caused no measurable change. Vagal stimulation did induce a decrease in arterial pressure and heart rate. In fact, the heart rate response to left vagus nerve stimulation was nearly abolished in the case when the stimulation pulses were 2 mA. In particular, trace 4 of the figure, which represents arterial pressure, shows a rapid fall during the time period in which current stimuli were 2 mA. Trace 3, representing the ECG, shows a complete cessation of the ECG recorded within the same period of time. Similarly, Fig. 3 shows four traces recorded as responses elicited in the heart, the lungs and the bladder by stimuli delivered for 10 s to GTE no. 4, which was closest to region 5, innervating the respiratory muscles. Trace 1, representing breathing frequency, shows complete cessation of breathing during the time period in which the magnitude of the current stimuli was 2 mA. Trace 2, representing changes in pressure within the bladder, shows no measurable change. Similarly, trace 3, representing ECG, does not show any disturbance within the stimulation period. Trace 4, representing arterial pressure, does not show any change in pressure. The four records of responses elicited in the heart, the lungs and the bladder by stimuli delivered for 10 s on GTE no. 1, which was closest to region 4, innervating the bladder, are presented in Fig. 4. Trace 1, representing breathing frequency, does not show any disturbance in breathing during the stimulation period. Similarly, as can J. Rozman and M. Bunc 2 0 34.8 34.6 Trace 2 0 1 Trace 3 Trace 4 0 5 10 15 20 Time (s) Figure 2. Physiological responses of heart, lungs, and bladder to stimulation with the group of 3 electrodes no. 9 Trace 1 shows the physiological response of the lungs, represented by breathing frequency. Trace 2 illustrates internal bladder pressure. Traces 3 and 4 illustrate the response of the heart as demostrated by ECG and arterial blood pressure, respectively. some overlap occurred and some disturbance in the heart rate was also observed. Discussion This work shows for the first time that a multi-electrode spiral cuff chronically implanted around the left vagus nerve of a dog can be used to modulate the function of the internal organs and glands selectively, via selective stimulation of the corresponding superficial regions of the nerve. Normal cardiac rhythm is controlled by pacemaker activity, which is spontaneously generated by SA nodal cells. The rhythm is strongly influenced by autonomic 34.6 34.8 0 2 Trace 1 Trace 2 0 1 Trace 3 Trace 4 100 ECG (mV) Bladder pressure Breathing (a.u) (mmHg) be seen in traces 3 and 4, respectively, vagal stimulation did not induce any disturbance in heart rate or in arterial pressure. Trace 2, representing the change in pressure within the bladder, however, shows a decrease in pressure from 34.8 mmHg at the beginning of stimulation to 34.74 mmHg at the end of stimulation. Figure 5 shows the response of the pancreas on consecutive selective stimulation of the superficial regions of the nerve using all 13 groups of three electrodes. The results of RIA of the blood samples drawn from the femoral artery after stimulation using each of the 13 GTEs showed that only GTE no. 10, which was close to region 7, caused a maximum increase in insulin and minor increase in glucagon secretion. Since GTE no. 10 is close to GTE no. 9, ABP (mmHg) Exp Physiol 89.6 pp 717–725 Trace 1 100 ABP (mmHg) ECG (mV) Bladder pressure Breathing (a.u) (mmHg) 722 0 5 10 15 Time (s) 20 Figure 3. Physiological responses of heart, lungs, and bladder to stimulation with the group of 3 electrodes no. 4 Traces as in Fig. 2. C The Physiological Society 2004 Left vagus nerve modulates visceral function 723 0 2 Trace 1 34.6 34.8 Bladder pressure Breathing (a.u) (mmHg) Exp Physiol 89.6 pp 717–725 Trace 2 Figure 4. Physiological responses of heart, lungs, and bladder to stimulation with the group of 3 electrodes no. 1 Traces as in Fig. 2. 1 0 Trace 4 100 ABP (mmHg) ECG (mV) Trace 3 0 nerves, with the vagus nerve being dominant over sympathetic influences at rest. Abnormal cardiac rhythms may occur when the SA node fails to function normally or when normal conduction pathways are not followed. Vagal activation decreases pacemaker rate, which decreases the slope of repolarization, thereby increasing the time to reach threshold. Vagal activity can also hyperpolarize the pacemaker cells during repolarization, which results in a long time to reach threshold voltage. According to the results of selective stimulation of the heart, represented by trace 2 in Fig. 2, showing the ECG, we could assume that the pacemaker cells were hyperpolarized owing to the intensity of applied current pulses. This illustrates that one difficulty observed during stimulation sessions was that the difference in the current level between threshold excitation and maximum recruitment was rather low. Consequently, the heart rate as shown by the R waves remained constant until it was abruptly terminated at the onset of stimulation and returned unchanged at the end of stimulation. The results of selective stimulation of the respiratory muscles represented by trace 4 in Fig. 3, showing the rhythm of breathing, shows the complete cessation of breathing during the stimulation period. However, to obtain more detailed information on the possibility of modifying lung function, different values of stimulation parameters such as current intensity and frequency of stimuli should be employed. The results of selective stimulation of the bladder, represented by trace 3 in Fig. 4, showing pressure within the bladder, reveals a minor decrease in pressure during the stimulation period. In this case, however, a degree of selectivity and the rate of intra-individual reproducibility are not so great as for heart and lung parameters. Finally, the results of selective stimulation of the pancreas, shown in Fig. 5 by the bars representing the C The Physiological Society 2004 5 10 15 20 Time (s) secretion of insulin and glucagon when different GTEs were used, reveal that a considerable increase in insulin and minor increase in glucagon secretion could be elicited. The results obtained in both animals show almost the same high degree of selectivity and high rate of intra-individual reproducibility in all stimulation sessions performed. With respect to selectivity, the problem of excitational overlap in the regions between the neighbouring electrodes and dislodgement of the spiral cuff during any significant movement remained unsolved. The results of the study could be used in various animal and especially human basic studies concerning the neurophysiology of the internal organs and endocrine glands and their relation to bodily changes and disease. Ultimately, the method could be used for both stimulation and recording of electroneurograms (ENGs) in different combinations when implanted around the aforementioned nerves. Figure 5. Physiological response of the pancreas elicited by stimulation with the group of 3 electrodes no. 10 The physiological responses of the pancreas are represented by changes in the secretion of insulin and glucagon. 724 J. Rozman and M. Bunc The methodology as well as the pertaining technological solutions developed could be used in transfer of the animal model to the human model of curing diseases of cardiovascular system and potentially diabetes mellitus. Namely, it is well known that cardiovascular disease is the leading cause of death in diabetes mellitus. Moreover, the methodology, and especially spiral cuffs, could be successfully used in the transfer of this animal model to human vagus nerve stimulation (VNS; Murphy & Patil, 2003). However, the condition for the potentially successful transfer of the model for selectively modifying the function of the internal organs and glands in man is a good functional result demonstrated by these animal experiments, even over a prolonged period of time. VNS is already established as a clinically useful anticonvulsant in drug-resistant patients with epilepsy, and it may have potential as an antidepressant treatment. It seems logical to suggest that selective VNS delivered with different parameters from those commonly used for epilepsy might produce different CNS effects that would in turn broaden the clinical indications. Therefore, this therapy could be significantly improved by the possibility of selective stimulation of the human left vagus nerve with the implanted multielectrode spiral cuff (Murphy & Patil, 2003). One application already being studied is the use of VNS in slowing the progress of coronary artery disease (Zamotrinsky et al. 2001). Another potential application of VNS is the control of Parkinsonian and essential tremor. Gastric secretory studies combined with selective vagal stimulation may be useful in patients with suspected gastric hypersecretion or with gastroparesis (Berthoud et al. 2002). Since recent behavioural and physiological evidence indicates that the vagus nerves conduct sensory information from the uterus to the brainstem, conducted information could also be modified using the selective VNS (Collins et al. 1999). The long-range goal of our research will be to understand how the various branches of the autonomic nervous system regulate the function of the internal organs and glands. More precisely, in our near future work, the physiological function of internal organs and glands modified via the selective stimulation of the superficial regions in the innervating sympathetic and parasympathetic nerves will be focused on a certain organ and studied in detail. References Ahrén B (2000). Autonomic regulation of islet hormone secretion – implications for health and disease. Diabetologia 43, 393–410. Ahrén B & Taborsky GJ (1986). The mechanism of vagal nerve stimulation of glucagon and insulin secretion in the dog. Endocrinology 118, 1551–1557. Exp Physiol 89.6 pp 717–725 Armour JA & Randall WC (1985). Rebound cardiovascular responses following stimulation of canine vagosympathetic complexes or cardiopulmonary nerves. Can J Physiol Pharmacol 63, 1122–1132. Bergman RN & Miller RE (1973). Direct enhancement of insulin secretion by vagal stimulation of the isolated pancreas. Am J Physiol 225, 481–486. Berthoud H-R, Fox EA & Powley TL (1990). Localization of vagal preganglionics that stimulate insulin and glucagon secretion. Am J Physiol 258, R160–R168. Berthoud HR, Hennig G, Campbell M, Volaufova J & Costa M (2002). Video-based spatio-temporal maps for analysis of gastric motility in vitro: effects of vagal stimulation in guinea-pigs. Neurogastroenterol Motil 14, 677–688. Berthoud HR & Neuhuber WL (2000). Functional and chemical anatomy of the afferent vagal system. Auton Neurosci 85, 1–17. Berthoud H-R & Powley TL (1987). Characteristics of gastric and pancreatic responses to vagal stimulation with varied frequencies: evidence for different fiber calibers? J Auton Nerv Syst 19, 77–84. Carlson MD, Geha AS, Hsu J, Martin PJ, Levy MN, Jacobs G & Waldo AL (1992). Selective stimulation of parasympathetic nerve fibers to the human sinoatrial node. Circulation 85, 1311–1317. Collins JJ, Lin CE, Berthoud HR & Papka RE (1999). Vagal afferents from the uterus and cervix provide direct connections to the brainstem. Cell Tissue Res 295, 43–54. Dexter F, Yang T & Levy MN (1992). Analysis of vagally induced sinus arrhythmias. J Theor Biol 159, 507–511. Dixon M, Jackson DM & Richards IM (1980). A study of the afferent and efferent nerve distribution to the lungs of dogs. Respiration 39, 144–149. Huikuri HV, Makikallio T, Airaksinen KE, Mitrani R, Castellanos A & Myerburg RJ (1999). Measurement of heart rate variability: a clinical tool or a research toy? J Am Coll Cardiol 34, 1878–1883. Levy MN (1984). Cardiac sympathetic–parasympathetic interactions. Fed Proc 43, 2598–2602. Levy MN (1997). Neural control of cardiac function. Baillieres Clin Neurol 6, 227–244. Levy MN & Martin PJ (1981). Neural regulation of the heart beat. Annu Rev Physiol 43, 443–453. Levy MN, Yang T & Wallick DW (1993). Assessment of beatby-beat control of heart rate by the autonomic nervous system: molecular biology techniques are necessary, but not sufficient. J Cardiovasc Electrophysiol 4, 183–193. Ma X, Abboud FM & Chapleau MW (2002). Analysis of afferent, central, and efferent components of the baroreceptor reflex in mice. Am J Physiol 283, R1033–R1040. Mace SE & Levy MN (1983). Autonomic nervous control of heart rate: sympathetic–parasympathetic interactions and age related differences. Cardiovasc Res 17, 547–552. Matheny RG & Shaar CJ (1997). Vagus nerve stimulation as a method to temporarily slow or arrest the heart. Ann Thorac Surg 63 (Suppl. 6), S28–S29. Matran R, Alving K & Lundberg JM (1991). Differential bronchial and pulmonary vascular responses to vagal stimulation in the pig. Acta Physiol Scand 143, 387–393. C The Physiological Society 2004 Exp Physiol 89.6 pp 717–725 Left vagus nerve modulates visceral function Miller RE (1981). Pancreatic neuroendocrinology: peripheral neural mechanisms in the regulation of the Islets of Langerhans. Endocr Rev 2, 471–494. Murphy JV & Patil A (2003). Stimulation of the nervous system for the management of seizures: current and future developments. CNS Drugs 17, 101–115. Roy MW, Lee KC, Jones MS & Miller RE (1984). Neural control of pancreatic insulin and somatostain secretion. Endocrinology 115, 770–775. Schemann M & Grundy D (1992). Electrophysiological identification of vagally innervated enteric neurons in guinea pig stomach. Am J Physiol 263, G709–G718. Seidel H, Herzel H & Eckberg DL (1997). Phase dependencies of the human baroreceptor reflex. Am J Physiol 272, H2040–H2053. Setty AB, Vaughn BV, Quint SR, Robertson KR & Messenheimer JA (1998). Heart period variability during vagal nerve stimulation. Seizure 7, 213–217. Sevre K & Rostrup M (2001). Measurements of heart rate variability and baroreflex sensitivity. Tidsskr Nor Laegeforen 121, 3059–3064. (In Norwegian) C The Physiological Society 2004 725 Woods SC & Porte D Jr (1987). Neural control of the endocrine pancreas. Physiol Rev 54, 596–619. Xenopoulos NP, Braden GA & Applegate RJ (1996). Severe right heart failure in a patient with Grave’s disease. Clin Cardiol 19, 903–905. Yang T & Levy MN (1992). Sequence of excitation as a factor in sympathetic–parasympathetic interactions in the heart. Circ Res 71, 898–905. Zamotrinsky AV, Kondratiev B & de Jong JW (2001). Vagal neurostimulation in patients with coronary artery disease. Auton Neurosci 88, 109–116. Acknowledgements This work was financed by Research Grant no. J2-0542 from the Ministry of Education, Science and Sport, Ljubljana, Republic of Slovenia and supported in part by the European Community’s Human Potential Programme under contract no. HPRN-CT2000-00030 [NeuralPRO].