* Your assessment is very important for improving the work of artificial intelligence, which forms the content of this project
Download Atomic Structure Scientists
Survey
Document related concepts
Transcript
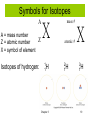
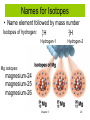
History of the Atom Chemistry Mrs. Herrmann Democritus 460-370 BC • Atomos: The point at which matter can no longer be subdivided. John Dalton 1766-1844 Dalton’s Atomic Theory 1803 • Elements consisted of tiny particles called atoms. • All atoms of an element were identical . • Atoms of each element were different from one another. (they had different masses, sizes, properties, etc…) • Compounds consist of atoms of different elements combined together. • Compounds have constant composition because they contain a fixed ratio of atoms. • Chemical reactions involve the rearrangement of combinations of those atoms. Dalton’s Atomic Model • Atoms were tiny, indivisible, indestructive particles and each one had a certain mass, size and chemical behavior. • The picture to the right illustrates Dalton’s idea of an atom. J.J. Thomson 1856-1940 • April 30, 1897 announced that cathode rays were made of negatively charged particles which he called “corpuscles”. These particles would later be called electrons. • Determined charge to mass ratio of the electron using the cathode ray tube, thus known for the negative one charge associated with the electron and the idea that atoms could be divided into smaller particles. • Won Nobel Prize in Physics in 1906 for discharge of electricity Thomson Experiment 1897, Joseph John Thomson: • discovered negatively charged particles--Electrons • calculated mass-to-charge ratio of electrons © 2013 Pearson Education, Inc. Chapter 3 7 Mass of Electron • Mass of electron calculated to be • • • 9.1 x 10–28 g 1837 times smaller than a proton 0.0005 amu (0 amu) • Mass of proton • 1.7 x 10 –24 g 0.0000000000000000000000017 • 1 amu g 0.00000000000000000000000000091 g 0.00000000000000000000000170091 g Chapter 3 8 Thomson’s Plum Pudding Atomic Model • Believed the model of an atom was much like plum pudding. The plums would be the electrons and the pudding would be the the sphere of positive charge • Ernest Rutherford 1871-1937 • Born in New Zealand • There were 12 children in his family: 7 boys and 5 girls • Age 16 first attended college • Most known for the discovery of the nucleus through the Gold Foil Experiment • Coined the term proton for the positive particles of the nucleus Model Explaining Rutherford’s Experiment Atoms • have positively charged nucleus • have a tiny, very dense nucleus • are made mostly of empty space Chapter 3 12 Structure of the Atom • Rutherford suggested: – Most mass in nucleus – positive charge in nucleus • James Chadwick, English physicist – discovered neutrons in 1932 • Nucleus: protons and neutrons – Neutrons have mass but no charge – Very small compared to rest of atom Chapter 3 13 James Chadwick 1891-1974 Subatomic Particles © 2013 Pearson Education, Inc. Chapter 3 15 Atomic Structure Atomic number = number protons in atom • whole number on periodic table • atoms of same element have same atomic # • For a neutral atom, #protons = #electrons 16 Chapter 3 Isotopes mass number = number protons + neutrons Mass # is NOT on periodic table! Chapter 3 17 Isotopes Isotopes have the same atomic number, but have different mass numbers (same number of protons, but different number of neutrons). © 2013 Pearson Education, Inc. Chapter 3 18 Symbols for Isotopes A A = mass number Z Z = atomic number X = symbol of element Isotopes of hydrogen: X 1H 1 Chapter 3 mass # atomic # 2H 1 X 3H 1 19 Names for Isotopes • Name element followed by mass number 1H 2H Isotopes of hydrogen: 1 1 Hydrogen-1 Hydrogen-2 Mg isotopes: magnesium-24 magnesium-25 magnesium-26 Chapter 3 20 Practice atomic number for cobalt? 27 How many protons in phosphorus atom? 15 How many electrons in tungsten atom? 74 mass # of sodium isotope with 12 neutrons? 11 + 12 = 23 Write isotope symbol for tin with mass number 119. 119 50Sn Chapter 3 50 p 50 e69 n 21 •